Region:Middle East
Author(s):Shubham
Product Code:KRAC2153
Pages:97
Published On:October 2025
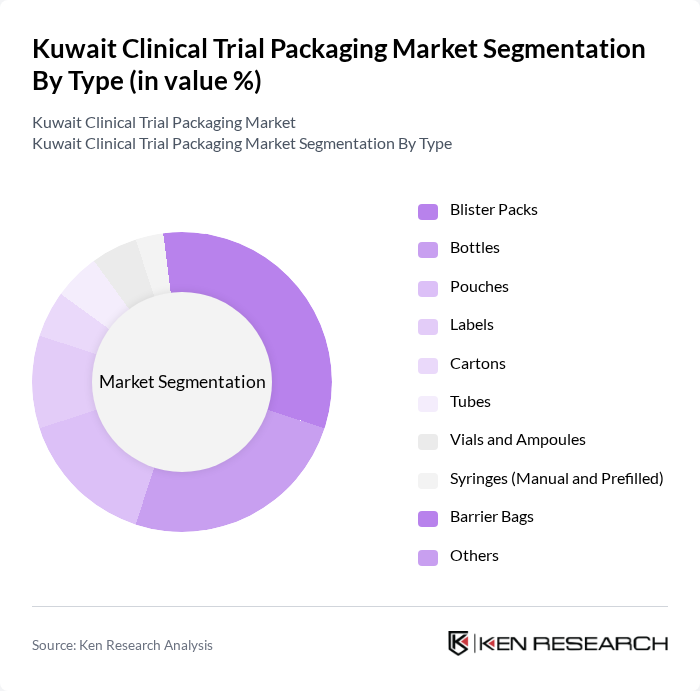
By Type:The market is segmented into various types of packaging solutions, including blister packs, bottles, pouches, labels, cartons, tubes, vials and ampoules, syringes (manual and prefilled), barrier bags, and others. Blister packs and bottles are the most widely used due to their effectiveness in protecting sensitive pharmaceutical products from environmental factors, ease of use, and ability to support patient adherence. The increasing adoption of serialization and traceability features, as well as sustainable packaging materials, is shaping the market landscape .
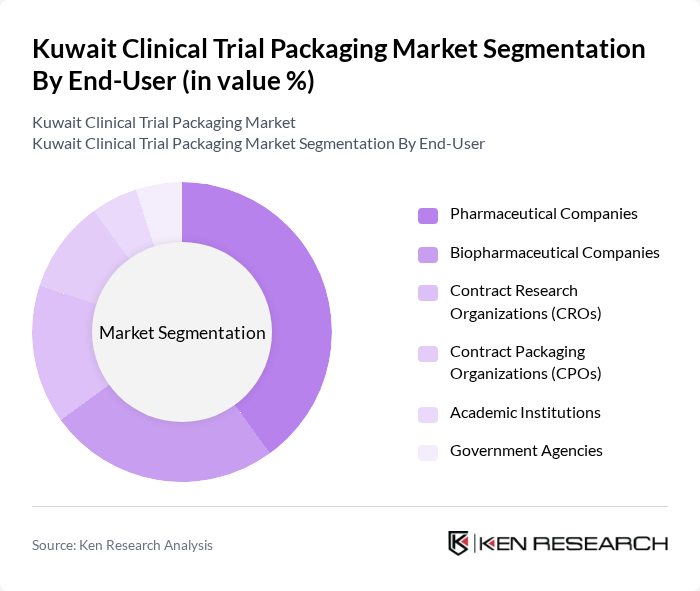
By End-User:The end-user segmentation includes pharmaceutical companies, biopharmaceutical companies, contract research organizations (CROs), contract packaging organizations (CPOs), academic institutions, and government agencies. Pharmaceutical companies are the leading end-users, driven by their need for reliable and compliant packaging solutions for clinical trials. The market is also witnessing increased participation from CROs and CPOs, reflecting the trend toward outsourcing and specialized packaging services to meet complex trial requirements .
The Kuwait Clinical Trial Packaging Market is characterized by a dynamic mix of regional and international players. Leading participants such as Catalent Pharma Solutions, PCI Pharma Services, Thermo Fisher Scientific (Patheon), Sharp Packaging Services, Almac Group, Gulf Pharmaceutical Industries (Julphar), Kuwait Pharmaceutical Industries (KPI), Gulf Biotech, Tjoapack, CCL Industries, ACG Worldwide, United Pharmaceutical Manufacturing Company, UDG Healthcare, Mediserv, Kuwait Institute for Scientific Research contribute to innovation, geographic expansion, and service delivery in this space.
The future of the Kuwait clinical trial packaging market appears promising, driven by advancements in technology and a growing emphasis on sustainability. As the healthcare sector continues to evolve, the integration of digital technologies in packaging processes is expected to enhance efficiency and traceability. Additionally, the increasing focus on personalized medicine will likely drive demand for innovative packaging solutions tailored to specific patient needs, fostering a more adaptable and responsive market environment.
| Segment | Sub-Segments |
|---|---|
| By Type | Blister Packs Bottles Pouches Labels Cartons Tubes Vials and Ampoules Syringes (Manual and Prefilled) Barrier Bags Others |
| By End-User | Pharmaceutical Companies Biopharmaceutical Companies Contract Research Organizations (CROs) Contract Packaging Organizations (CPOs) Academic Institutions Government Agencies |
| By Packaging Material | Plastic Glass Metal Paperboard Cyclo Olefin Copolymer/Polymer |
| By Application | Oncology Trials Cardiovascular Trials Neurology Trials Infectious Disease Trials Other Therapeutic Areas |
| By Distribution Channel | Direct Sales Online Sales Distributors |
| By Regulatory Compliance Level | Fully Compliant Partially Compliant Non-Compliant |
| By Price Range | Low Price Mid Price High Price |
| Scope Item/Segment | Sample Size | Target Respondent Profiles |
|---|---|---|
| Pharmaceutical Clinical Trials | 100 | Clinical Trial Managers, Regulatory Affairs Specialists |
| Biotechnology Clinical Trials | 60 | Research Scientists, Project Coordinators |
| Medical Device Trials | 50 | Product Development Managers, Quality Assurance Officers |
| Contract Research Organizations (CROs) | 40 | Business Development Managers, Operations Directors |
| Regulatory Compliance in Trials | 45 | Compliance Officers, Legal Advisors |
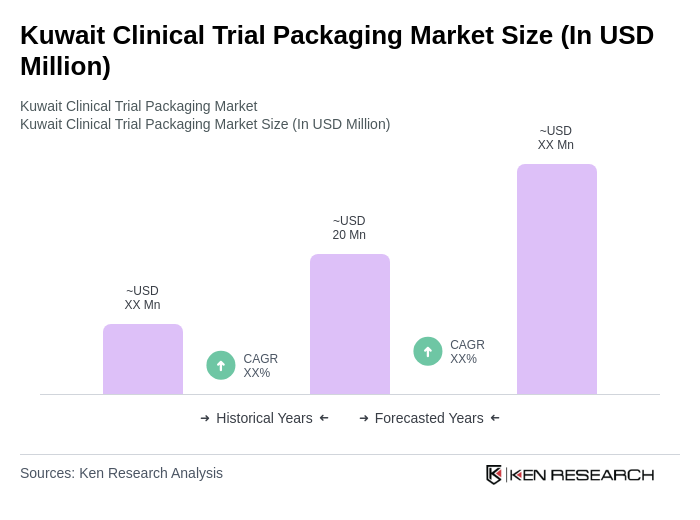
The Kuwait Clinical Trial Packaging Market is valued at approximately USD 20 million, based on a five-year historical analysis. This market is experiencing growth due to the increasing number of clinical trials and the demand for efficient packaging solutions.