Region:Middle East
Author(s):Rebecca
Product Code:KRAD4912
Pages:100
Published On:December 2025
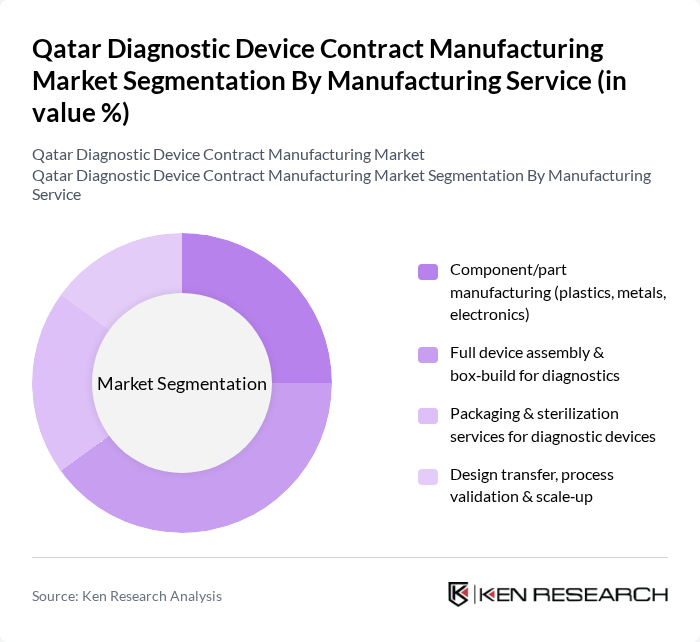
By Manufacturing Service:The manufacturing services segment encompasses various sub-segments, including component/part manufacturing, full device assembly, packaging and sterilization services, and design transfer. Among these, full device assembly is currently the leading sub-segment, driven by the increasing complexity of diagnostic devices and the demand for integrated solutions. Manufacturers are focusing on providing comprehensive services that cover the entire lifecycle of diagnostic devices, from design to final assembly, which enhances efficiency and reduces time-to-market.
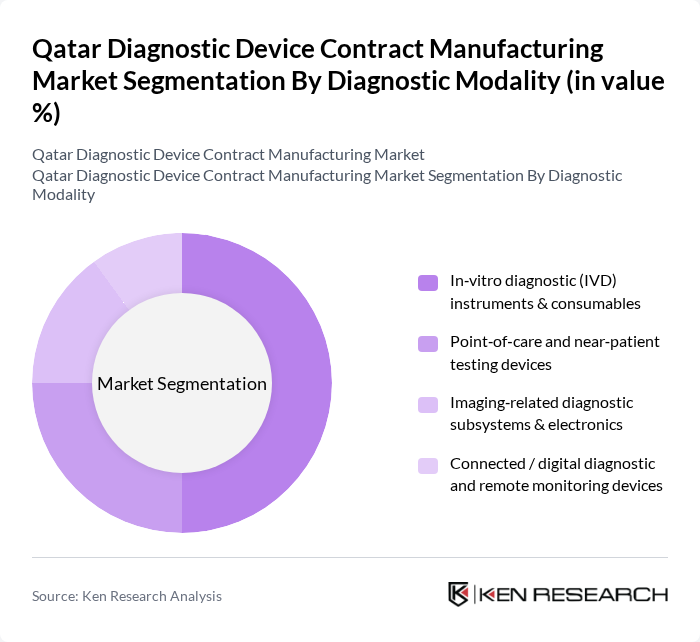
By Diagnostic Modality:The diagnostic modality segment includes in-vitro diagnostic instruments, point-of-care testing devices, imaging-related diagnostic subsystems, and connected digital diagnostic devices. The in-vitro diagnostic (IVD) instruments and consumables sub-segment is the most significant, driven by the growing demand for rapid and accurate testing solutions. The rise in chronic diseases and the need for early detection have led to increased investments in IVD technologies, making it a focal point for manufacturers in the region. The Middle East in-vitro diagnostics market was valued at USD 2,093 million in 2024, with Qatar representing a key manufacturing hub for diagnostic device contract manufacturing services.
The Qatar Diagnostic Device Contract Manufacturing Market is characterized by a dynamic mix of regional and international players. Leading participants such as Sanmina Corporation, Jabil Inc., Flex Ltd. (Flex Health Solutions), Celestica Inc., Integer Holdings Corporation, Viant Medical, Inc., Gerresheimer AG, Phillips?Medisize (a Molex company), Eurofins CDMO (diagnostic device services), Forefront Medical Technology, Qatar Pharma for Pharmaceutical Industries, Sidra Medicine Innovation & Manufacturing Center, Hamad Medical Corporation – Medical Devices & Technology Management Department, Aspetar – Orthopaedic and Sports Medicine Hospital (diagnostic technology collaborations), SeaskyMedical contribute to innovation, geographic expansion, and service delivery in this space.
The future of the Qatar diagnostic device contract manufacturing market appears promising, driven by technological advancements and increasing healthcare investments. The integration of artificial intelligence in diagnostic devices is expected to enhance accuracy and efficiency, while the expansion of telemedicine will facilitate remote diagnostics. As the government continues to support local manufacturing initiatives, contract manufacturers are likely to benefit from favorable policies and increased collaboration with international firms, fostering innovation and growth in the sector.
| Segment | Sub-Segments |
|---|---|
| By Manufacturing Service | Component/part manufacturing (plastics, metals, electronics) Full device assembly & box?build for diagnostics Packaging & sterilization services for diagnostic devices Design transfer, process validation & scale?up |
| By Diagnostic Modality | In?vitro diagnostic (IVD) instruments & consumables Point?of?care and near?patient testing devices Imaging?related diagnostic subsystems & electronics Connected / digital diagnostic and remote monitoring devices |
| By Contract Type | Contract manufacturing only (build?to?print) Contract development and manufacturing (CDMO) Long?term strategic manufacturing partnerships Short?term / project?based contracts |
| By Client Type | Global IVD and diagnostic OEMs Regional/MENA diagnostic manufacturers Local Qatari healthcare & diagnostic startups Academic and research spin?offs |
| By Application Area | Infectious disease and respiratory diagnostics Oncology and genetic testing Cardio?metabolic and chronic disease diagnostics Women’s health, fertility and prenatal diagnostics |
| By Geography of Client Base | Domestic (Qatar?based clients) GCC and wider Middle East Europe Asia?Pacific & North America |
| By Ownership & Investment | State?linked and government?backed entities Private domestic investors Foreign direct investment (FDI) backed facilities Joint ventures and PPP models |
| Scope Item/Segment | Sample Size | Target Respondent Profiles |
|---|---|---|
| Hospital Procurement Departments | 90 | Procurement Managers, Supply Chain Coordinators |
| Diagnostic Device Manufacturers | 60 | Product Managers, R&D Directors |
| Healthcare Professionals | 110 | Doctors, Lab Technicians |
| Regulatory Bodies | 40 | Compliance Officers, Policy Makers |
| Healthcare Distributors | 70 | Sales Managers, Distribution Coordinators |
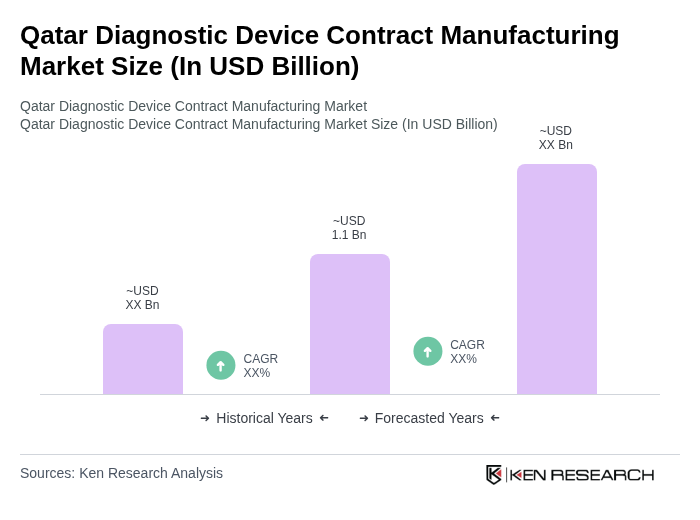
The Qatar Diagnostic Device Contract Manufacturing Market is valued at approximately USD 1.1 billion, reflecting a significant growth driven by the demand for advanced diagnostic technologies and the increasing prevalence of chronic diseases.