About the Report
Base Year 2024Poland Healthcare Diagnostics and Labs Market Overview
- The Poland Healthcare Diagnostics and Labs Market is valued at USD 5.5 billion, based on a five-year historical analysis. This growth is primarily driven by the increasing prevalence of chronic diseases, advancements in diagnostic technologies, and a growing emphasis on preventive healthcare. The market benefits from rising healthcare funding, an aging population, and the adoption of advanced diagnostic tools such as molecular diagnostics and AI-driven solutions. The demand for accurate and timely diagnostic services continues to propel the sector, making it a critical component of the national healthcare system.
- Warsaw, Kraków, and Wroc?aw are the dominant cities in the Poland Healthcare Diagnostics and Labs Market. Warsaw, as the capital, hosts numerous healthcare facilities and research institutions, while Kraków and Wroc?aw benefit from their strong academic presence and healthcare infrastructure. These cities are pivotal in driving innovation and accessibility in diagnostic services, contributing significantly to market growth.
- The regulation titled “Rozporz?dzenie Ministra Zdrowia z dnia 15 kwietnia 2023 r. w sprawie elektronicznej dokumentacji medycznej” (Regulation of the Minister of Health of April 15, 2023, on Electronic Medical Documentation) mandates the integration of electronic health records (EHR) across all healthcare facilities in Poland. Issued by the Ministry of Health, this regulation requires healthcare providers, including diagnostic laboratories, to implement standardized EHR systems, ensuring secure data sharing, improved patient care efficiency, and comprehensive access to patient histories. The regulation aims to streamline operations and foster collaboration among healthcare providers, ultimately benefiting patient outcomes.
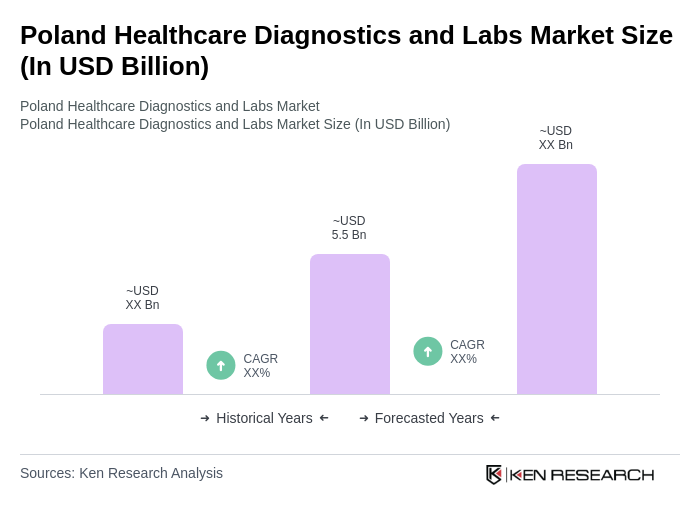
Poland Healthcare Diagnostics and Labs Market Segmentation
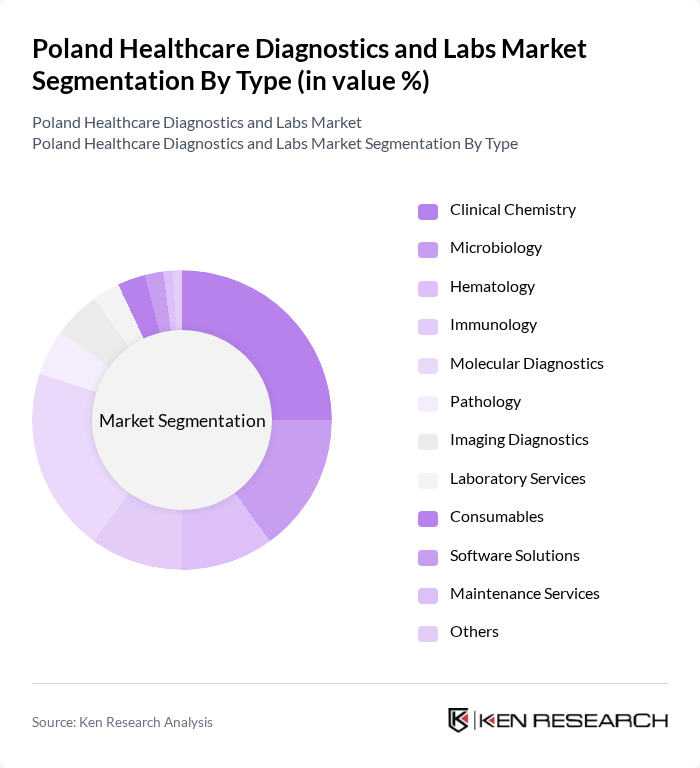
By Type:The market is segmented into various types, including Clinical Chemistry, Microbiology, Hematology, Immunology, Molecular Diagnostics, Pathology, Imaging Diagnostics, Laboratory Services, Consumables, Software Solutions, Maintenance Services, and Others. Each of these segments plays a crucial role in the overall diagnostics landscape, with specific applications and technologies driving their growth.
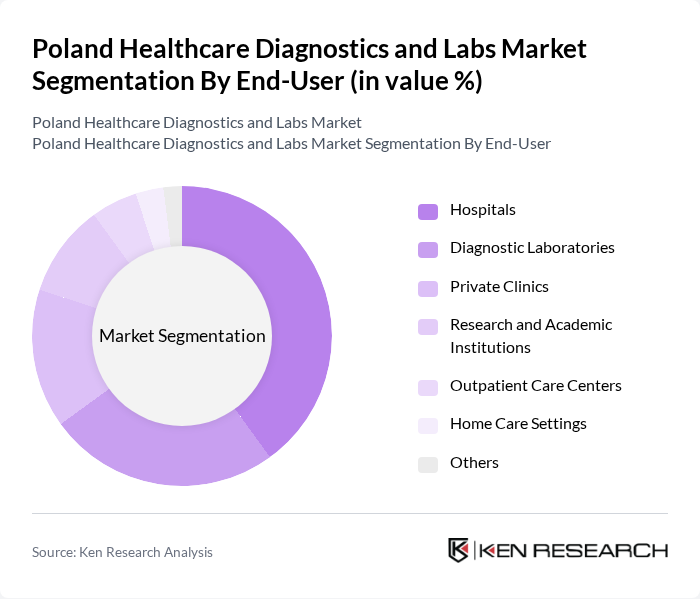
By End-User:The end-user segmentation includes Hospitals, Diagnostic Laboratories, Private Clinics, Research and Academic Institutions, Outpatient Care Centers, Home Care Settings, and Others. Each end-user category has distinct needs and preferences, influencing the demand for various diagnostic services and technologies.
Poland Healthcare Diagnostics and Labs Market Competitive Landscape
The Poland Healthcare Diagnostics and Labs Market is characterized by a dynamic mix of regional and international players. Leading participants such as Diagnostyka S.A., Synevo Poland, Grupa Alab, Voxel S.A., PZU Zdrowie, Roche Diagnostics Polska, Siemens Healthineers Poland, Abbott Laboratories Poland, Philips Healthcare Poland, GE Healthcare Poland, Canon Medical Systems Poland, BioMérieux Poland, Mindray Medical Poland, Becton, Dickinson and Company Poland, Thermo Fisher Scientific Poland contribute to innovation, geographic expansion, and service delivery in this space.
Poland Healthcare Diagnostics and Labs Market Industry Analysis
Growth Drivers
- Increasing Prevalence of Chronic Diseases:The rise in chronic diseases such as diabetes and cardiovascular conditions is a significant growth driver for the healthcare diagnostics market in Poland. In future, approximately2.5 millionPoles are expected to be diagnosed with diabetes, reflecting a 10% increase from 2020. This growing patient population necessitates advanced diagnostic services, leading to increased demand for laboratory tests and diagnostic imaging, which are crucial for effective disease management and monitoring.
- Advancements in Diagnostic Technologies:The healthcare diagnostics sector in Poland is experiencing rapid technological advancements, particularly in molecular diagnostics and imaging technologies. In future, the market for advanced diagnostic equipment is projected to reachUSD 1.1 billion, driven by innovations such as next-generation sequencing and portable diagnostic devices. These technologies enhance accuracy and speed in disease detection, thereby improving patient outcomes and driving market growth as healthcare providers adopt these solutions.
- Rising Healthcare Expenditure:Poland's healthcare expenditure is projected to reachUSD 60 billionin future, reflecting a steady increase from the previous year. This rise in spending is primarily due to government initiatives aimed at improving healthcare infrastructure and access to diagnostic services. Increased funding allows for the procurement of advanced diagnostic tools and technologies, thereby enhancing the capabilities of laboratories and diagnostic centers to meet the growing healthcare demands of the population.
Market Challenges
- High Operational Costs:The operational costs associated with running diagnostic laboratories in Poland are a significant challenge. In future, the average operational cost for a mid-sized laboratory is estimated to be aroundUSD 1.5 millionannually. These costs include equipment maintenance, staffing, and compliance with regulatory standards. High operational expenses can limit the ability of smaller labs to compete effectively, potentially leading to reduced service availability in certain regions.
- Stringent Regulatory Requirements:The healthcare diagnostics market in Poland faces stringent regulatory requirements that can hinder market entry and operational efficiency. Compliance with EU medical device regulations requires significant investment in quality assurance and documentation processes. In future, it is estimated that compliance costs for new diagnostic products could reachUSD 400,000, creating barriers for smaller companies and stifling innovation within the sector.
Poland Healthcare Diagnostics and Labs Market Future Outlook
The future of the healthcare diagnostics market in Poland appears promising, driven by technological advancements and increasing healthcare investments. The integration of artificial intelligence in diagnostics is expected to enhance accuracy and efficiency, while the expansion of telemedicine services will improve access to diagnostic testing. As the population ages and chronic diseases become more prevalent, the demand for innovative diagnostic solutions will continue to grow, fostering a dynamic and competitive market landscape.
Market Opportunities
- Expansion of Telemedicine Services:The growth of telemedicine presents a significant opportunity for the diagnostics market. In future, telemedicine consultations in Poland are expected to exceed5 millionannually, facilitating remote diagnostics and increasing patient access to healthcare services. This trend can lead to higher demand for at-home diagnostic tests, enhancing convenience and efficiency in patient care.
- Integration of AI in Diagnostics:The integration of artificial intelligence in diagnostic processes is poised to revolutionize the market. In future, AI-driven diagnostic tools are projected to account forUSD 300 millionin revenue, improving diagnostic accuracy and reducing turnaround times. This technological advancement will not only enhance patient outcomes but also streamline laboratory operations, creating a competitive edge for early adopters in the market.
Scope of the Report
| Segment | Sub-Segments |
|---|---|
| By Type | Clinical Chemistry Microbiology Hematology Immunology Molecular Diagnostics Pathology Imaging Diagnostics Laboratory Services Consumables Software Solutions Maintenance Services Others |
| By End-User | Hospitals Diagnostic Laboratories Private Clinics Research and Academic Institutions Outpatient Care Centers Home Care Settings Others |
| By Application | Disease Diagnosis Health Monitoring Drug Development Clinical Trials |
| By Distribution Channel | Direct Sales Online Sales Distributors |
| By Pricing Model | Premium Pricing Competitive Pricing Value-Based Pricing |
| By Service Type | Routine Testing Specialized Testing Emergency Testing |
| By Technology | Automated Systems Manual Systems Hybrid Systems |
Key Target Audience
Investors and Venture Capitalist Firms
Government and Regulatory Bodies (e.g., Ministry of Health, National Health Fund)
Manufacturers and Producers of Diagnostic Equipment
Clinical Laboratories and Diagnostic Centers
Healthcare Providers and Hospitals
Pharmaceutical Companies
Health Insurance Companies
Biotechnology Firms
Players Mentioned in the Report:
Diagnostyka S.A.
Synevo Poland
Grupa Alab
Voxel S.A.
PZU Zdrowie
Roche Diagnostics Polska
Siemens Healthineers Poland
Abbott Laboratories Poland
Philips Healthcare Poland
GE Healthcare Poland
Canon Medical Systems Poland
BioMerieux Poland
Mindray Medical Poland
Becton, Dickinson and Company Poland
Thermo Fisher Scientific Poland
Table of Contents
Market Assessment Phase
1. Executive Summary and Approach
2. Poland Healthcare Diagnostics and Labs Market Overview
2.1 Key Insights and Strategic Recommendations
2.2 Poland Healthcare Diagnostics and Labs Market Overview
2.3 Definition and Scope
2.4 Evolution of Market Ecosystem
2.5 Timeline of Key Regulatory Milestones
2.6 Value Chain & Stakeholder Mapping
2.7 Business Cycle Analysis
2.8 Policy & Incentive Landscape
3. Poland Healthcare Diagnostics and Labs Market Analysis
3.1 Growth Drivers
3.1.1 Increasing prevalence of chronic diseases
3.1.2 Advancements in diagnostic technologies
3.1.3 Rising healthcare expenditure
3.1.4 Growing demand for preventive healthcare
3.2 Market Challenges
3.2.1 High operational costs
3.2.2 Stringent regulatory requirements
3.2.3 Limited access in rural areas
3.2.4 Competition from international players
3.3 Market Opportunities
3.3.1 Expansion of telemedicine services
3.3.2 Integration of AI in diagnostics
3.3.3 Collaborations with research institutions
3.3.4 Growth in personalized medicine
3.4 Market Trends
3.4.1 Shift towards home-based diagnostics
3.4.2 Increasing focus on patient-centric services
3.4.3 Rise of point-of-care testing
3.4.4 Adoption of digital health solutions
3.5 Government Regulation
3.5.1 Compliance with EU medical device regulations
3.5.2 National health policies promoting diagnostics
3.5.3 Quality assurance standards for laboratories
3.5.4 Data protection regulations in healthcare
4. SWOT Analysis
5. Stakeholder Analysis
6. Porter's Five Forces Analysis
7. Poland Healthcare Diagnostics and Labs Market Market Size, 2019-2024
7.1 By Value
7.2 By Volume
7.3 By Average Selling Price
8. Poland Healthcare Diagnostics and Labs Market Segmentation
8.1 By Type
8.1.1 Clinical Chemistry
8.1.2 Microbiology
8.1.3 Hematology
8.1.4 Immunology
8.1.5 Molecular Diagnostics
8.1.6 Pathology
8.1.7 Imaging Diagnostics
8.1.8 Laboratory Services
8.1.9 Consumables
8.1.10 Software Solutions
8.1.11 Maintenance Services
8.1.12 Others
8.2 By End-User
8.2.1 Hospitals
8.2.2 Diagnostic Laboratories
8.2.3 Private Clinics
8.2.4 Research and Academic Institutions
8.2.5 Outpatient Care Centers
8.2.6 Home Care Settings
8.2.7 Others
8.3 By Application
8.3.1 Disease Diagnosis
8.3.2 Health Monitoring
8.3.3 Drug Development
8.3.4 Clinical Trials
8.4 By Distribution Channel
8.4.1 Direct Sales
8.4.2 Online Sales
8.4.3 Distributors
8.5 By Pricing Model
8.5.1 Premium Pricing
8.5.2 Competitive Pricing
8.5.3 Value-Based Pricing
8.6 By Service Type
8.6.1 Routine Testing
8.6.2 Specialized Testing
8.6.3 Emergency Testing
8.7 By Technology
8.7.1 Automated Systems
8.7.2 Manual Systems
8.7.3 Hybrid Systems
9. Poland Healthcare Diagnostics and Labs Market Competitive Analysis
9.1 Market Share of Key Players
9.2 Cross Comparison of Key Players
9.2.1 Company Name
9.2.2 Group Size (Large, Medium, or Small as per industry convention)
9.2.3 Revenue Growth Rate (YoY %)
9.2.4 Market Penetration Rate (% of total diagnostic tests or labs served)
9.2.5 Customer Retention Rate (%)
9.2.6 Average Price per Test (PLN)
9.2.7 Product/Service Portfolio Breadth
9.2.8 Operational Efficiency Ratio (EBITDA margin, FCF margin)
9.2.9 Innovation Index (R&D spend as % of revenue, patents filed)
9.2.10 Geographic Coverage (number of locations, regions served)
9.2.11 Brand Recognition Score (survey-based or market share proxy)
9.3 SWOT Analysis of Top Players
9.4 Pricing Analysis
9.5 Detailed Profile of Major Companies
9.5.1 Diagnostyka S.A.
9.5.2 Synevo Poland
9.5.3 Grupa Alab
9.5.4 Voxel S.A.
9.5.5 PZU Zdrowie
9.5.6 Roche Diagnostics Polska
9.5.7 Siemens Healthineers Poland
9.5.8 Abbott Laboratories Poland
9.5.9 Philips Healthcare Poland
9.5.10 GE Healthcare Poland
9.5.11 Canon Medical Systems Poland
9.5.12 BioMérieux Poland
9.5.13 Mindray Medical Poland
9.5.14 Becton, Dickinson and Company Poland
9.5.15 Thermo Fisher Scientific Poland
10. Poland Healthcare Diagnostics and Labs Market End-User Analysis
10.1 Procurement Behavior of Key Ministries
10.1.1 Ministry of Health
10.1.2 Ministry of Finance
10.1.3 Ministry of Education
10.2 Corporate Spend on Infrastructure & Energy
10.2.1 Investment in Diagnostic Facilities
10.2.2 Funding for Research and Development
10.2.3 Budget Allocation for Health Programs
10.3 Pain Point Analysis by End-User Category
10.3.1 Hospitals
10.3.2 Diagnostic Labs
10.3.3 Research Institutions
10.4 User Readiness for Adoption
10.4.1 Training and Support Needs
10.4.2 Technology Acceptance Levels
10.4.3 Infrastructure Readiness
10.5 Post-Deployment ROI and Use Case Expansion
10.5.1 Measurement of ROI
10.5.2 Expansion into New Use Cases
10.5.3 Long-term Sustainability Considerations
11. Poland Healthcare Diagnostics and Labs Market Future Size, 2025-2030
11.1 By Value
11.2 By Volume
11.3 By Average Selling Price
Go-To-Market Strategy Phase
1. Whitespace Analysis + Business Model Canvas
1.1 Market Gaps Identification
1.2 Business Model Framework
2. Marketing and Positioning Recommendations
2.1 Branding Strategies
2.2 Product USPs
3. Distribution Plan
3.1 Urban Retail Strategies
3.2 Rural NGO Tie-ups
4. Channel & Pricing Gaps
4.1 Underserved Routes
4.2 Pricing Bands Analysis
5. Unmet Demand & Latent Needs
5.1 Category Gaps
5.2 Consumer Segments
6. Customer Relationship
6.1 Loyalty Programs
6.2 After-sales Service
7. Value Proposition
7.1 Sustainability Initiatives
7.2 Integrated Supply Chains
8. Key Activities
8.1 Regulatory Compliance
8.2 Branding Efforts
8.3 Distribution Setup
9. Entry Strategy Evaluation
9.1 Domestic Market Entry Strategy
9.1.1 Product Mix Considerations
9.1.2 Pricing Band Strategy
9.1.3 Packaging Solutions
9.2 Export Entry Strategy
9.2.1 Target Countries Identification
9.2.2 Compliance Roadmap Development
10. Entry Mode Assessment
10.1 Joint Ventures
10.2 Greenfield Investments
10.3 Mergers & Acquisitions
10.4 Distributor Model Evaluation
11. Capital and Timeline Estimation
11.1 Capital Requirements
11.2 Timelines for Implementation
12. Control vs Risk Trade-Off
12.1 Ownership Considerations
12.2 Partnerships Evaluation
13. Profitability Outlook
13.1 Breakeven Analysis
13.2 Long-term Sustainability
14. Potential Partner List
14.1 Distributors
14.2 Joint Ventures
14.3 Acquisition Targets
15. Execution Roadmap
15.1 Phased Plan for Market Entry
15.1.1 Market Setup
15.1.2 Market Entry
15.1.3 Growth Acceleration
15.1.4 Scale & Stabilize
15.2 Key Activities and Milestones
15.2.1 Milestone Planning
15.2.2 Activity Tracking
Research Methodology
Phase 1: Approach1
Desk Research
- Analysis of healthcare expenditure reports from the Polish Ministry of Health
- Review of industry publications and market reports from healthcare associations
- Examination of demographic and epidemiological data from national health surveys
Primary Research
- Interviews with laboratory directors and healthcare administrators in Poland
- Telephonic surveys with key opinion leaders in the diagnostics field
- Focus group discussions with healthcare professionals and lab technicians
Validation & Triangulation
- Cross-validation of findings with data from international health organizations
- Triangulation of insights from primary interviews and secondary data sources
- Sanity checks through expert panel reviews comprising industry veterans
Phase 2: Market Size Estimation1
Top-down Assessment
- Estimation of total healthcare spending in Poland as a baseline for diagnostics
- Segmentation of the market by diagnostic categories and laboratory types
- Incorporation of trends in preventive healthcare and chronic disease management
Bottom-up Modeling
- Collection of revenue data from leading diagnostic labs and healthcare providers
- Operational cost analysis based on service pricing and volume of tests conducted
- Estimation of market size based on test frequency and patient demographics
Forecasting & Scenario Analysis
- Multi-factor regression analysis incorporating technological advancements and policy changes
- Scenario modeling based on potential shifts in healthcare funding and patient behavior
- Development of baseline, optimistic, and pessimistic forecasts through 2030
Phase 3: CATI Sample Composition1
| Scope Item/Segment | Sample Size | Target Respondent Profiles |
|---|---|---|
| Clinical Laboratory Services | 60 | Laboratory Managers, Pathologists |
| Diagnostic Imaging Centers | 45 | Radiologists, Imaging Technicians |
| Point-of-Care Testing | 40 | Healthcare Practitioners, Clinic Managers |
| Genetic Testing Services | 40 | Genetic Counselors, Laboratory Directors |
| Microbiology and Infectious Disease Labs | 50 | Microbiologists, Infection Control Specialists |
Frequently Asked Questions
What is the current value of the Poland Healthcare Diagnostics and Labs Market?
The Poland Healthcare Diagnostics and Labs Market is valued at approximately USD 5.5 billion, driven by factors such as the increasing prevalence of chronic diseases, advancements in diagnostic technologies, and a growing emphasis on preventive healthcare.