Region:Asia
Author(s):Geetanshi
Product Code:KRAC9467
Pages:83
Published On:November 2025
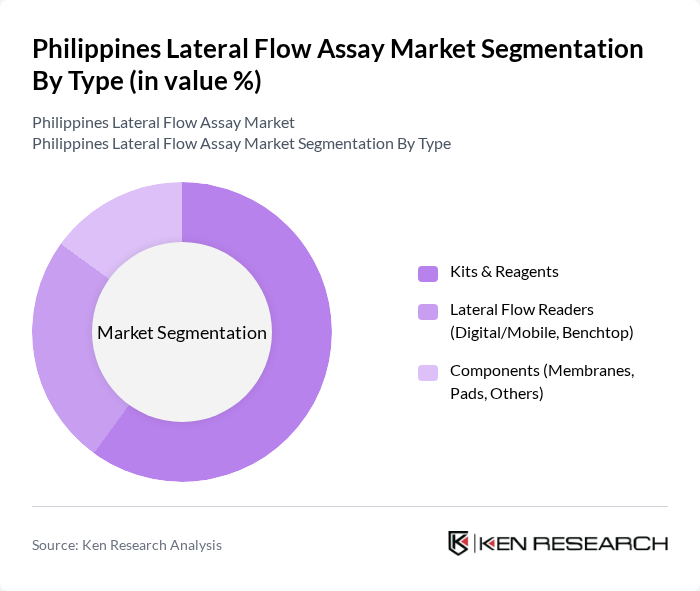
By Type:The market is segmented into Kits & Reagents, Lateral Flow Readers (Digital/Mobile, Benchtop), and Components (Membranes, Pads, Others). Among these, Kits & Reagents dominate the market due to their essential role in the testing process, providing the necessary materials for conducting tests. The increasing prevalence of infectious diseases, the growing trend of home testing, and the need for rapid results in both clinical and non-clinical settings have further fueled the demand for these kits. Lateral Flow Readers are also gaining traction, especially digital/mobile versions, as they enhance the testing experience by providing quick and accurate results. Components such as membranes and pads remain critical for manufacturers and laboratory use .
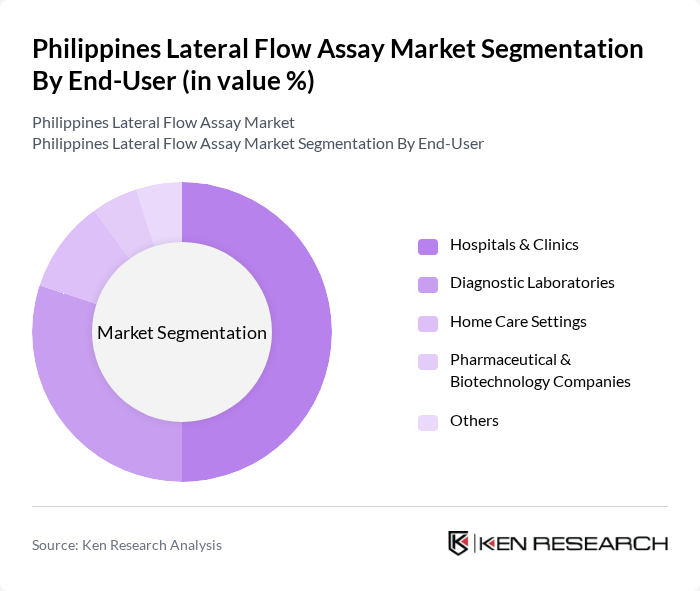
By End-User:The end-user segmentation includes Hospitals & Clinics, Diagnostic Laboratories, Home Care Settings, Pharmaceutical & Biotechnology Companies, and Others. Hospitals & Clinics are the leading end-users, driven by the need for rapid testing solutions in patient care. The increasing number of diagnostic laboratories also contributes to the market, as they require lateral flow assays for various testing applications. Home care settings are emerging as a significant segment due to the growing trend of self-testing among consumers, particularly for infectious diseases and chronic condition monitoring. Pharmaceutical and biotechnology companies utilize lateral flow assays for research, development, and quality control purposes .
The Philippines Lateral Flow Assay Market is characterized by a dynamic mix of regional and international players. Leading participants such as Abbott Laboratories, Roche Diagnostics, Siemens Healthineers, QuidelOrtho Corporation, BD (Becton, Dickinson and Company), Bio-Rad Laboratories, Cepheid, Hologic, Inc., Medline Industries, LP, Thermo Fisher Scientific, GenMark Diagnostics, Ortho Clinical Diagnostics, Mylab Discovery Solutions Pvt. Ltd., Eiken Chemical Co., Ltd., AccuBioTech Co., Ltd., SD Biosensor, Inc., Bioeasy Biotechnology Co., Ltd., CTK Biotech, Inc., Wondfo Biotech Co., Ltd., Fujirebio, Inc. contribute to innovation, geographic expansion, and service delivery in this space.
The future of the lateral flow assay market in the Philippines appears promising, driven by ongoing advancements in technology and increasing healthcare investments. As the government continues to prioritize healthcare infrastructure, the integration of digital health technologies is expected to enhance diagnostic capabilities. Additionally, the growing trend towards home-based testing solutions will likely expand the market further, providing patients with convenient access to rapid diagnostic tests and improving overall health outcomes.
| Segment | Sub-Segments |
|---|---|
| By Type | Kits & Reagents Lateral Flow Readers (Digital/Mobile, Benchtop) Components (Membranes, Pads, Others) |
| By End-User | Hospitals & Clinics Diagnostic Laboratories Home Care Settings Pharmaceutical & Biotechnology Companies Others |
| By Application | Infectious Disease Testing (e.g., COVID-19, Influenza, Tuberculosis) Pregnancy & Fertility Testing Drug Abuse Testing Veterinary Diagnostics Food Safety & Environmental Testing Others |
| By Distribution Channel | Direct Sales Online Sales Retail Pharmacies Others |
| By Region | Luzon Visayas Mindanao |
| By Technology | Colloidal Gold Fluorescent Labels Latex Beads Others |
| By Policy Support | Government Subsidies Tax Incentives Research Grants Others |
| Scope Item/Segment | Sample Size | Target Respondent Profiles |
|---|---|---|
| Healthcare Providers | 100 | Doctors, Nurses, Laboratory Technicians |
| Manufacturers of Lateral Flow Assays | 60 | Product Managers, R&D Managers |
| Distributors and Suppliers | 50 | Sales Managers, Supply Chain Coordinators |
| Regulatory Bodies | 40 | Policy Makers, Compliance Officers |
| End-users (Patients) | 80 | Patients, Caregivers, Health Advocates |
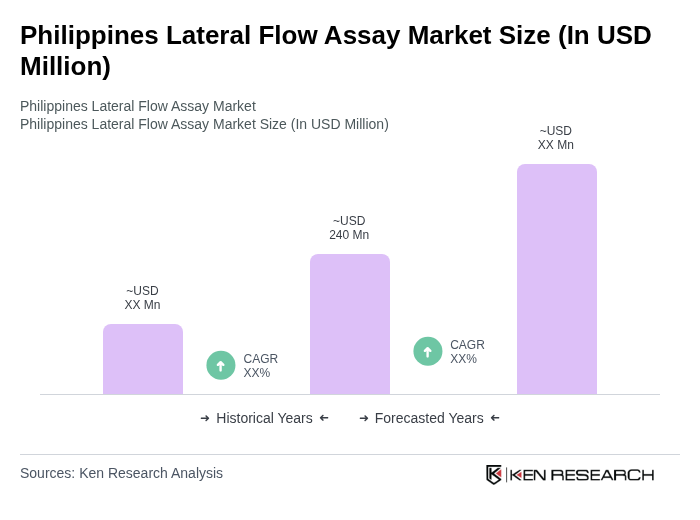
The Philippines Lateral Flow Assay Market is valued at approximately USD 240 million, reflecting a significant increase driven by the rising demand for rapid diagnostic tests, particularly in response to infectious diseases and the COVID-19 pandemic.