Region:Middle East
Author(s):Rebecca
Product Code:KRAC3201
Pages:83
Published On:October 2025
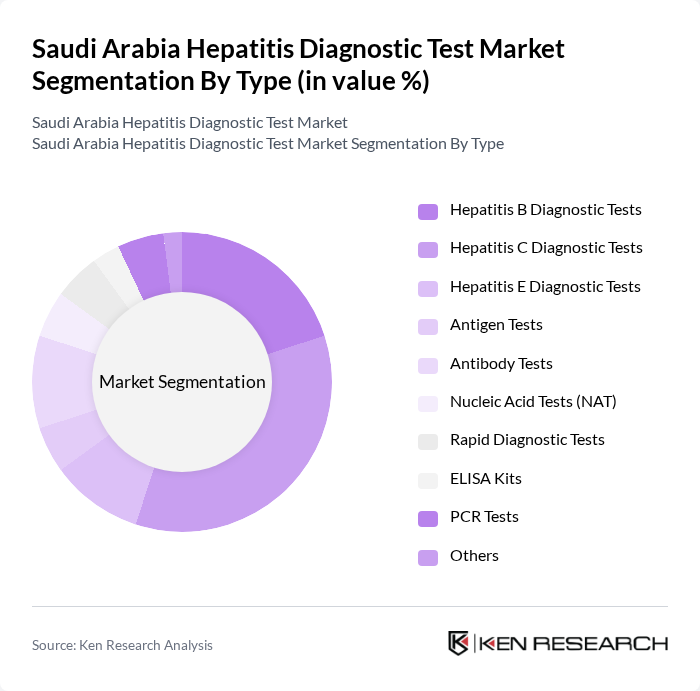
By Type:The market is segmented into hepatitis B, C, and E diagnostic tests, as well as antigen tests, antibody tests, nucleic acid tests (NAT), rapid diagnostic tests, ELISA kits, PCR tests, and others. Among these, hepatitis C diagnostic tests account for the largest share, reflecting the high prevalence of hepatitis C in Saudi Arabia and the growing demand for sensitive molecular and serological screening solutions. Hepatitis B diagnostics also represent a significant segment, supported by national immunization and screening programs .
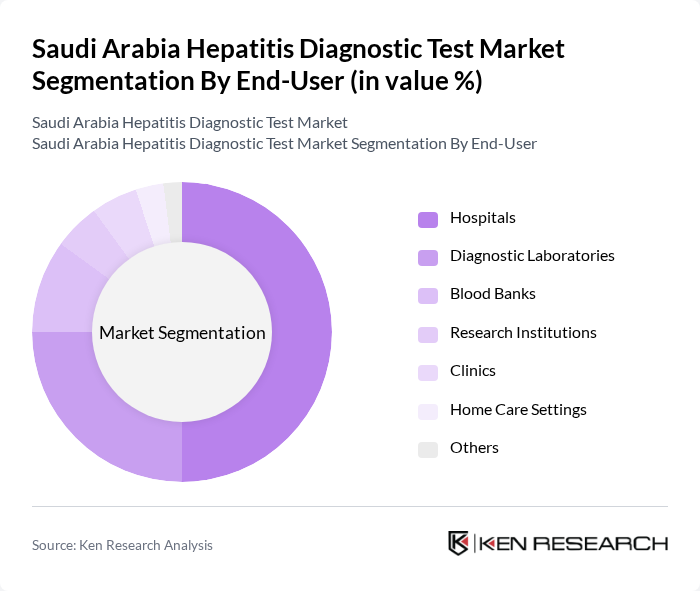
By End-User:The end-user segmentation includes hospitals, diagnostic laboratories, blood banks, research institutions, clinics, home care settings, and others. Hospitals remain the leading end-users, driven by their comprehensive service offerings, high patient throughput, and integration with national screening and vaccination initiatives. Diagnostic laboratories and blood banks also constitute significant market shares due to their role in routine screening, blood safety, and disease surveillance .
The Saudi Arabia Hepatitis Diagnostic Test Market is characterized by a dynamic mix of regional and international players. Leading participants such as Abbott Laboratories, Roche Diagnostics, Siemens Healthineers, Bio-Rad Laboratories, Hologic, Inc., Cepheid, Grifols S.A., Ortho Clinical Diagnostics, Thermo Fisher Scientific, Becton, Dickinson and Company, Quidel Corporation, DiaSorin S.p.A., GenMark Diagnostics, Mylab Discovery Solutions, MedMira Inc., Al Borg Diagnostics, National Unified Procurement Company (NUPCO), Al-Dawaa Pharmacies, Al Faisaliah Medical Systems, Sysmex Corporation contribute to innovation, geographic expansion, and service delivery in this space.
The future of the Saudi Arabia hepatitis diagnostic test market appears promising, driven by ongoing government initiatives and technological advancements. The integration of telemedicine is expected to enhance access to diagnostic services, particularly in underserved areas. Additionally, the growing emphasis on personalized medicine will likely lead to the development of tailored diagnostic solutions, improving patient outcomes. As healthcare infrastructure continues to expand, the market is poised for significant growth, addressing the rising demand for effective hepatitis management and prevention strategies.
| Segment | Sub-Segments |
|---|---|
| By Type | Hepatitis B Diagnostic Tests Hepatitis C Diagnostic Tests Hepatitis E Diagnostic Tests Antigen Tests Antibody Tests Nucleic Acid Tests (NAT) Rapid Diagnostic Tests ELISA Kits PCR Tests Others |
| By End-User | Hospitals Diagnostic Laboratories Blood Banks Research Institutions Clinics Home Care Settings Others |
| By Distribution Channel | Direct Sales Distributors Online Sales Retail Pharmacies Hospitals Others |
| By Region | Central Region Eastern Region Western Region Southern Region Northern Region Others |
| By Test Purpose | Screening Tests Confirmatory Tests Follow-up Tests Others |
| By Sample Type | Blood Samples Serum Samples Plasma Samples Others |
| By Price Range | Low Price Range Mid Price Range High Price Range Others |
| Scope Item/Segment | Sample Size | Target Respondent Profiles |
|---|---|---|
| Hepatitis Diagnostic Test Manufacturers | 100 | Product Managers, Sales Directors |
| Healthcare Providers and Laboratories | 80 | Laboratory Technicians, Medical Directors |
| Government Health Officials | 50 | Policy Makers, Public Health Administrators |
| Patient Advocacy Groups | 40 | Advocacy Leaders, Community Health Workers |
| Healthcare Market Analysts | 40 | Market Researchers, Industry Analysts |
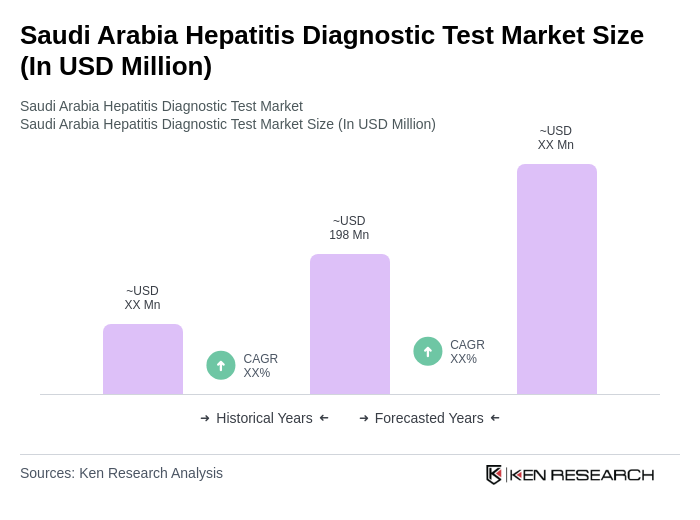
The Saudi Arabia Hepatitis Diagnostic Test Market is valued at approximately USD 198 million, reflecting a significant segment of the broader liver disease diagnostics market, driven by the rising prevalence of hepatitis infections and advancements in diagnostic technologies.