Region:Asia
Author(s):Shubham
Product Code:KRAC2820
Pages:89
Published On:October 2025
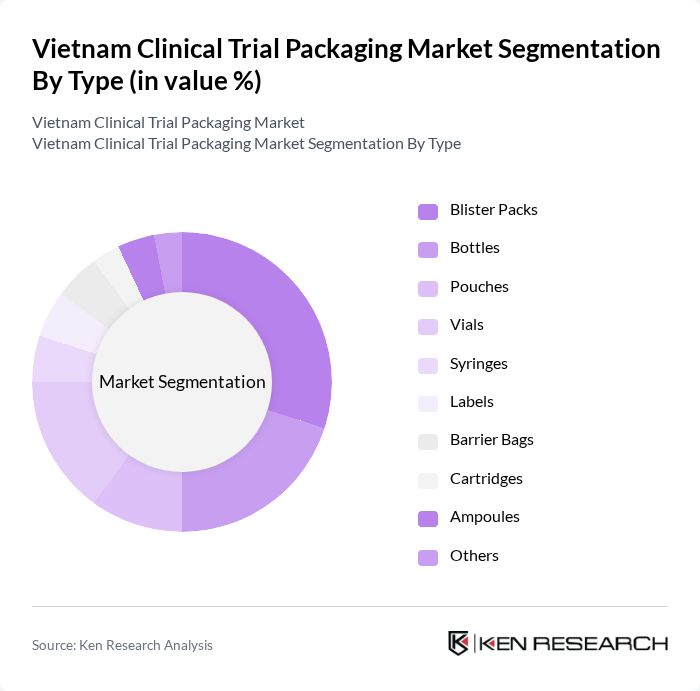
By Type:The clinical trial packaging market is segmented into blister packs, bottles, pouches, vials, syringes, labels, barrier bags, cartridges, ampoules, and others. Among these,blister packsandvialsare particularly prominent due to their effectiveness in protecting sensitive materials, ensuring dosage accuracy, and supporting compliance with regulatory standards. The demand for these packaging types is driven by their ability to enhance patient compliance, reduce contamination risk, and support traceability, which is increasingly important for clinical trial logistics .
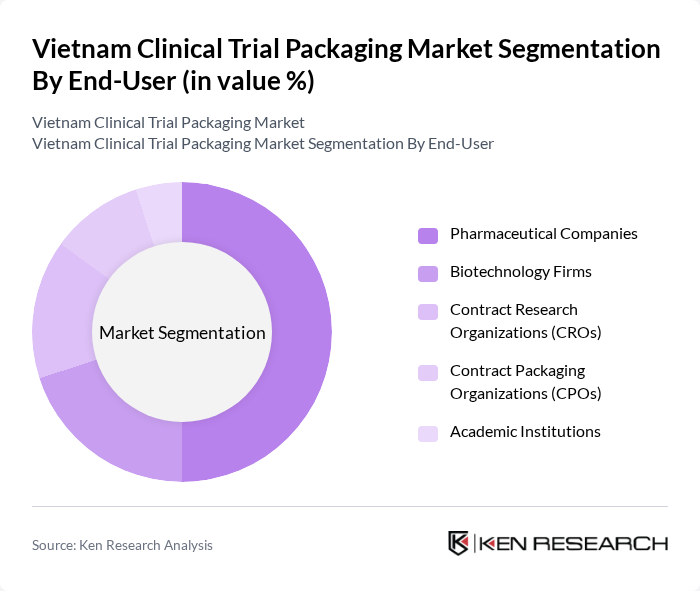
By End-User:The end-user segmentation includes pharmaceutical companies, biotechnology firms, contract research organizations (CROs), contract packaging organizations (CPOs), and academic institutions.Pharmaceutical companiesare the leading end-users, driven by their need for reliable, compliant packaging solutions to support clinical trials and product launches. The increasing number of drug development projects and the growing presence of international sponsors are further propelling demand for clinical trial packaging among these entities .
The Vietnam Clinical Trial Packaging Market is characterized by a dynamic mix of regional and international players. Leading participants such as Vina Packaging Co., Ltd., Amcor Flexibles Vietnam, Huhtamaki Vietnam Ltd., Sonoco Products Company, WestRock Company, Sealed Air Corporation, Berry Global, Inc., Smurfit Kappa Group, Mondi Group, Constantia Flexibles, Schott AG, Nipro Corporation, Catalent Pharma Solutions, PCI Pharma Services, Sharp Packaging Services, Almac Group, Tjoapack Group, CCL Industries, ACG Capsules, UDG Healthcare, and Marken contribute to innovation, geographic expansion, and service delivery in this space.
The future of Vietnam's clinical trial packaging market appears promising, driven by ongoing advancements in technology and increasing international collaboration. As the country continues to attract foreign investment, the demand for innovative packaging solutions is expected to rise. Additionally, the focus on patient-centric designs and sustainable practices will likely shape the market landscape, encouraging companies to adopt eco-friendly materials and technologies that enhance patient safety and compliance in clinical trials.
| Segment | Sub-Segments |
|---|---|
| By Type | Blister Packs Bottles Pouches Vials Syringes Labels Barrier Bags Cartridges Ampoules Others |
| By End-User | Pharmaceutical Companies Biotechnology Firms Contract Research Organizations (CROs) Contract Packaging Organizations (CPOs) Academic Institutions |
| By Application | Oncology Trials Cardiovascular Trials Infectious Disease Trials Neurology Trials Other Therapeutic Areas |
| By Distribution Channel | Direct Sales Online Sales Distributors |
| By Packaging Material | Plastic Glass Metal Cyclo Olefin Copolymer Cyclo Olefin Polymer |
| By Regulatory Compliance Level | Fully Compliant Partially Compliant |
| By Price Range | Low Medium High |
| Scope Item/Segment | Sample Size | Target Respondent Profiles |
|---|---|---|
| Pharmaceutical Clinical Trials | 100 | Clinical Trial Managers, Regulatory Affairs Specialists |
| Biotechnology Clinical Trials | 60 | Research Scientists, Project Managers |
| Medical Device Trials | 50 | Product Development Managers, Quality Assurance Officers |
| Contract Research Organizations (CROs) | 40 | Operations Directors, Business Development Managers |
| Regulatory Compliance in Trials | 70 | Compliance Officers, Legal Advisors |
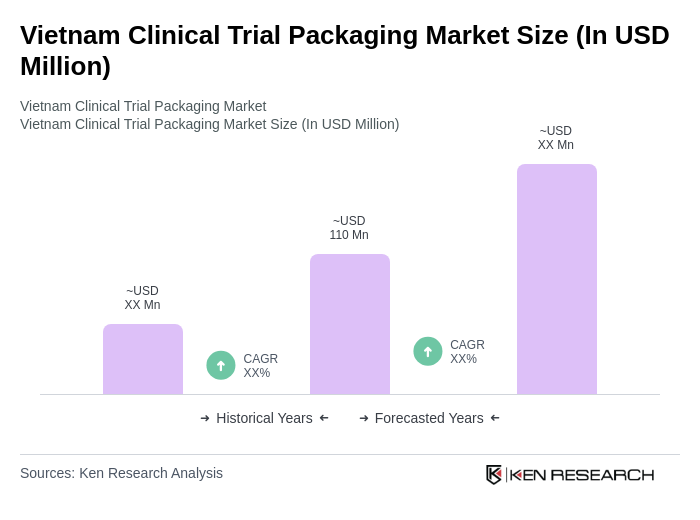
The Vietnam Clinical Trial Packaging Market is valued at approximately USD 110 million, reflecting its significant share within the broader clinical trials and healthcare packaging sectors, driven by increasing clinical trials and investments in the pharmaceutical industry.