Region:Asia
Author(s):Rebecca
Product Code:KRAA6085
Pages:97
Published On:January 2026
By Type:The market is segmented into various types, including API Manufacturing, Finished Dosage Formulations, Biologics Manufacturing Services, Pharmaceutical Manufacturing Services, and Others. Each of these segments plays a crucial role in meeting the diverse needs of pharmaceutical companies.
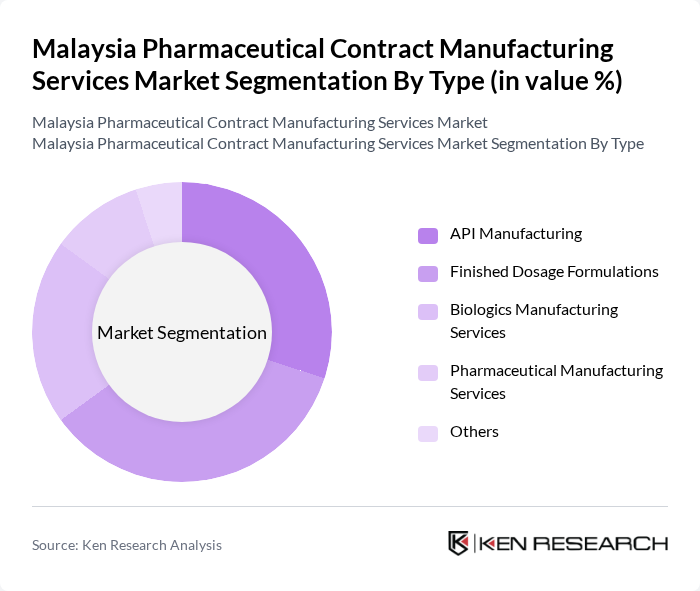
The Finished Dosage Formulations segment is currently dominating the market due to the increasing demand for ready-to-use medications and the growing trend of outsourcing manufacturing processes, with particular strength in solid dosage forms, generic drug production, and halal-certified pharmaceuticals. This segment benefits from the rise in chronic diseases, which necessitate a steady supply of various dosage forms. Additionally, the shift towards personalized medicine and the need for efficient production processes further enhance the attractiveness of this segment.
By End-User:The market is segmented based on end-users, including Large Pharmaceutical Companies, Small & Mid-size Pharmaceutical Companies, Generic Pharmaceutical Companies, and Biopharmaceutical Companies. Each segment has unique requirements and contributes differently to the overall market dynamics.
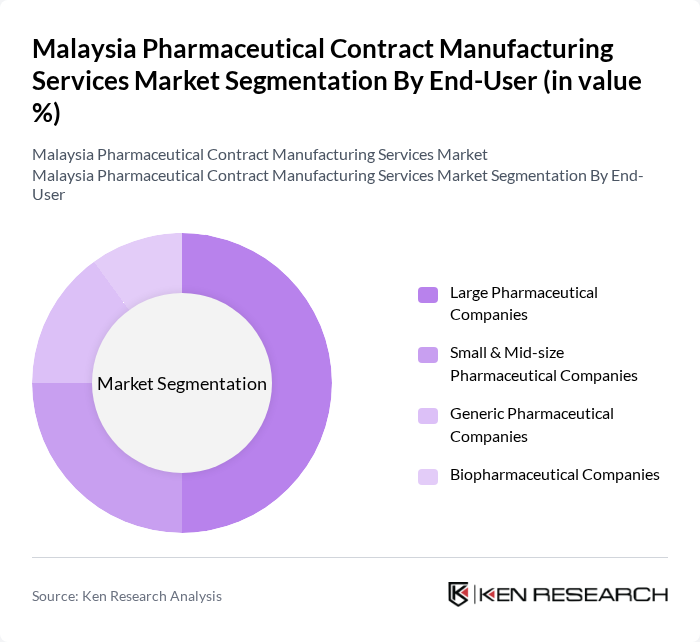
Large Pharmaceutical Companies dominate the market due to their extensive resources, established distribution networks, and strong brand recognition, with pharmaceutical and biopharmaceutical companies representing the largest end-use segment. These companies often engage in contract manufacturing to optimize production costs and focus on core competencies such as research and development. Their ability to leverage economies of scale further solidifies their leading position in the market.
The Malaysia Pharmaceutical Contract Manufacturing Services Market is characterized by a dynamic mix of regional and international players. Leading participants such as Pharmaniaga Berhad, Duopharma Biotech Berhad, Hovid Berhad, Kotra Pharma (M) Sdn Bhd, CCM Duopharma Biotech Berhad, Apex Healthcare Berhad, YSP Southeast Asia Holding Berhad, Pharmaniaga Logistics Sdn Bhd, Biocon Sdn Bhd, SPP Pharma Sdn Bhd, Sandoz (Malaysia) Sdn Bhd, Merck Sharp & Dohme (Malaysia) Sdn Bhd, GSK Malaysia Sdn Bhd, Sanofi-Aventis (Malaysia) Sdn Bhd, Pfizer Malaysia Sdn Bhd contribute to innovation, geographic expansion, and service delivery in this space.
The future of the Malaysian pharmaceutical contract manufacturing services market appears promising, driven by increasing demand for generics and government support for local production. As healthcare expenditure rises, manufacturers are likely to invest in advanced technologies to enhance production efficiency. Additionally, the trend towards outsourcing manufacturing will continue, allowing local firms to expand their capabilities and focus on core competencies, ultimately fostering a more competitive landscape in the region.
| Segment | Sub-Segments |
|---|---|
| By Type | API Manufacturing Finished Dosage Formulations Biologics Manufacturing Services Pharmaceutical Manufacturing Services Others |
| By End-User | Large Pharmaceutical Companies Small & Mid-size Pharmaceutical Companies Generic Pharmaceutical Companies Biopharmaceutical Companies |
| By Product Formulation | Solid Dosage Forms Semi-solid Formulations Liquid Formulations Injectables Others |
| By Therapeutic Area | Cardiovascular Oncology Neurology Infectious Diseases Others |
| By Scale of Operation | Preclinical & Clinical Phase Commercial Scale Manufacturing |
| By Region | Klang Valley (Selangor, Kuala Lumpur) Penang East Malaysia (Sabah, Sarawak) Others |
| Scope Item/Segment | Sample Size | Target Respondent Profiles |
|---|---|---|
| Pharmaceutical Contract Manufacturing | 120 | Operations Managers, Business Development Executives |
| Regulatory Compliance in Manufacturing | 100 | Quality Assurance Managers, Regulatory Affairs Specialists |
| Market Trends in Biopharmaceuticals | 80 | Research and Development Heads, Product Managers |
| Outsourcing Strategies in Pharmaceuticals | 70 | Procurement Officers, Supply Chain Managers |
| Emerging Technologies in Manufacturing | 60 | IT Managers, Innovation Leads |
The Malaysia Pharmaceutical Contract Manufacturing Services Market is valued at approximately USD 3.1 billion, reflecting significant growth driven by the demand for generic drugs, chronic disease prevalence, and government support for local manufacturing capabilities.