Region:Asia
Author(s):Geetanshi
Product Code:KRAE1667
Pages:100
Published On:March 2026
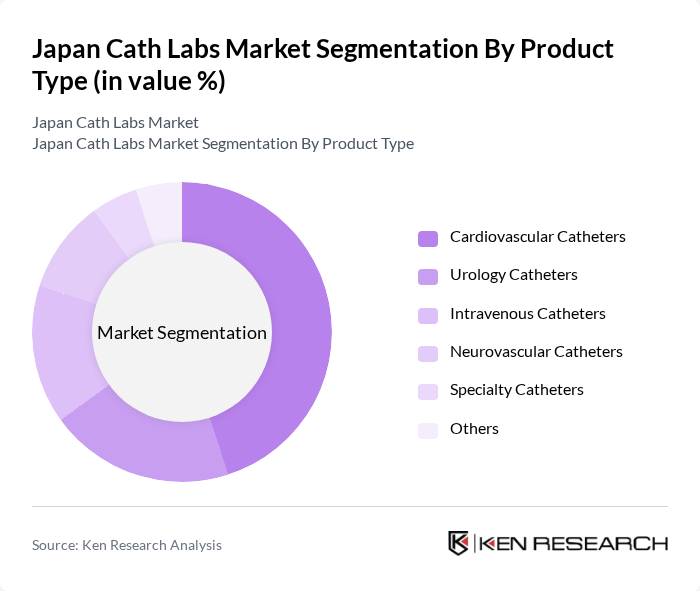
By Product Type:The product type segmentation includes various categories of catheters used in cath labs. The primary subsegments are cardiovascular catheters, urology catheters, intravenous catheters, neurovascular catheters, specialty catheters, and others. Among these, cardiovascular catheters dominate the market due to the high incidence of heart-related diseases and the growing preference for catheter-based interventions over traditional surgical methods.
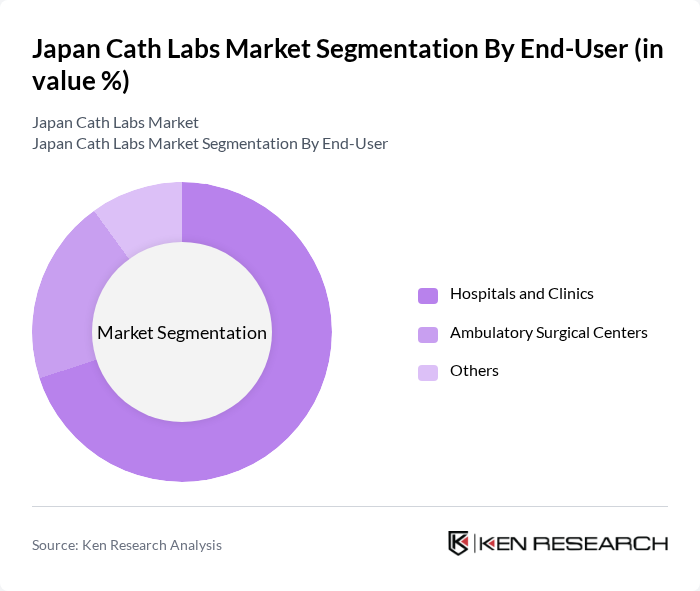
By End-User:The end-user segmentation encompasses hospitals and clinics, ambulatory surgical centers, electrophysiology labs, and other healthcare facilities. Hospitals and clinics are the leading end-users, primarily due to their extensive patient base and the need for advanced diagnostic and therapeutic procedures. The increasing number of outpatient procedures is also driving growth in ambulatory surgical centers and specialized electrophysiology facilities.
The Japan Cath Labs Market is characterized by a dynamic mix of regional and international players. Leading participants such as Siemens Healthineers, GE Healthcare, Philips Healthcare, Canon Medical Systems, Hitachi Medical Corporation, Toshiba Medical Systems, Boston Scientific, Medtronic, Abbott Laboratories, Terumo Corporation, Johnson & Johnson, B. Braun Melsungen AG, Cook Medical, Edwards Lifesciences, and Abbott (St. Jude Medical) contribute to innovation, geographic expansion, and service delivery in this space.
The future of the Japan cath labs market appears promising, driven by technological advancements and an increasing focus on patient-centered care. As healthcare providers adopt minimally invasive techniques and integrate artificial intelligence into diagnostics, the efficiency and effectiveness of cath lab procedures are expected to improve. Additionally, the ongoing expansion of telemedicine services will facilitate remote monitoring and consultations, enhancing patient access to care and potentially increasing the volume of procedures performed in cath labs.
| Segment | Sub-Segments |
|---|---|
| By Product Type | Cardiovascular Catheters Urology Catheters Intravenous Catheters Neurovascular Catheters Specialty Catheters Others |
| By End-User | Hospitals and Clinics Ambulatory Surgical Centers Others |
| By Procedure Type | Diagnostic Procedures Interventional Procedures Electrophysiology Procedures Others |
| By Imaging Technology | Angiography Systems Intravascular Ultrasound (IVUS) Optical Coherence Tomography (OCT) Others |
| By Ownership | Hospital-owned Cath Labs Standalone Cath Labs Others |
| By Region | Kanto Region Kansai/Kinki Region Central/Chubu Region Kyushu-Okinawa Region Tohoku Region Others |
| Scope Item/Segment | Sample Size | Target Respondent Profiles |
|---|---|---|
| Cardiologist Insights | 100 | Interventional Cardiologists, Electrophysiologists |
| Cath Lab Equipment Manufacturers | 75 | Product Managers, Sales Directors |
| Healthcare Administrators | 60 | Hospital Administrators, Financial Officers |
| Regulatory Bodies | 50 | Health Policy Analysts, Compliance Officers |
| Patient Advocacy Groups | 40 | Patient Representatives, Health Educators |
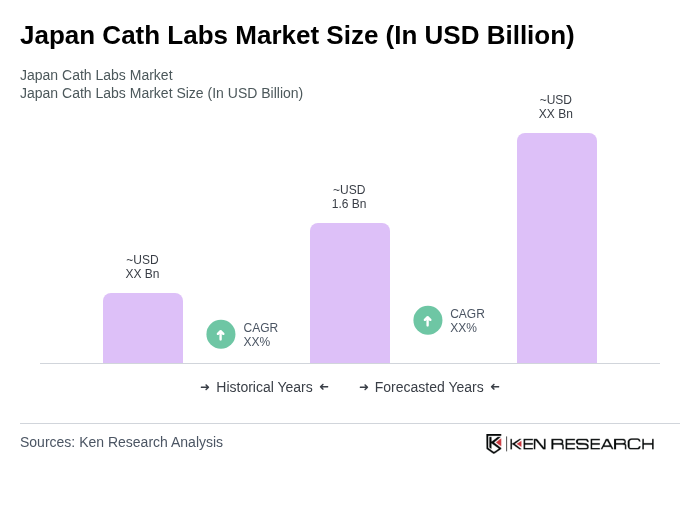
The Japan Cath Labs Market is valued at approximately USD 1.6 billion, reflecting a significant growth driven by the increasing prevalence of cardiovascular diseases and advancements in catheter technology.