Region:Asia
Author(s):Dev
Product Code:KRAA6067
Pages:86
Published On:January 2026
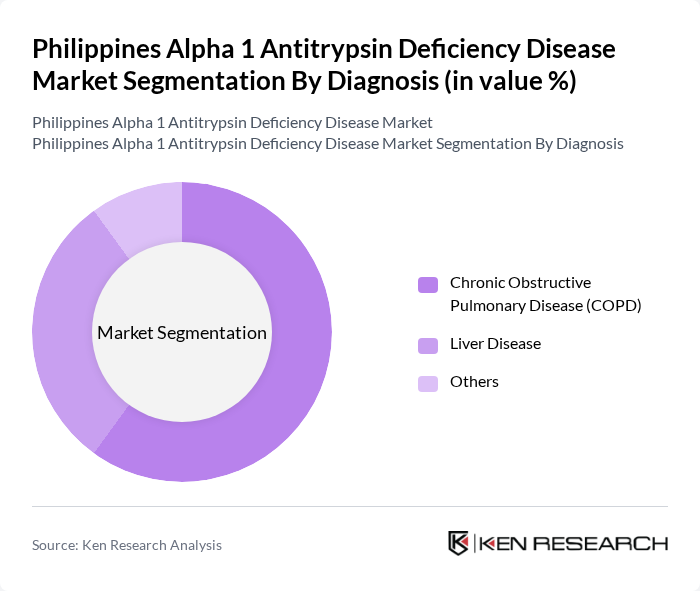
By Diagnosis:The diagnosis of Alpha 1 Antitrypsin Deficiency is crucial for effective management and treatment. The primary subsegments include Chronic Obstructive Pulmonary Disease (COPD), Liver Disease, and Others. COPD is the leading diagnosis due to its high prevalence among patients with this deficiency, as it significantly impacts lung function and quality of life. Liver disease also represents a significant concern, as it can lead to severe complications. The growing awareness and advancements in diagnostic methods are driving the demand for accurate and timely diagnosis.
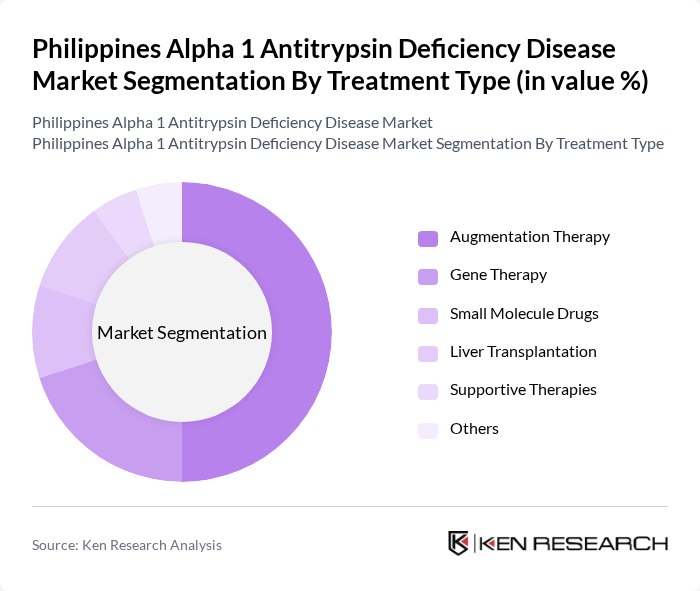
By Treatment Type:The treatment options for Alpha 1 Antitrypsin Deficiency include Augmentation Therapy, Gene Therapy, Small Molecule Drugs, Liver Transplantation, Supportive Therapies, and Others. Augmentation therapy is the most widely used treatment, as it directly addresses the deficiency of the protein in patients. Gene therapy is emerging as a promising option, while liver transplantation remains a critical intervention for patients with severe liver disease. The increasing availability of innovative therapies is enhancing treatment outcomes and patient quality of life.
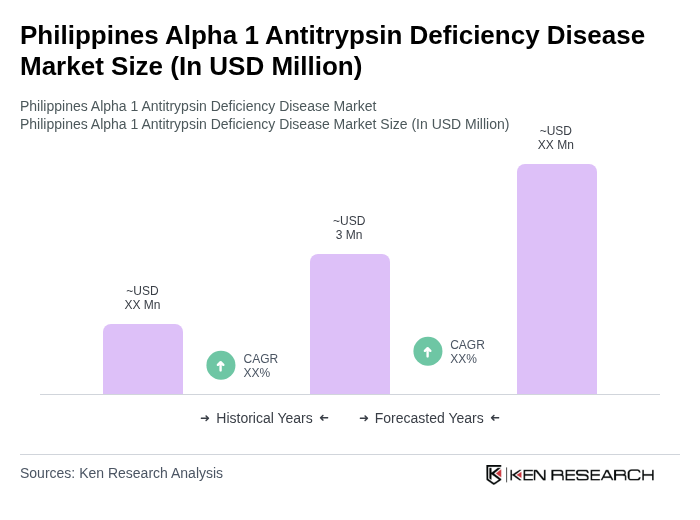
The Philippines Alpha 1 Antitrypsin Deficiency Disease Market is characterized by a dynamic mix of regional and international players. Leading participants such as Grifols, CSL Behring, Takeda Pharmaceutical Company, Shire (now part of Takeda), Vertex Pharmaceuticals, Alnylam Pharmaceuticals, Biogen, AbbVie, Regeneron Pharmaceuticals, Amgen, Roche, Sanofi, Novartis, Pfizer, Merck & Co. contribute to innovation, geographic expansion, and service delivery in this space.
The future of the Philippines Alpha 1 Antitrypsin Deficiency market appears promising, driven by ongoing advancements in healthcare infrastructure and increased government support. As awareness continues to grow, more patients are likely to seek diagnosis and treatment, leading to improved health outcomes. Additionally, the integration of telemedicine and digital health technologies is expected to enhance access to care, particularly in underserved areas, fostering a more equitable healthcare landscape for AATD patients across the nation.
| Segment | Sub-Segments |
|---|---|
| By Diagnosis | Chronic Obstructive Pulmonary Disease (COPD) Liver Disease Others |
| By Treatment Type | Augmentation Therapy Gene Therapy Small Molecule Drugs Liver Transplantation Supportive Therapies Others |
| By Drug Class | Protease Inhibitors Antioxidants Anti-inflammatory Agents Enzyme Replacement Therapy Others |
| By End-User | Hospitals Specialty Clinics Pharmacies Home Care Settings Others |
| By Distribution Channel | Hospital Pharmacies Retail Pharmacies Online Pharmacies Specialty Clinics Others |
| By Patient Demographics | Pediatric Patients Adult Patients Others |
| Scope Item/Segment | Sample Size | Target Respondent Profiles |
|---|---|---|
| Healthcare Providers Treating AATD | 100 | Pulmonologists, Hepatologists, General Practitioners |
| Patients Diagnosed with AATD | 80 | AATD Patients, Caregivers, Patient Advocates |
| Pharmaceutical Representatives | 60 | Sales Managers, Product Specialists in Rare Diseases |
| Health Insurance Providers | 50 | Policy Analysts, Claims Managers, Underwriters |
| Government Health Officials | 40 | Health Policy Makers, Program Directors, Epidemiologists |
Alpha 1 Antitrypsin Deficiency (AATD) is a genetic disorder that results in low levels of the protein alpha-1 antitrypsin, which protects the lungs and liver from damage. This deficiency can lead to serious health issues, including chronic obstructive pulmonary disease (COPD) and liver disease.