Region:Middle East
Author(s):Dev
Product Code:KRAB2348
Pages:87
Published On:January 2026
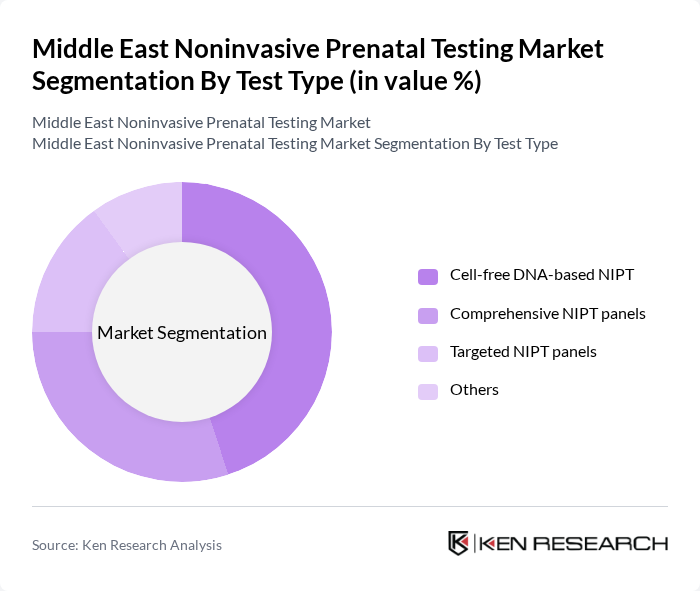
By Test Type:The market is segmented into various test types, including Cell-free DNA-based NIPT, Comprehensive NIPT panels, Targeted NIPT panels, and Others. Cell-free DNA-based NIPT is the most widely adopted technology platform in the region, supported by strong clinical evidence for trisomy 21, 18, and 13 screening and its favorable safety profile. Comprehensive NIPT panels, which extend analysis to sex chromosome aneuploidies and selected microdeletion or monogenic disease panels, are gaining traction among high-risk and self?pay populations who seek broader genomic information and are increasingly offered through partnerships between regional laboratories and global test developers. Targeted NIPT panels remain relevant for payers and providers focused on cost?effective screening for a limited set of high?prevalence conditions, while the Others category includes emerging approaches such as combined NIPT plus carrier screening workflows and reflex testing algorithms.
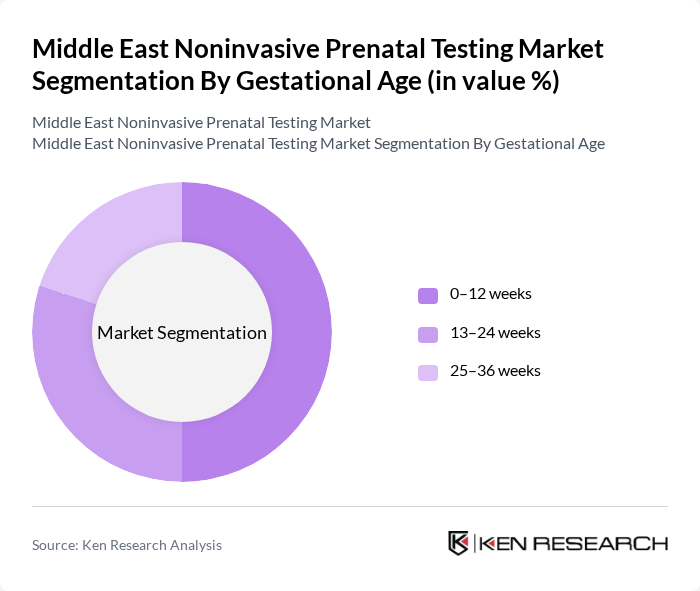
By Gestational Age:The market is categorized based on gestational age into three segments: 0–12 weeks, 13–24 weeks, and 25–36 weeks. In practice, most NIPT is ordered from around 10 weeks of gestation onward, and the 13–24 weeks segment currently accounts for the largest share of regional revenue, reflecting common clinical workflows in which women are referred for NIPT following first?trimester ultrasound or biochemical screening. However, the 0–12 weeks segment is the fastest growing, as earlier prenatal visits, rising awareness of early fetal anomaly detection, and the availability of validated NIPT protocols from 10 weeks support a shift toward earlier screening and more timely pregnancy decision?making. The 25–36 weeks segment remains comparatively smaller and is largely associated with late presenters, repeat testing in specific clinical scenarios, and complex high?risk pregnancies managed in tertiary centers.
The Middle East Noninvasive Prenatal Testing Market is characterized by a dynamic mix of regional and international players. Leading participants such as Illumina, Inc., Natera, Inc., Roche Diagnostics (Roche Holding AG), Laboratory Corporation of America Holdings (Labcorp), Quest Diagnostics Incorporated, PerkinElmer, Inc., Eurofins Scientific SE, Myriad Genetics, Inc., BGI Genomics Co., Ltd., MedGenome Inc., Centogene N.V., Qiagen N.V., CooperSurgical, Inc., Regional hospital laboratories (selected examples), Emerging Middle East-based NIPT providers contribute to innovation, geographic expansion, and service delivery in this space.
The future of the Middle East noninvasive prenatal testing market appears promising, driven by ongoing advancements in technology and increasing healthcare investments. As the region's healthcare expenditure is projected to reach $220 billion in future, the focus on maternal health will likely intensify. Additionally, the integration of telemedicine and digital health solutions is expected to enhance access to prenatal care, particularly in underserved areas, fostering a more inclusive healthcare environment for expectant mothers.
| Segment | Sub-Segments |
|---|---|
| By Test Type | Cell-free DNA-based NIPT Comprehensive NIPT panels Targeted NIPT panels Others |
| By Gestational Age | –12 weeks –24 weeks –36 weeks |
| By Condition Screened | Trisomy 21 (Down syndrome) Trisomy 18 and Trisomy 13 Sex chromosome aneuploidies Microdeletions and other genetic disorders |
| By Technology Platform | Next-generation sequencing (NGS) PCR-based methods Microarray-based methods Others |
| By End User | Hospitals and maternity centers Diagnostic and reference laboratories Specialized fertility and women’s health clinics Others |
| By Country | Saudi Arabia United Arab Emirates Qatar Kuwait Oman Rest of Middle East |
| Scope Item/Segment | Sample Size | Target Respondent Profiles |
|---|---|---|
| Obstetricians and Gynecologists | 120 | Healthcare Providers, Specialists in Prenatal Care |
| Laboratory Managers | 90 | Technical Directors, Laboratory Technicians |
| Expectant Parents | 140 | First-time Parents, Couples Considering NIPT |
| Healthcare Policy Makers | 60 | Government Officials, Health Program Directors |
| Genetic Counselors | 60 | Genetic Counselors, Patient Educators |
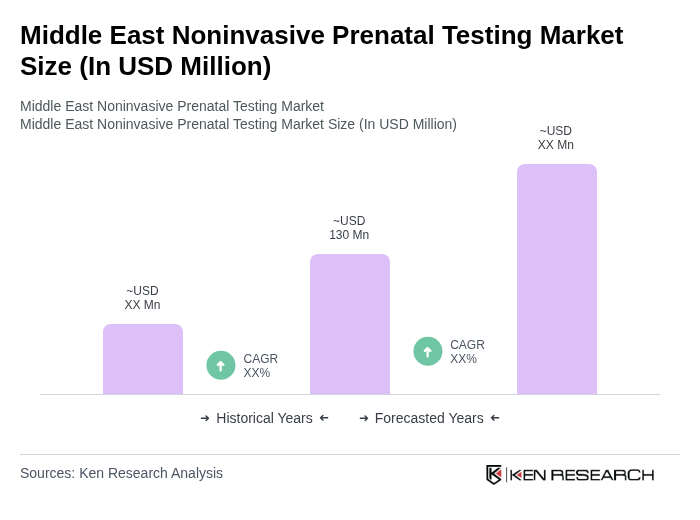
The Middle East Noninvasive Prenatal Testing Market is valued at approximately USD 130 million, reflecting a significant growth trend driven by advancements in technology and increased awareness of prenatal health among expectant parents in the region.