About the Report
Base Year 2024Kuwait Arrhythmia Monitoring Devices Market Overview
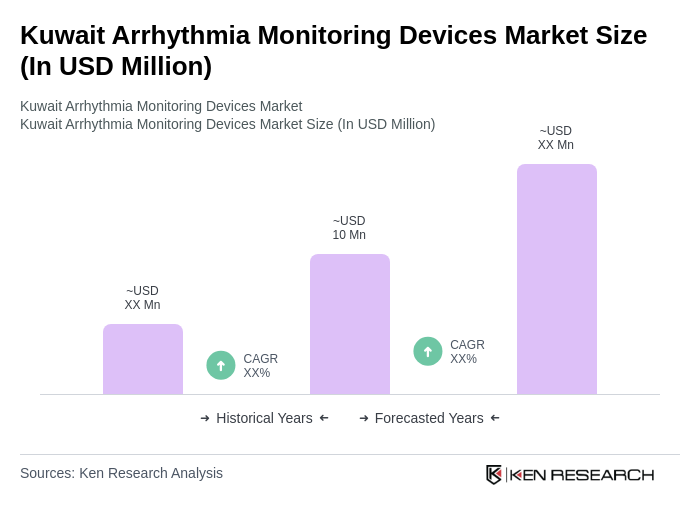
- The Kuwait Arrhythmia Monitoring Devices Market is valued at USD 10 million, based on a five-year historical analysis. This growth is primarily driven by the increasing prevalence of cardiovascular diseases, rising geriatric population, and advancements in technology that enhance monitoring capabilities. The demand for arrhythmia monitoring devices has surged as healthcare providers focus on early detection and management of heart conditions, leading to improved patient outcomes. The increasing adoption of Holter ECG devices, driven by rising cardiovascular disease prevalence including 4,500 heart attack cases reported nationwide in May 2023, has been instrumental in accelerating market growth. Additionally, the integration of wearable technology advancements, artificial intelligence and machine learning capabilities, and remote patient monitoring systems are transforming cardiac care delivery and expanding market opportunities.
- Kuwait City is the dominant region in the market due to its advanced healthcare infrastructure and concentration of specialized medical facilities. The city benefits from a high standard of living and increased healthcare spending, which supports the adoption of innovative medical technologies. Additionally, the presence of key healthcare institutions and a growing awareness of heart health among the population further contribute to its market leadership.
- Kuwait's healthcare sector operates under the oversight of the Ministry of Health (MOH), which enforces the Medical Devices Regulation Law No. 25 of 2016. This legislation establishes comprehensive requirements for the registration, import, distribution, and use of medical devices, including arrhythmia monitoring equipment. The law mandates that all medical devices must obtain approval from the MOH before being marketed or used in healthcare facilities, with specific standards for safety, efficacy, and quality control. Devices are classified based on risk levels, with higher-risk cardiac monitoring equipment subject to rigorous premarket evaluation, clinical data submission, and ongoing post-market surveillance. Healthcare facilities must ensure compliance with installation, maintenance, and operational protocols, while distributors are required to maintain proper documentation and traceability throughout the supply chain.
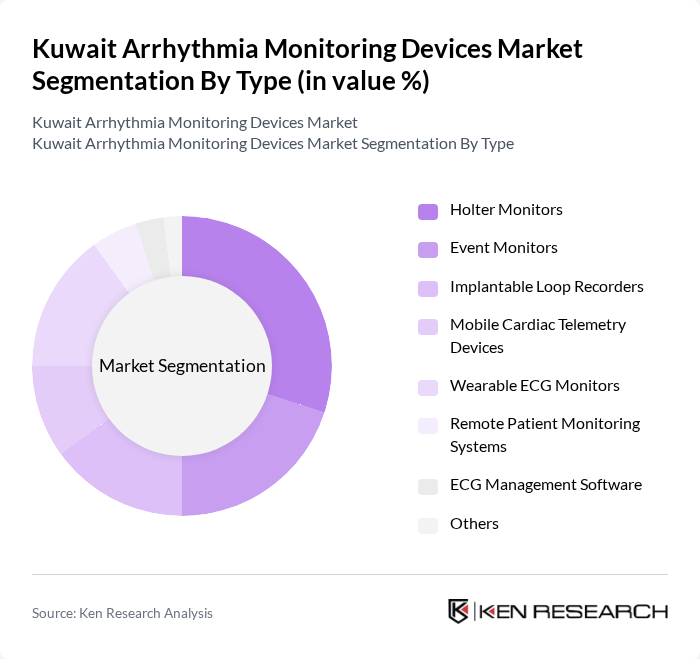
Kuwait Arrhythmia Monitoring Devices Market Segmentation
By Type:The market is segmented into various types of arrhythmia monitoring devices, including Holter Monitors, Event Monitors, Implantable Loop Recorders, Mobile Cardiac Telemetry Devices, Wearable ECG Monitors, Remote Patient Monitoring Systems, ECG Management Software, and Others. Among these, Holter Monitors and Wearable ECG Monitors are gaining significant traction due to their convenience and ability to provide continuous monitoring. The increasing consumer preference for non-invasive and user-friendly devices is driving the growth of these segments.
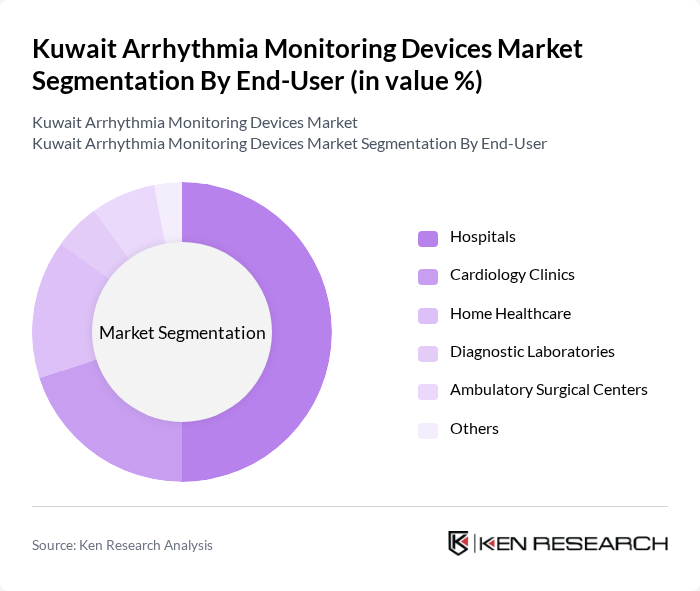
By End-User:The end-user segmentation includes Hospitals, Cardiology Clinics, Home Healthcare, Diagnostic Laboratories, Ambulatory Surgical Centers, and Others. Hospitals are the leading end-users due to their comprehensive cardiac care services and the need for advanced monitoring solutions. The increasing number of cardiac procedures and the emphasis on patient safety and monitoring in hospital settings are driving the demand for arrhythmia monitoring devices.
Kuwait Arrhythmia Monitoring Devices Market Competitive Landscape
The Kuwait Arrhythmia Monitoring Devices Market is characterized by a dynamic mix of regional and international players. Leading participants such as Medtronic plc, Abbott Laboratories, Boston Scientific Corporation, Biotronik SE & Co. KG, Koninklijke Philips N.V. (Philips Healthcare), GE Healthcare, Siemens Healthineers AG, Nihon Kohden Corporation, Zoll Medical Corporation, Hillrom Services, Inc. (Welch Allyn), iRhythm Technologies, Inc., Cardiac Science Corporation, LivaNova PLC, AliveCor, Inc., Spacelabs Healthcare (OSI Systems, Inc.), Schiller AG, Bionet Co., Ltd., Biotricity Inc., Applied Cardiac Systems, Inc. contribute to innovation, geographic expansion, and service delivery in this space.
Kuwait Arrhythmia Monitoring Devices Market Industry Analysis
Growth Drivers
- Increasing Prevalence of Arrhythmias:The prevalence of arrhythmias in Kuwait is rising, with approximately 1.5 million individuals affected by various heart rhythm disorders in future. This increase is attributed to lifestyle changes, including high rates of obesity and diabetes, which affect 35% and 25% of the adult population, respectively. The growing patient base drives demand for arrhythmia monitoring devices, as healthcare providers seek effective solutions to manage these conditions and improve patient outcomes.
- Advancements in Monitoring Technology:Technological innovations in arrhythmia monitoring devices are transforming patient care. In future, the introduction of devices featuring real-time data transmission and AI-driven analytics is expected to enhance diagnostic accuracy. The global market for wearable health technology is projected to reach $60 billion, with a significant portion attributed to cardiac monitoring devices. These advancements facilitate timely interventions, thereby increasing the adoption of such devices in Kuwait's healthcare system.
- Rising Healthcare Expenditure:Kuwait's healthcare expenditure is projected to reach $8.5 billion in future, reflecting a commitment to improving health services. This increase is driven by government initiatives aimed at enhancing healthcare infrastructure and access to advanced medical technologies. As healthcare spending rises, hospitals and clinics are more likely to invest in arrhythmia monitoring devices, thereby expanding the market and improving patient care capabilities across the nation.
Market Challenges
- High Cost of Advanced Devices:The high cost of advanced arrhythmia monitoring devices poses a significant barrier to market growth in Kuwait. Many state-of-the-art devices range from $1,500 to $5,000, making them unaffordable for a substantial portion of the population. This financial constraint limits access to necessary monitoring solutions, particularly in public healthcare settings, where budget allocations are often insufficient to cover such expenses.
- Limited Reimbursement Policies:The lack of comprehensive reimbursement policies for arrhythmia monitoring devices in Kuwait presents a challenge for market expansion. Currently, only 40% of healthcare providers offer reimbursement for these devices, which discourages patients from seeking necessary monitoring. This limitation affects the overall adoption rate of advanced technologies, as many patients are unable to afford out-of-pocket expenses for essential health monitoring solutions.
Kuwait Arrhythmia Monitoring Devices Market Future Outlook
The future of the Kuwait arrhythmia monitoring devices market appears promising, driven by technological advancements and increasing healthcare investments. As the healthcare sector continues to evolve, the integration of telemedicine and remote monitoring solutions is expected to gain traction in future. Additionally, the focus on preventive healthcare will likely lead to greater demand for innovative monitoring devices, enabling early detection and management of arrhythmias, ultimately improving patient outcomes and reducing healthcare costs.
Market Opportunities
- Expansion of Telemedicine Services:The growth of telemedicine services in Kuwait presents a significant opportunity for arrhythmia monitoring devices. With an estimated 60% of healthcare consultations expected to be conducted remotely in future, the demand for devices that facilitate remote monitoring will increase. This trend allows for continuous patient engagement and timely interventions, enhancing overall healthcare delivery.
- Development of Portable Monitoring Devices:The rising demand for portable monitoring devices is another key opportunity in the market. As patients seek more convenient and user-friendly solutions, manufacturers are focusing on developing compact, wearable devices. This trend aligns with the global shift towards personalized healthcare, enabling patients to monitor their heart health effectively and share data with healthcare providers seamlessly.
Scope of the Report
| Segment | Sub-Segments |
|---|---|
| By Type | Holter Monitors Event Monitors Implantable Loop Recorders Mobile Cardiac Telemetry Devices Wearable ECG Monitors Remote Patient Monitoring Systems ECG Management Software Others |
| By End-User | Hospitals Cardiology Clinics Home Healthcare Diagnostic Laboratories Ambulatory Surgical Centers Others |
| By Distribution Channel | Direct Sales Online Retail Medical Device Distributors Others |
| By Application | Atrial Fibrillation Monitoring Bradycardia Monitoring Tachycardia Monitoring Post-Operative Monitoring Premature Contraction Detection Others |
| By Technology | Wireless Monitoring Wired Monitoring Cloud-Based Solutions AI-Enabled Monitoring Others |
| By Price Range | Low-End Devices Mid-Range Devices High-End Devices |
| By Region | Central Kuwait Southern Kuwait Northern Kuwait Eastern Kuwait Western Kuwait Others |
Key Target Audience
Investors and Venture Capitalist Firms
Government and Regulatory Bodies (e.g., Ministry of Health, Kuwait Food and Drug Administration)
Manufacturers and Producers
Distributors and Retailers
Healthcare Providers and Hospitals
Medical Device Importers and Exporters
Industry Associations and Societies
Health Insurance Companies
Players Mentioned in the Report:
Medtronic plc
Abbott Laboratories
Boston Scientific Corporation
Biotronik SE & Co. KG
Koninklijke Philips N.V. (Philips Healthcare)
GE Healthcare
Siemens Healthineers AG
Nihon Kohden Corporation
Zoll Medical Corporation
Hillrom Services, Inc. (Welch Allyn)
iRhythm Technologies, Inc.
Cardiac Science Corporation
LivaNova PLC
AliveCor, Inc.
Spacelabs Healthcare (OSI Systems, Inc.)
Schiller AG
Bionet Co., Ltd.
Biotricity Inc.
Applied Cardiac Systems, Inc.
Table of Contents
Market Assessment Phase
1. Executive Summary and Approach
2. Kuwait Arrhythmia Monitoring Devices Market Overview
2.1 Key Insights and Strategic Recommendations
2.2 Kuwait Arrhythmia Monitoring Devices Market Overview
2.3 Definition and Scope
2.4 Evolution of Market Ecosystem
2.5 Timeline of Key Regulatory Milestones
2.6 Value Chain & Stakeholder Mapping
2.7 Business Cycle Analysis
2.8 Policy & Incentive Landscape
3. Kuwait Arrhythmia Monitoring Devices Market Analysis
3.1 Growth Drivers
3.1.1 Increasing prevalence of arrhythmias
3.1.2 Advancements in monitoring technology
3.1.3 Rising healthcare expenditure
3.1.4 Growing awareness of heart health
3.2 Market Challenges
3.2.1 High cost of advanced devices
3.2.2 Limited reimbursement policies
3.2.3 Shortage of skilled healthcare professionals
3.2.4 Regulatory hurdles in device approval
3.3 Market Opportunities
3.3.1 Expansion of telemedicine services
3.3.2 Development of portable monitoring devices
3.3.3 Increasing partnerships with tech companies
3.3.4 Growing demand for preventive healthcare solutions
3.4 Market Trends
3.4.1 Integration of AI in monitoring devices
3.4.2 Shift towards home-based monitoring
3.4.3 Rise in wearable health technology
3.4.4 Focus on patient-centric care models
3.5 Government Regulation
3.5.1 Stricter quality control measures
3.5.2 Enhanced data privacy regulations
3.5.3 Incentives for local manufacturing
3.5.4 Guidelines for telehealth services
4. SWOT Analysis
5. Stakeholder Analysis
6. Porter's Five Forces Analysis
7. Kuwait Arrhythmia Monitoring Devices Market Market Size, 2019-2024
7.1 By Value
7.2 By Volume
7.3 By Average Selling Price
8. Kuwait Arrhythmia Monitoring Devices Market Segmentation
8.1 By Type
8.1.1 Holter Monitors
8.1.2 Event Monitors
8.1.3 Implantable Loop Recorders
8.1.4 Mobile Cardiac Telemetry Devices
8.1.5 Wearable ECG Monitors
8.1.6 Remote Patient Monitoring Systems
8.1.7 ECG Management Software
8.1.8 Others
8.2 By End-User
8.2.1 Hospitals
8.2.2 Cardiology Clinics
8.2.3 Home Healthcare
8.2.4 Diagnostic Laboratories
8.2.5 Ambulatory Surgical Centers
8.2.6 Others
8.3 By Distribution Channel
8.3.1 Direct Sales
8.3.2 Online Retail
8.3.3 Medical Device Distributors
8.3.4 Others
8.4 By Application
8.4.1 Atrial Fibrillation Monitoring
8.4.2 Bradycardia Monitoring
8.4.3 Tachycardia Monitoring
8.4.4 Post-Operative Monitoring
8.4.5 Premature Contraction Detection
8.4.6 Others
8.5 By Technology
8.5.1 Wireless Monitoring
8.5.2 Wired Monitoring
8.5.3 Cloud-Based Solutions
8.5.4 AI-Enabled Monitoring
8.5.5 Others
8.6 By Price Range
8.6.1 Low-End Devices
8.6.2 Mid-Range Devices
8.6.3 High-End Devices
8.7 By Region
8.7.1 Central Kuwait
8.7.2 Southern Kuwait
8.7.3 Northern Kuwait
8.7.4 Eastern Kuwait
8.7.5 Western Kuwait
8.7.6 Others
9. Kuwait Arrhythmia Monitoring Devices Market Competitive Analysis
9.1 Market Share of Key Players
9.2 Cross Comparison of Key Players
9.2.1 Company Name
9.2.2 Group Size (Large, Medium, or Small as per industry convention)
9.2.3 Revenue Growth Rate (Kuwait Market)
9.2.4 Market Penetration Rate (Devices Installed per 100,000 Population)
9.2.5 Customer Retention Rate (Annual %)
9.2.6 Average Selling Price (USD per Device)
9.2.7 Product Portfolio Breadth (Number of Arrhythmia Monitoring SKUs)
9.2.8 R&D Intensity (% of Revenue Spent on R&D)
9.2.9 Distribution Network Coverage (Number of Hospitals/Clinics Served)
9.2.10 Regulatory Approval Track Record (Number of Kuwait MOH Approvals)
9.2.11 Brand Recognition Score (Survey-Based Index)
9.3 SWOT Analysis of Top Players
9.4 Pricing Analysis
9.5 Detailed Profile of Major Companies
9.5.1 Medtronic plc
9.5.2 Abbott Laboratories
9.5.3 Boston Scientific Corporation
9.5.4 Biotronik SE & Co. KG
9.5.5 Koninklijke Philips N.V. (Philips Healthcare)
9.5.6 GE Healthcare
9.5.7 Siemens Healthineers AG
9.5.8 Nihon Kohden Corporation
9.5.9 Zoll Medical Corporation
9.5.10 Hillrom Services, Inc. (Welch Allyn)
9.5.11 iRhythm Technologies, Inc.
9.5.12 Cardiac Science Corporation
9.5.13 LivaNova PLC
9.5.14 AliveCor, Inc.
9.5.15 Spacelabs Healthcare (OSI Systems, Inc.)
9.5.16 Schiller AG
9.5.17 Bionet Co., Ltd.
9.5.18 Biotricity Inc.
9.5.19 Applied Cardiac Systems, Inc.
10. Kuwait Arrhythmia Monitoring Devices Market End-User Analysis
10.1 Procurement Behavior of Key Ministries
10.1.1 Ministry of Health
10.1.2 Ministry of Finance
10.1.3 Ministry of Defense
10.2 Corporate Spend on Infrastructure & Energy
10.2.1 Healthcare Infrastructure Investments
10.2.2 Technology Upgrades in Hospitals
10.3 Pain Point Analysis by End-User Category
10.3.1 Hospitals
10.3.2 Clinics
10.3.3 Home Care Providers
10.4 User Readiness for Adoption
10.4.1 Training Needs for Healthcare Professionals
10.4.2 Patient Education Initiatives
10.5 Post-Deployment ROI and Use Case Expansion
10.5.1 Monitoring Efficiency Improvements
10.5.2 Cost Savings from Early Detection
11. Kuwait Arrhythmia Monitoring Devices Market Future Size, 2025-2030
11.1 By Value
11.2 By Volume
11.3 By Average Selling Price
Go-To-Market Strategy Phase
1. Whitespace Analysis + Business Model Canvas
1.1 Market Gaps Identification
1.2 Business Model Framework
2. Marketing and Positioning Recommendations
2.1 Branding Strategies
2.2 Product USPs
3. Distribution Plan
3.1 Urban Retail vs Rural NGO Tie-Ups
4. Channel & Pricing Gaps
4.1 Underserved Routes
4.2 Pricing Bands
5. Unmet Demand & Latent Needs
5.1 Category Gaps
5.2 Consumer Segments
6. Customer Relationship
6.1 Loyalty Programs
6.2 After-Sales Service
7. Value Proposition
7.1 Sustainability
7.2 Integrated Supply Chains
8. Key Activities
8.1 Regulatory Compliance
8.2 Branding
8.3 Distribution Setup
9. Entry Strategy Evaluation
9.1 Domestic Market Entry Strategy
9.1.1 Product Mix
9.1.2 Pricing Band
9.1.3 Packaging
9.2 Export Entry Strategy
9.2.1 Target Countries
9.2.2 Compliance Roadmap
10. Entry Mode Assessment
10.1 JV, Greenfield, M&A, Distributor Model
11. Capital and Timeline Estimation
11.1 Capital Requirements
11.2 Timelines
12. Control vs Risk Trade-Off
12.1 Ownership vs Partnerships
13. Profitability Outlook
13.1 Breakeven Analysis
13.2 Long-Term Sustainability
14. Potential Partner List
14.1 Distributors
14.2 JVs
14.3 Acquisition Targets
15. Execution Roadmap
15.1 Phased Plan for Market Entry
15.1.1 Market Setup
15.1.2 Market Entry
15.1.3 Growth Acceleration
15.1.4 Scale & Stabilize
15.2 Key Activities and Milestones
15.2.1 Activity Timeline
15.2.2 Milestone Tracking
Research Methodology
Phase 1: Approach1
Desk Research
- Analysis of market reports from healthcare organizations and industry associations in Kuwait
- Review of published articles and white papers on arrhythmia monitoring technologies
- Examination of government health statistics and regulatory frameworks affecting medical devices
Primary Research
- Interviews with cardiologists and healthcare professionals specializing in arrhythmia management
- Surveys with medical device distributors and suppliers operating in the Kuwaiti market
- Focus groups with patients using arrhythmia monitoring devices to gather user experience insights
Validation & Triangulation
- Cross-validation of findings through multiple data sources, including clinical studies and market surveys
- Triangulation of insights from healthcare professionals, manufacturers, and regulatory bodies
- Sanity checks conducted through expert panel reviews to ensure data reliability
Phase 2: Market Size Estimation1
Top-down Assessment
- Estimation of the total addressable market based on Kuwait's healthcare expenditure and population demographics
- Segmentation of the market by device type, including wearable monitors and implantable devices
- Incorporation of trends in telemedicine and remote patient monitoring impacting device adoption
Bottom-up Modeling
- Collection of sales data from leading arrhythmia monitoring device manufacturers in Kuwait
- Estimation of market share based on unit sales and pricing strategies of key players
- Volume x price analysis to derive revenue projections for each device category
Forecasting & Scenario Analysis
- Multi-factor regression analysis considering factors such as aging population and prevalence of cardiovascular diseases
- Scenario modeling based on potential regulatory changes and technological advancements
- Development of baseline, optimistic, and pessimistic forecasts through 2030
Phase 3: CATI Sample Composition1
| Scope Item/Segment | Sample Size | Target Respondent Profiles |
|---|---|---|
| Cardiologists and Electrophysiologists | 100 | Cardiologists, Electrophysiologists |
| Healthcare Administrators | 60 | Hospital Administrators, Health Policy Makers |
| Medical Device Distributors | 50 | Sales Managers, Distribution Executives |
| Patients Using Monitoring Devices | 75 | Patients, Caregivers |
| Regulatory Bodies | 40 | Regulatory Officials, Compliance Officers |
Frequently Asked Questions
What is the current value of the Kuwait Arrhythmia Monitoring Devices Market?
The Kuwait Arrhythmia Monitoring Devices Market is valued at approximately USD 10 million, reflecting a five-year historical analysis. This growth is driven by the increasing prevalence of cardiovascular diseases and advancements in monitoring technology.