About the Report
Base Year 2024Japan Drug Discovery Development Laboratory Services Market Overview
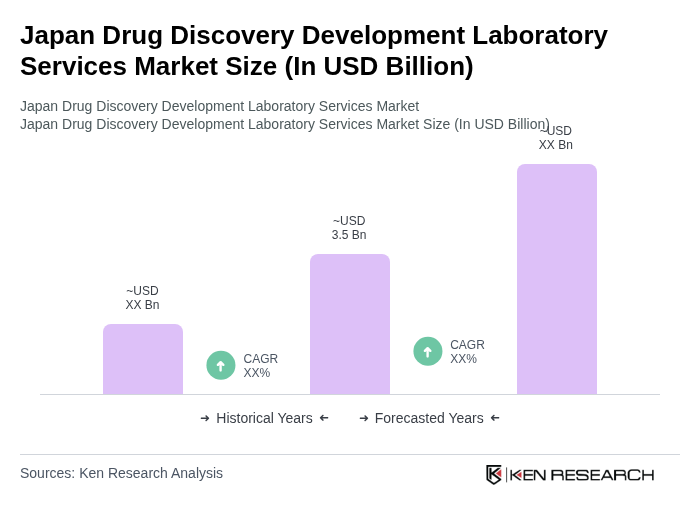
- The Japan Drug Discovery Development Laboratory Services Market is valued at USD 3.5 billion, based on a five-year historical analysis. This growth is primarily driven by the increasing demand for innovative drug development solutions, advancements in biotechnology, and a robust pharmaceutical sector that emphasizes research and development. The market is also supported by the rising prevalence of chronic diseases, necessitating the need for effective therapeutic solutions.
- Key players in this market are concentrated in major cities such as Tokyo, Osaka, and Yokohama. These cities dominate due to their advanced infrastructure, access to skilled labor, and proximity to leading pharmaceutical and biotechnology companies. The presence of numerous research institutions and universities further enhances the innovation ecosystem, making these regions pivotal for drug discovery and development activities.
- In 2023, the Japanese government implemented the "Pharmaceutical and Medical Device Act," which aims to streamline the approval process for new drugs and medical devices. This regulation encourages faster market entry for innovative therapies while ensuring safety and efficacy, thereby fostering a more competitive environment for drug discovery and development laboratory services.
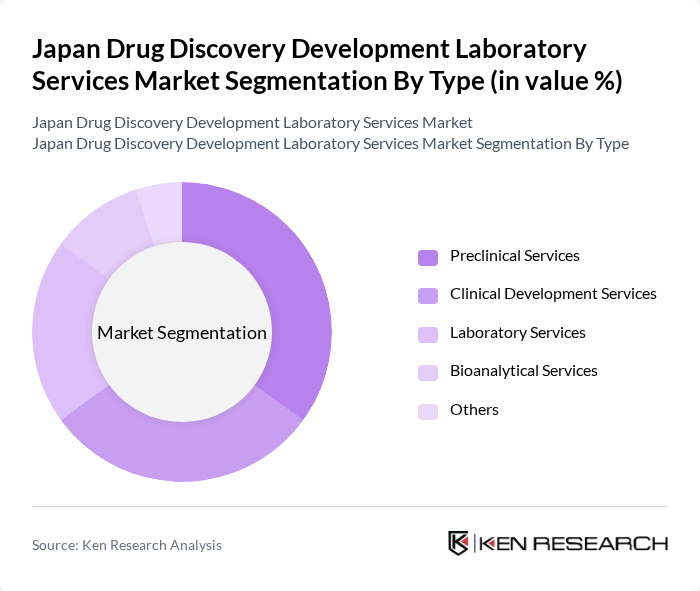
Japan Drug Discovery Development Laboratory Services Market Segmentation
By Type:The market is segmented into various types of services that cater to different stages of drug development. Among these, preclinical services and clinical development services are particularly significant, as they encompass essential processes in drug testing and validation. The laboratory services segment also plays a crucial role, providing necessary analytical support throughout the drug development lifecycle.
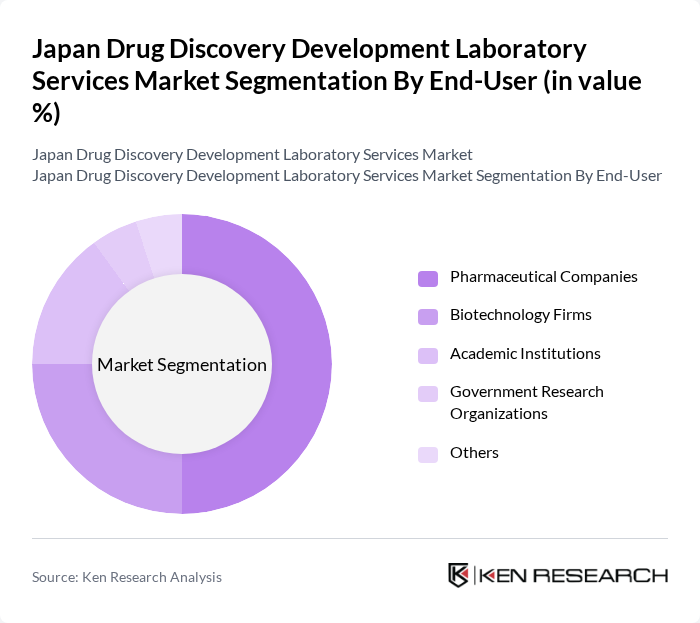
By End-User:The end-user segmentation includes various stakeholders involved in drug discovery and development. Pharmaceutical companies are the largest segment, driven by their need for comprehensive laboratory services to support extensive R&D activities. Biotechnology firms also represent a significant portion of the market, focusing on innovative therapies and personalized medicine, while academic institutions and government research organizations contribute to the overall landscape through research initiatives.
Japan Drug Discovery Development Laboratory Services Market Competitive Landscape
The Japan Drug Discovery Development Laboratory Services Market is characterized by a dynamic mix of regional and international players. Leading participants such as Takeda Pharmaceutical Company Limited, Astellas Pharma Inc., Daiichi Sankyo Company, Limited, Chugai Pharmaceutical Co., Ltd., Sumitomo Dainippon Pharma Co., Ltd., Mitsubishi Tanabe Pharma Corporation, Eisai Co., Ltd., Kyowa Kirin Co., Ltd., Otsuka Pharmaceutical Co., Ltd., Santen Pharmaceutical Co., Ltd., Hoya Corporation, Sysmex Corporation, Fujifilm Holdings Corporation, RIKEN Genesis Co., Ltd., Medipal Holdings Corporation contribute to innovation, geographic expansion, and service delivery in this space.
Japan Drug Discovery Development Laboratory Services Market Industry Analysis
Growth Drivers
- Increasing R&D Investments:Japan's pharmaceutical R&D expenditure reached approximately ¥1.5 trillion (around $13.5 billion) in future, reflecting a robust commitment to innovation. The government has been actively promoting R&D through initiatives like the "Innovative Drug Discovery and Development Program," which allocated ¥100 billion ($900 million) in future. This financial backing is crucial for enhancing drug discovery capabilities, fostering a conducive environment for laboratory services, and ultimately driving market growth in the drug discovery sector.
- Rising Demand for Personalized Medicine:The personalized medicine market in Japan is projected to grow significantly, with an estimated value of ¥1 trillion ($9 billion) by future. This surge is driven by advancements in genomics and biotechnology, leading to tailored therapies that cater to individual patient needs. As healthcare providers increasingly adopt personalized approaches, the demand for specialized laboratory services will rise, creating opportunities for growth in drug discovery development services across the country.
- Advancements in Biotechnology:Japan's biotechnology sector is experiencing rapid growth, with investments reaching ¥600 billion ($5.4 billion) in future. Innovations in biopharmaceuticals, gene therapy, and regenerative medicine are reshaping drug discovery processes. The integration of cutting-edge technologies, such as CRISPR and monoclonal antibodies, is enhancing the efficiency and effectiveness of laboratory services. This trend is expected to drive demand for specialized drug discovery services, positioning Japan as a leader in the global biotechnology landscape.
Market Challenges
- High Operational Costs:The operational costs for drug discovery laboratories in Japan are notably high, averaging around ¥2 billion ($18 million) annually. This includes expenses related to advanced equipment, skilled personnel, and compliance with stringent regulations. Such financial burdens can deter smaller firms from entering the market, limiting competition and innovation. Consequently, high operational costs pose a significant challenge to the growth and sustainability of drug discovery development services in Japan.
- Stringent Regulatory Requirements:Japan's regulatory landscape for drug development is complex, with multiple layers of oversight from agencies like the Pharmaceuticals and Medical Devices Agency (PMDA). Compliance with Good Laboratory Practice (GLP) and other stringent guidelines can extend timelines and increase costs, with an average approval process taking over 10 years. These regulatory hurdles can impede the agility of laboratory services, making it challenging for companies to adapt to market demands swiftly.
Japan Drug Discovery Development Laboratory Services Market Future Outlook
The future of Japan's drug discovery development laboratory services market appears promising, driven by technological advancements and increasing collaboration between academia and industry. As the demand for personalized medicine continues to rise, laboratories will need to adapt their services to meet evolving healthcare needs. Furthermore, the integration of artificial intelligence and machine learning in drug discovery processes is expected to enhance efficiency and reduce time-to-market for new therapies, positioning Japan as a key player in the global pharmaceutical landscape.
Market Opportunities
- Expansion of CRO Services:The contract research organization (CRO) market in Japan is projected to grow to ¥500 billion ($4.5 billion) by future. This expansion presents significant opportunities for laboratory services to partner with CROs, enhancing their capabilities and reach. By leveraging CRO networks, laboratories can access a broader client base and streamline their service offerings, ultimately driving growth in the drug discovery sector.
- Integration of AI in Drug Discovery:The application of artificial intelligence in drug discovery is expected to revolutionize the industry, with investments in AI technologies projected to exceed ¥300 billion ($2.7 billion) by future. This integration will enable laboratories to analyze vast datasets more efficiently, identify potential drug candidates faster, and reduce costs. As AI becomes more prevalent, laboratories that adopt these technologies will gain a competitive edge in the market.
Scope of the Report
| Segment | Sub-Segments |
|---|---|
| By Type | Preclinical Services Clinical Development Services Laboratory Services Bioanalytical Services Others |
| By End-User | Pharmaceutical Companies Biotechnology Firms Academic Institutions Government Research Organizations Others |
| By Therapeutic Area | Oncology Cardiovascular Neurology Infectious Diseases Others |
| By Service Model | Full-Service CROs Niche Service Providers In-House Services Others |
| By Region | Kanto Kansai Chubu Others |
| By Technology | High-Throughput Screening Genomics Proteomics Others |
| By Funding Source | Public Funding Private Investment Venture Capital Others |
Key Target Audience
Investors and Venture Capitalist Firms
Government and Regulatory Bodies (e.g., Pharmaceuticals and Medical Devices Agency, Ministry of Health, Labour and Welfare)
Biotechnology Companies
Pharmaceutical Manufacturers
Contract Research Organizations (CROs)
Clinical Research Institutions
Healthcare Technology Companies
Biopharmaceutical Associations
Players Mentioned in the Report:
Takeda Pharmaceutical Company Limited
Astellas Pharma Inc.
Daiichi Sankyo Company, Limited
Chugai Pharmaceutical Co., Ltd.
Sumitomo Dainippon Pharma Co., Ltd.
Mitsubishi Tanabe Pharma Corporation
Eisai Co., Ltd.
Kyowa Kirin Co., Ltd.
Otsuka Pharmaceutical Co., Ltd.
Santen Pharmaceutical Co., Ltd.
Hoya Corporation
Sysmex Corporation
Fujifilm Holdings Corporation
RIKEN Genesis Co., Ltd.
Medipal Holdings Corporation
Table of Contents
Market Assessment Phase
1. Executive Summary and Approach
2. Japan Drug Discovery Development Laboratory Services Market Overview
2.1 Key Insights and Strategic Recommendations
2.2 Japan Drug Discovery Development Laboratory Services Market Overview
2.3 Definition and Scope
2.4 Evolution of Market Ecosystem
2.5 Timeline of Key Regulatory Milestones
2.6 Value Chain & Stakeholder Mapping
2.7 Business Cycle Analysis
2.8 Policy & Incentive Landscape
3. Japan Drug Discovery Development Laboratory Services Market Analysis
3.1 Growth Drivers
3.1.1 Increasing R&D Investments
3.1.2 Rising Demand for Personalized Medicine
3.1.3 Advancements in Biotechnology
3.1.4 Growing Collaborations between Academia and Industry
3.2 Market Challenges
3.2.1 High Operational Costs
3.2.2 Stringent Regulatory Requirements
3.2.3 Limited Access to Advanced Technologies
3.2.4 Shortage of Skilled Workforce
3.3 Market Opportunities
3.3.1 Expansion of CRO Services
3.3.2 Integration of AI in Drug Discovery
3.3.3 Increasing Focus on Rare Diseases
3.3.4 Growth in Biopharmaceuticals
3.4 Market Trends
3.4.1 Shift Towards Outsourcing
3.4.2 Emphasis on Data-Driven Decision Making
3.4.3 Rise of Virtual Trials
3.4.4 Adoption of Cloud-Based Solutions
3.5 Government Regulation
3.5.1 Pharmaceutical Affairs Law
3.5.2 Good Laboratory Practice (GLP) Guidelines
3.5.3 Clinical Trials Act
3.5.4 Health Insurance System Regulations
4. SWOT Analysis
5. Stakeholder Analysis
6. Porter's Five Forces Analysis
7. Japan Drug Discovery Development Laboratory Services Market Size, 2019-2024
7.1 By Value
7.2 By Volume
7.3 By Average Selling Price
8. Japan Drug Discovery Development Laboratory Services Market Segmentation
8.1 By Type
8.1.1 Preclinical Services
8.1.2 Clinical Development Services
8.1.3 Laboratory Services
8.1.4 Bioanalytical Services
8.1.5 Others
8.2 By End-User
8.2.1 Pharmaceutical Companies
8.2.2 Biotechnology Firms
8.2.3 Academic Institutions
8.2.4 Government Research Organizations
8.2.5 Others
8.3 By Therapeutic Area
8.3.1 Oncology
8.3.2 Cardiovascular
8.3.3 Neurology
8.3.4 Infectious Diseases
8.3.5 Others
8.4 By Service Model
8.4.1 Full-Service CROs
8.4.2 Niche Service Providers
8.4.3 In-House Services
8.4.4 Others
8.5 By Region
8.5.1 Kanto
8.5.2 Kansai
8.5.3 Chubu
8.5.4 Others
8.6 By Technology
8.6.1 High-Throughput Screening
8.6.2 Genomics
8.6.3 Proteomics
8.6.4 Others
8.7 By Funding Source
8.7.1 Public Funding
8.7.2 Private Investment
8.7.3 Venture Capital
8.7.4 Others
9. Japan Drug Discovery Development Laboratory Services Market Competitive Analysis
9.1 Market Share of Key Players
9.2 Cross Comparison of Key Players
9.2.1 Company Name
9.2.2 Group Size (Large, Medium, or Small as per industry convention)
9.2.3 Revenue Growth Rate
9.2.4 Client Retention Rate
9.2.5 Market Penetration Rate
9.2.6 Pricing Strategy
9.2.7 Service Diversification Index
9.2.8 Customer Satisfaction Score
9.2.9 Innovation Rate
9.2.10 Operational Efficiency Ratio
9.3 SWOT Analysis of Top Players
9.4 Pricing Analysis
9.5 Detailed Profile of Major Companies
9.5.1 Takeda Pharmaceutical Company Limited
9.5.2 Astellas Pharma Inc.
9.5.3 Daiichi Sankyo Company, Limited
9.5.4 Chugai Pharmaceutical Co., Ltd.
9.5.5 Sumitomo Dainippon Pharma Co., Ltd.
9.5.6 Mitsubishi Tanabe Pharma Corporation
9.5.7 Eisai Co., Ltd.
9.5.8 Kyowa Kirin Co., Ltd.
9.5.9 Otsuka Pharmaceutical Co., Ltd.
9.5.10 Santen Pharmaceutical Co., Ltd.
9.5.11 Hoya Corporation
9.5.12 Sysmex Corporation
9.5.13 Fujifilm Holdings Corporation
9.5.14 RIKEN Genesis Co., Ltd.
9.5.15 Medipal Holdings Corporation
10. Japan Drug Discovery Development Laboratory Services Market End-User Analysis
10.1 Procurement Behavior of Key Ministries
10.1.1 Ministry of Health, Labour and Welfare
10.1.2 Ministry of Education, Culture, Sports, Science and Technology
10.1.3 Ministry of Economy, Trade and Industry
10.1.4 Others
10.2 Corporate Spend on Infrastructure & Energy
10.2.1 Pharmaceutical Sector Investments
10.2.2 Biotechnology Sector Investments
10.2.3 Academic Research Funding
10.2.4 Others
10.3 Pain Point Analysis by End-User Category
10.3.1 Pharmaceutical Companies
10.3.2 Biotechnology Firms
10.3.3 Academic Institutions
10.3.4 Government Research Organizations
10.4 User Readiness for Adoption
10.4.1 Awareness of New Technologies
10.4.2 Training and Support Needs
10.4.3 Budget Constraints
10.4.4 Others
10.5 Post-Deployment ROI and Use Case Expansion
10.5.1 Measurement of Success Metrics
10.5.2 Scalability of Solutions
10.5.3 Long-term Partnerships
10.5.4 Others
11. Japan Drug Discovery Development Laboratory Services Market Future Size, 2025-2030
11.1 By Value
11.2 By Volume
11.3 By Average Selling Price
Go-To-Market Strategy Phase
1. Whitespace Analysis + Business Model Canvas
1.1 Market Gaps Identification
1.2 Business Model Development
2. Marketing and Positioning Recommendations
2.1 Branding Strategies
2.2 Product USPs
3. Distribution Plan
3.1 Urban Retail Strategies
3.2 Rural NGO Tie-ups
4. Channel & Pricing Gaps
4.1 Underserved Routes
4.2 Pricing Bands
5. Unmet Demand & Latent Needs
5.1 Category Gaps
5.2 Consumer Segments
6. Customer Relationship
6.1 Loyalty Programs
6.2 After-sales Service
7. Value Proposition
7.1 Sustainability
7.2 Integrated Supply Chains
8. Key Activities
8.1 Regulatory Compliance
8.2 Branding
8.3 Distribution Setup
9. Entry Strategy Evaluation
9.1 Domestic Market Entry Strategy
9.1.1 Product Mix
9.1.2 Pricing Band
9.1.3 Packaging
9.2 Export Entry Strategy
9.2.1 Target Countries
9.2.2 Compliance Roadmap
10. Entry Mode Assessment
10.1 Joint Ventures
10.2 Greenfield Investments
10.3 Mergers & Acquisitions
10.4 Distributor Model
11. Capital and Timeline Estimation
11.1 Capital Requirements
11.2 Timelines
12. Control vs Risk Trade-Off
12.1 Ownership vs Partnerships
13. Profitability Outlook
13.1 Breakeven Analysis
13.2 Long-term Sustainability
14. Potential Partner List
14.1 Distributors
14.2 Joint Ventures
14.3 Acquisition Targets
15. Execution Roadmap
15.1 Phased Plan for Market Entry
15.1.1 Market Setup
15.1.2 Market Entry
15.1.3 Growth Acceleration
15.1.4 Scale & Stabilize
15.2 Key Activities and Milestones
15.2.1 Milestone Planning
15.2.2 Activity Tracking
Research Methodology
Phase 1: Approach1
Desk Research
- Analysis of industry reports from Japanese pharmaceutical associations and government health agencies
- Review of published scientific literature on drug discovery and development trends in Japan
- Examination of market data from trade publications and market research firms focusing on laboratory services
Primary Research
- Interviews with R&D leaders at major pharmaceutical companies operating in Japan
- Surveys with laboratory service providers to understand service offerings and pricing structures
- Field interviews with regulatory experts to gather insights on compliance and market entry barriers
Validation & Triangulation
- Cross-validation of findings through multiple data sources, including industry reports and expert opinions
- Triangulation of quantitative data with qualitative insights from interviews
- Sanity checks through expert panel reviews to ensure data accuracy and relevance
Phase 2: Market Size Estimation1
Top-down Assessment
- Estimation of the overall pharmaceutical market size in Japan as a basis for laboratory services demand
- Segmentation of the market by therapeutic areas and types of laboratory services offered
- Incorporation of government funding and initiatives supporting drug discovery and development
Bottom-up Modeling
- Collection of data on service volumes from leading laboratory service providers in Japan
- Operational cost analysis based on service pricing and service delivery models
- Volume x cost calculations to derive revenue estimates for each service segment
Forecasting & Scenario Analysis
- Multi-factor regression analysis incorporating factors such as R&D spending and technological advancements
- Scenario modeling based on potential changes in regulatory frameworks and market dynamics
- Development of baseline, optimistic, and pessimistic forecasts through 2030
Phase 3: CATI Sample Composition1
| Scope Item/Segment | Sample Size | Target Respondent Profiles |
|---|---|---|
| Pharmaceutical R&D Laboratories | 100 | R&D Directors, Lab Managers |
| Biotechnology Firms | 80 | Chief Scientific Officers, Project Managers |
| Contract Research Organizations (CROs) | 70 | Business Development Managers, Operations Heads |
| Regulatory Affairs Consultancies | 60 | Regulatory Affairs Specialists, Compliance Officers |
| Academic Research Institutions | 90 | Principal Investigators, Research Coordinators |
Frequently Asked Questions
What is the current value of the Japan Drug Discovery Development Laboratory Services Market?
The Japan Drug Discovery Development Laboratory Services Market is valued at approximately USD 3.5 billion, reflecting significant growth driven by increasing demand for innovative drug development solutions and advancements in biotechnology.