Region:Middle East
Author(s):Dev
Product Code:KRAC4807
Pages:97
Published On:October 2025
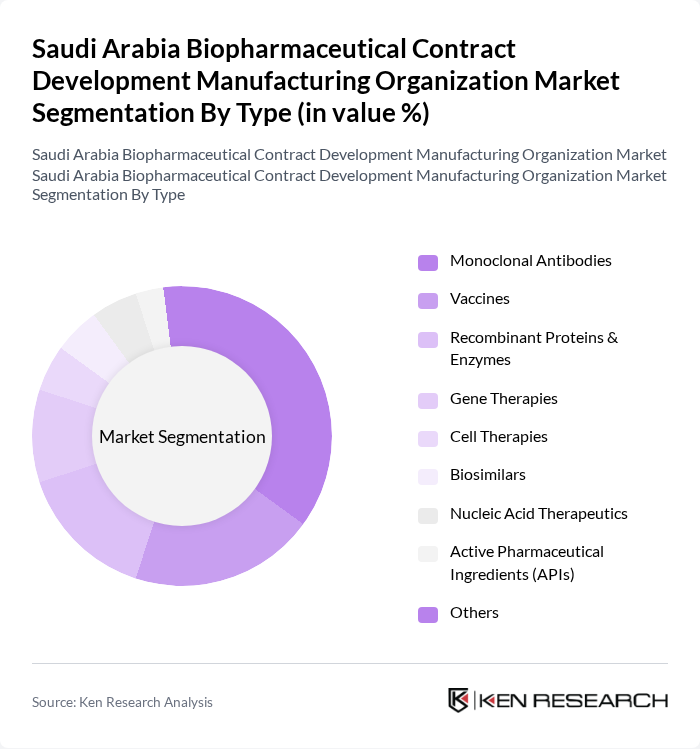
By Type:The biopharmaceutical contract development manufacturing organization market can be segmented into various types, including Monoclonal Antibodies, Vaccines, Recombinant Proteins & Enzymes, Gene Therapies, Cell Therapies, Biosimilars, Nucleic Acid Therapeutics, Active Pharmaceutical Ingredients (APIs), and Others. Among these, Monoclonal Antibodies have emerged as a leading sub-segment due to their widespread application in treating cancer, autoimmune disorders, and infectious diseases. The increasing focus on personalized medicine, the growing number of approvals for monoclonal antibody therapies, and the expansion of local manufacturing capabilities have significantly contributed to their dominance in the market .
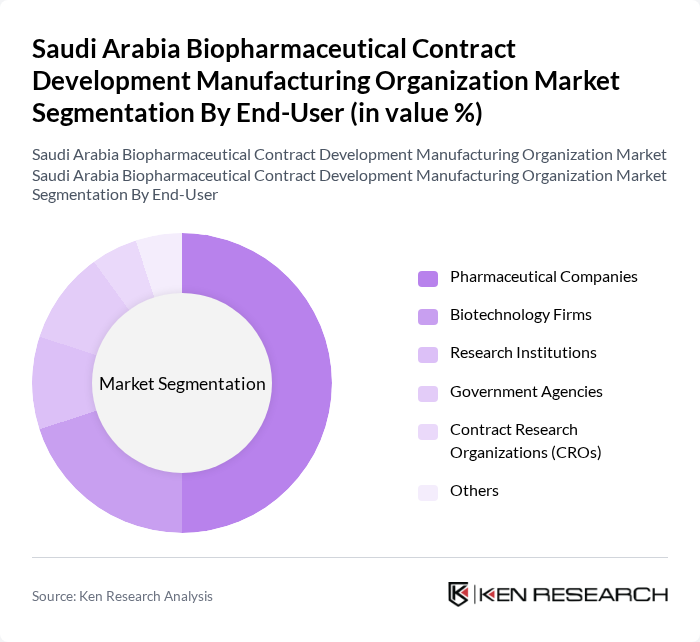
By End-User:The end-user segmentation includes Pharmaceutical Companies, Biotechnology Firms, Research Institutions, Government Agencies, Contract Research Organizations (CROs), and Others. Pharmaceutical Companies are the dominant end-user segment, driven by their need for efficient manufacturing solutions to meet the growing demand for biopharmaceutical products. The increasing trend of outsourcing manufacturing processes to specialized CDMOs allows pharmaceutical companies to focus on their core competencies while ensuring high-quality production. Biotechnology firms and research institutions are also expanding their engagement with CDMOs as the sector shifts towards advanced therapies and biologics .
The Saudi Arabia Biopharmaceutical Contract Development Manufacturing Organization Market is characterized by a dynamic mix of regional and international players. Leading participants such as Lifera, Saudi Bioventure, Tabuk Pharmaceuticals Manufacturing Company, Riyadh Pharma (Saudi Pharmaceutical Industries & Medical Appliances Corporation), Balsam Clinical Research, Sudair Pharma Company, Jamjoom Pharma, SPIMACO (Saudi Pharmaceutical Industries & Medical Appliances Corporation), Gulf Pharmaceutical Industries (Julphar), Dar Al Dawa Development and Investment Company, Aljomaih and Shell Lubricating Oil Company (for bioprocessing partnerships), Al-Jeel Medical & Trading Co., Al Nahdi Medical Company, Badr Pharma, United Pharmacies contribute to innovation, geographic expansion, and service delivery in this space.
The future of the biopharmaceutical contract development manufacturing organization market in Saudi Arabia appears promising, driven by government initiatives and technological advancements. The focus on local manufacturing capabilities is expected to enhance self-sufficiency in biopharmaceutical production. Additionally, the increasing prevalence of chronic diseases will likely spur demand for innovative therapies, creating a conducive environment for growth. As the market evolves, collaboration with international firms will play a crucial role in knowledge transfer and capacity building, further strengthening the sector.
| Segment | Sub-Segments |
|---|---|
| By Type | Monoclonal Antibodies Vaccines Recombinant Proteins & Enzymes Gene Therapies Cell Therapies Biosimilars Nucleic Acid Therapeutics Active Pharmaceutical Ingredients (APIs) Others |
| By End-User | Pharmaceutical Companies Biotechnology Firms Research Institutions Government Agencies Contract Research Organizations (CROs) Others |
| By Application | Therapeutics Diagnostics Research & Development Clinical Trials Others |
| By Distribution Channel | Direct Sales Distributors Online Platforms Others |
| By Region | Central Region (Riyadh) Eastern Region (Dammam, Khobar) Western Region (Jeddah, Makkah) Southern Region Others |
| By Pricing Strategy | Premium Pricing Competitive Pricing Value-Based Pricing Others |
| By Regulatory Compliance | Local Regulations (SFDA) International Standards (GMP, EMA, FDA) Quality Assurance Protocols Others |
| Scope Item/Segment | Sample Size | Target Respondent Profiles |
|---|---|---|
| Biopharmaceutical CDMO Operations | 60 | Operations Managers, Production Directors |
| Regulatory Compliance in Biopharmaceuticals | 50 | Regulatory Affairs Specialists, Quality Assurance Managers |
| Market Trends in Biopharmaceuticals | 45 | Market Analysts, Business Development Managers |
| R&D Innovations in Biopharmaceuticals | 40 | R&D Directors, Scientific Advisors |
| Investment Opportunities in Biopharmaceuticals | 50 | Investment Analysts, Venture Capitalists |
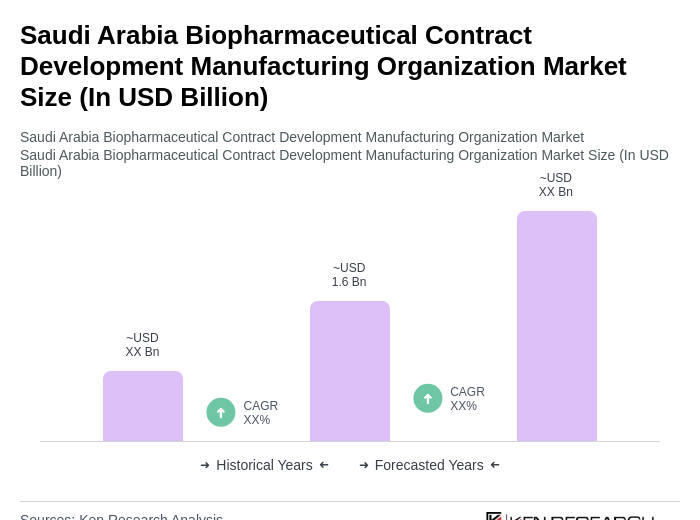
The Saudi Arabia Biopharmaceutical Contract Development Manufacturing Organization Market is valued at approximately USD 1.6 billion, driven by increased healthcare investments, rising chronic disease prevalence, and demand for innovative biopharmaceuticals.