Region:Middle East
Author(s):Dev
Product Code:KRAA0641
Pages:92
Published On:January 2026
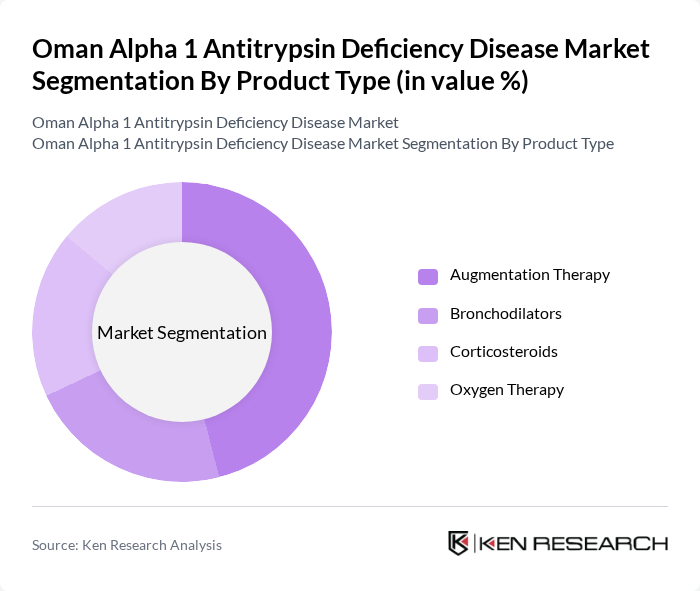
By Product Type:The product type segmentation includes Augmentation Therapy, Bronchodilators, Corticosteroids, and Oxygen Therapy. Among these, Augmentation Therapy is the leading sub-segment due to its effectiveness in managing the deficiency and improving lung function. The increasing adoption of this therapy, driven by patient preference for effective treatment options, has solidified its position in the market. Augmentation therapy accounts for approximately 46% of the market revenue share, establishing it as the dominant segment. The other therapies, while important, are often used as adjuncts to Augmentation Therapy, which remains the cornerstone of treatment.
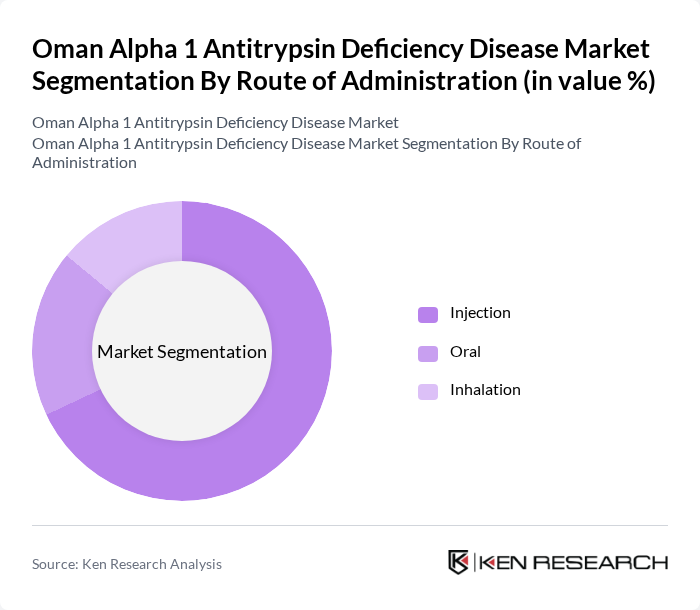
By Route of Administration:The route of administration includes Injection, Oral, and Inhalation. The Injection sub-segment dominates the market due to the direct delivery of medication into the bloodstream, ensuring rapid therapeutic effects. Patients and healthcare providers prefer this method for its efficacy, particularly in acute management scenarios. Augmentation therapy is primarily administered intravenously through infusion of purified alpha-1 antitrypsin protein to restore protective levels in the lungs. Oral and Inhalation routes are also utilized, but they are generally considered secondary options for ongoing management.
The Oman Alpha 1 Antitrypsin Deficiency Disease Market is characterized by a dynamic mix of regional and international players. Leading participants such as Grifols, CSL Behring, Takeda Pharmaceutical Company, AstraZeneca, Vertex Pharmaceuticals, Alnylam Pharmaceuticals, Shire (now part of Takeda), Biogen, Regeneron Pharmaceuticals, Amgen, Roche, Novartis, Sanofi, Merck & Co., and Pfizer contribute to innovation, geographic expansion, and service delivery in this space. Grifols leads the augmentation therapy segment with its PROLASTIN product, commanding the largest market share among therapeutic options.
The future of the AATD market in Oman appears promising, driven by ongoing advancements in personalized medicine and telehealth solutions. As healthcare infrastructure improves, the integration of digital health technologies will facilitate better patient management and follow-up care. Additionally, increased collaboration between pharmaceutical companies and research institutions is expected to foster innovation in treatment options, ultimately enhancing patient outcomes and accessibility to therapies for AATD in the region.
| Segment | Sub-Segments |
|---|---|
| By Product Type | Augmentation Therapy Bronchodilators Corticosteroids Oxygen Therapy |
| By Route of Administration | Injection Oral Inhalation |
| By End-User | Hospitals Specialty Clinics Pharmacies |
| By Distribution Channel | Hospital Pharmacies Retail Pharmacies Online Pharmacies |
| Scope Item/Segment | Sample Size | Target Respondent Profiles |
|---|---|---|
| Healthcare Providers | 100 | Doctors, Genetic Counselors, Pulmonologists |
| Patients with Alpha 1 Antitrypsin Deficiency | 40 | Diagnosed Patients, Caregivers |
| Pharmaceutical Representatives | 60 | Sales Managers, Product Specialists |
| Health Policy Makers | 50 | Government Officials, Health Administrators |
| Patient Advocacy Groups | 40 | Leaders, Community Organizers |
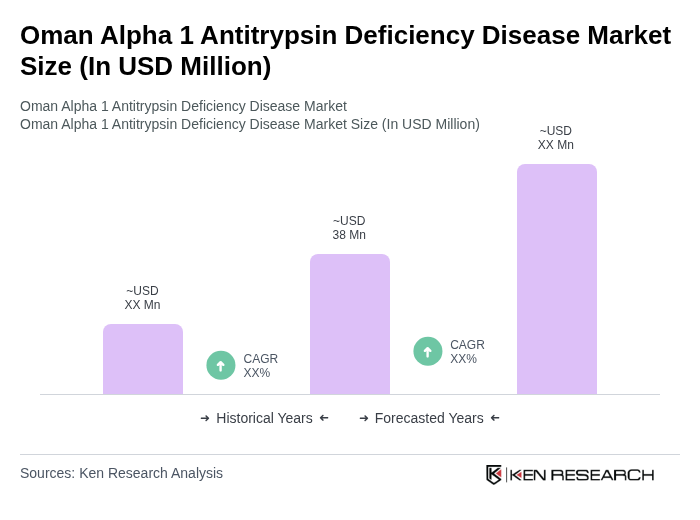
The Oman Alpha 1 Antitrypsin Deficiency Disease market is valued at approximately USD 38 million, driven by increased awareness, advancements in diagnostics, and the rising prevalence of related respiratory diseases.