GCC Lateral Flow Assay Market Overview
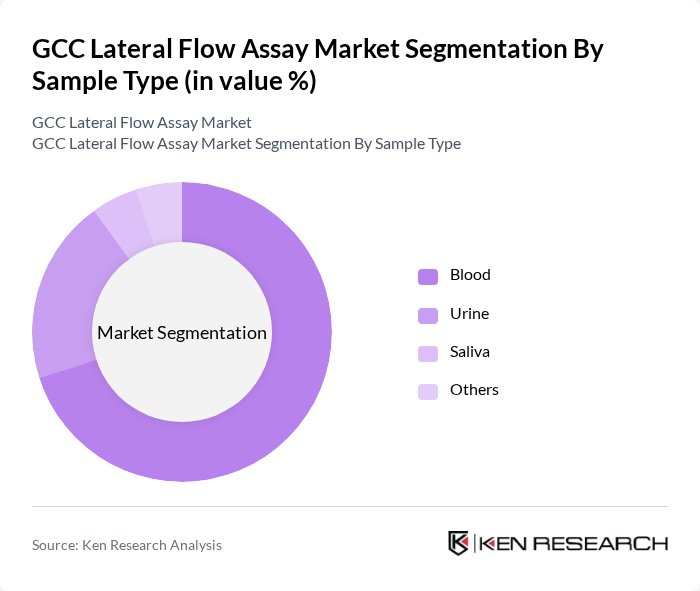
- The GCC Lateral Flow Assay Market is valued at USD 1.1 billion, based on a five-year historical analysis. This growth is primarily driven by the increasing prevalence of infectious diseases, rising demand for rapid diagnostic tests, and growing adoption of point-of-care testing among healthcare professionals and patients. The market has seen significant advancements, including smartphone-compatible readers, AI-powered analysis, and multiplex-capable assays, which have improved accuracy, connectivity, and diagnostic efficiency. The expansion of home-based and self-testing kits, as well as integration with telehealth platforms, are notable trends shaping market growth.
- Key players in this market include Saudi Arabia, the United Arab Emirates, and Qatar. These countries dominate the market due to their advanced healthcare infrastructure, substantial investment in healthcare technologies, and robust government initiatives aimed at enhancing diagnostic capabilities. The presence of major healthcare facilities, a growing population, and increased focus on preventive healthcare further contribute to market expansion in these regions.
- The Saudi Food and Drug Authority (SFDA) issued the "Guidelines for Registration of In-Vitro Diagnostic Medical Devices, 2023," mandating the registration and use of rapid diagnostic tests in primary healthcare settings. This regulation requires all rapid diagnostic test manufacturers and distributors to obtain SFDA approval, adhere to performance and safety standards, and ensure traceability of products. The initiative aims to improve early detection and treatment of infectious and chronic diseases, supporting the broader strategy to modernize the healthcare system and enhance public health outcomes.
GCC Lateral Flow Assay Market Segmentation
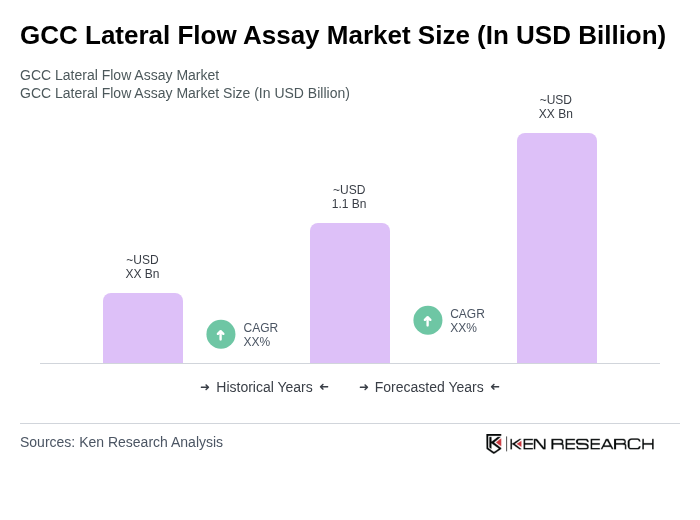
By Product Type:The product type segmentation includes Kits & Reagents, Readers/Analyzers, and Others. Kits & Reagents remain the dominant subsegment, reflecting their essential role in conducting diagnostic tests across clinical and home settings. The surge in demand for home testing kits, combined with the convenience and accessibility they offer, has reinforced their market leadership. Readers/Analyzers are increasingly adopted in clinical environments, driven by the need for rapid, accurate, and digitally integrated results.
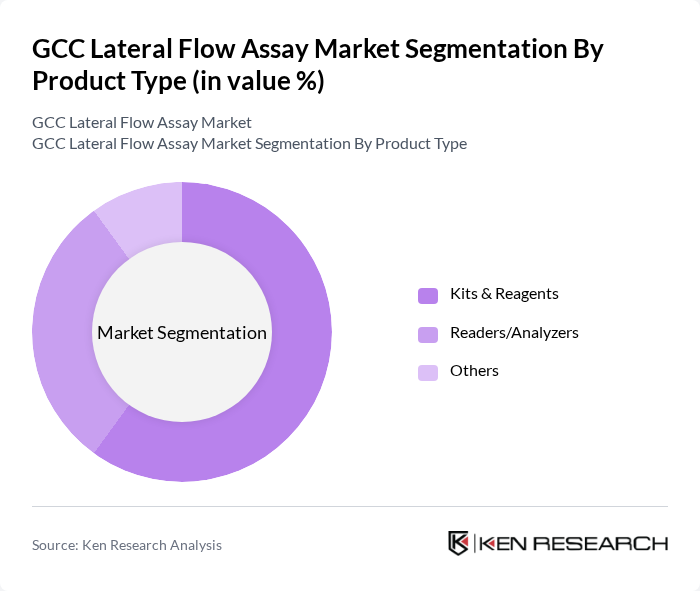
By Sample Type:The sample type segmentation includes Blood, Urine, Saliva, and Others. Blood samples account for the largest share, owing to their widespread use in infectious disease detection, cardiac marker analysis, and other critical diagnostics. The reliability and accuracy of blood-based tests make them the preferred choice for healthcare providers. Urine and saliva samples are gaining traction, particularly in home and self-testing scenarios, but currently represent a smaller share compared to blood.
GCC Lateral Flow Assay Market Competitive Landscape
The GCC Lateral Flow Assay Market is characterized by a dynamic mix of regional and international players. Leading participants such as Abbott Laboratories, Roche Diagnostics, Siemens Healthineers, QuidelOrtho Corporation, BD (Becton, Dickinson and Company), Bio-Rad Laboratories, Cepheid, Hologic, Inc., Thermo Fisher Scientific, Mylab Discovery Solutions, BioSure UK, SDBiosensor, Alere (now part of Abbott), BTNX Inc., Omega Diagnostics Group PLC contribute to innovation, geographic expansion, and service delivery in this space.
GCC Lateral Flow Assay Market Industry Analysis
Growth Drivers
- Increasing Prevalence of Infectious Diseases:The GCC region has witnessed a significant rise in infectious diseases, with the World Health Organization reporting over 1.6 million cases of communicable diseases in future. This surge has heightened the demand for rapid diagnostic tools, particularly lateral flow assays, which provide quick results. The increasing incidence of diseases such as COVID-19, influenza, and hepatitis is driving healthcare providers to adopt these tests, thereby expanding the market significantly.
- Rising Demand for Rapid Diagnostic Tests:The GCC healthcare sector is experiencing a paradigm shift towards rapid diagnostic tests, with a reported increase of 35% in demand for such tests in future. This trend is fueled by the need for timely diagnosis and treatment, especially in emergency settings. Lateral flow assays, known for their ease of use and quick turnaround times, are becoming essential tools in hospitals and clinics, further propelling market growth in the region.
- Technological Advancements in Assay Development:The lateral flow assay market is benefiting from continuous technological innovations, with investments in R&D reaching approximately $600 million in future. These advancements include improved sensitivity and specificity of tests, enabling better disease detection. The integration of nanotechnology and microfluidics is enhancing assay performance, making them more reliable and efficient, thus attracting more healthcare providers to adopt these solutions in the GCC region.
Market Challenges
- High Competition Among Key Players:The GCC lateral flow assay market is characterized by intense competition, with over 60 companies vying for market share. This saturation leads to price wars, which can erode profit margins. Companies are compelled to invest heavily in marketing and product differentiation to maintain their competitive edge, which can strain financial resources and impact overall market stability.
- Regulatory Hurdles in Product Approval:Navigating the regulatory landscape in the GCC can be challenging, with approval processes for new lateral flow assays often taking up to 20 months. Stringent regulations imposed by health authorities require comprehensive clinical validation and quality control measures. These hurdles can delay product launches and increase costs, posing a significant challenge for companies looking to enter or expand in the market.
GCC Lateral Flow Assay Market Future Outlook
The future of the GCC lateral flow assay market appears promising, driven by ongoing innovations and a growing emphasis on point-of-care testing. As healthcare systems increasingly prioritize rapid diagnostics, the integration of digital technologies and telemedicine is expected to enhance the accessibility and efficiency of these tests. Furthermore, the rising focus on personalized medicine will likely lead to the development of tailored assays, catering to specific patient needs and conditions, thereby expanding market potential.
Market Opportunities
- Expansion into Emerging Markets:The GCC region presents significant opportunities for lateral flow assay manufacturers to expand into emerging markets, where healthcare infrastructure is rapidly developing. With a projected increase in healthcare spending of 12% annually, companies can tap into new customer bases, enhancing their market presence and driving revenue growth.
- Development of Novel Assays for Diverse Applications:There is a growing opportunity for the development of novel lateral flow assays targeting various applications, including chronic disease management and infectious disease detection. With an estimated market demand for such innovations reaching $250 million by future, companies that invest in R&D for diverse applications can significantly enhance their competitive advantage and market share.