Region:Middle East
Author(s):Geetanshi
Product Code:KRAB1544
Pages:97
Published On:January 2026
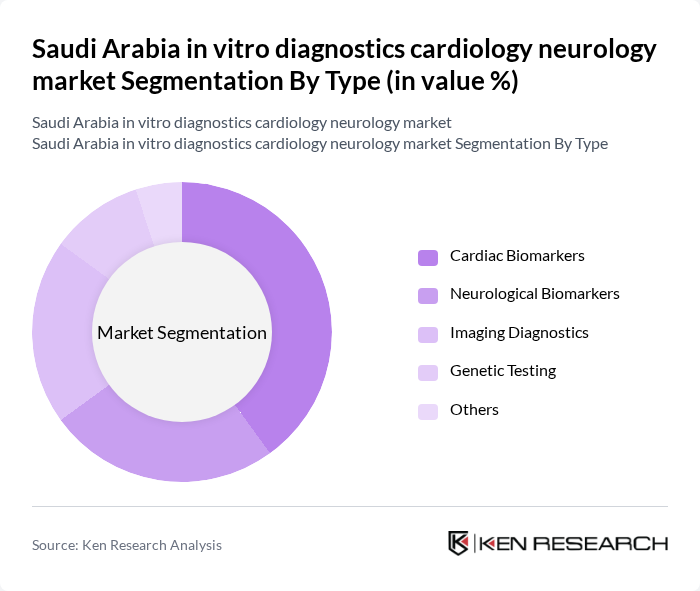
By Type:The market is segmented into various types, including cardiac biomarkers, neurological biomarkers, imaging diagnostics, genetic testing, and others. Among these, cardiac biomarkers are leading the market due to their critical role in diagnosing heart diseases, which are prevalent in the region. The increasing focus on preventive healthcare and the growing awareness of cardiovascular health are driving the demand for these diagnostics.
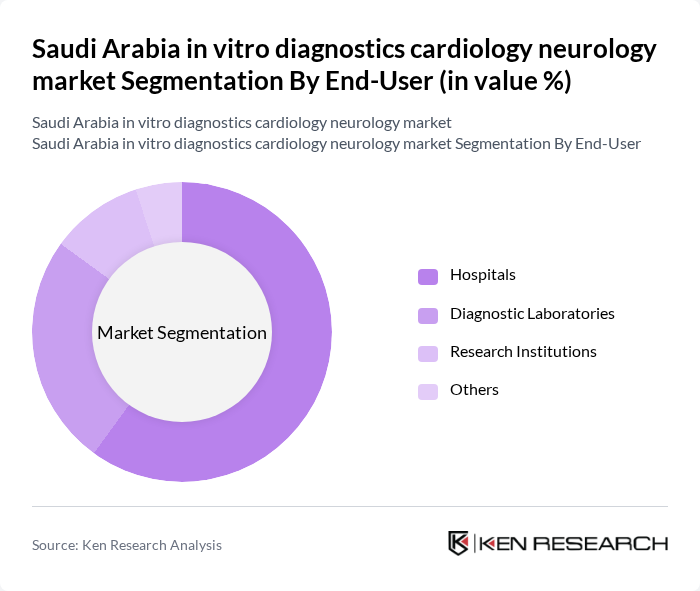
By End-User:The end-user segmentation includes hospitals, diagnostic laboratories, research institutions, home care settings, and others. Hospitals are the dominant end-user segment, driven by the increasing number of patients requiring diagnostic tests for cardiovascular and neurological conditions. The trend towards integrated healthcare services and the expansion of hospital networks are further enhancing the demand for in vitro diagnostics in this sector.
The Saudi Arabia in vitro diagnostics cardiology neurology market is characterized by a dynamic mix of regional and international players. Leading participants such as Siemens Healthineers, Roche Diagnostics, Abbott Laboratories, Bio-Rad Laboratories, Ortho Clinical Diagnostics, Thermo Fisher Scientific, Beckman Coulter, Sysmex Corporation, Mindray Medical International, Philips Healthcare, GE Healthcare, Becton Dickinson, Fujifilm Holdings Corporation, Canon Medical Systems, Agilent Technologies contribute to innovation, geographic expansion, and service delivery in this space.
The future of the in vitro diagnostics market in Saudi Arabia is poised for significant growth, driven by technological advancements and increasing healthcare investments. The integration of artificial intelligence and machine learning into diagnostic processes is expected to enhance accuracy and efficiency. Additionally, the rising focus on preventive healthcare will likely lead to increased demand for early diagnostic solutions, positioning the market for robust expansion in the future.
| Segment | Sub-Segments |
|---|---|
| By Type | Cardiac Biomarkers Neurological Biomarkers Imaging Diagnostics Genetic Testing Others |
| By End-User | Hospitals Diagnostic Laboratories Research Institutions Home Care Settings Others |
| By Application | Cardiovascular Diseases Neurological Disorders Preventive Health Screening Others |
| By Technology | Immunoassays Molecular Diagnostics Point-of-Care Testing Others |
| By Distribution Channel | Direct Sales Distributors Online Sales Others |
| By Region | Central Region Eastern Region Western Region Southern Region |
| By Customer Type | Public Sector Private Sector Others |
| Scope Item/Segment | Sample Size | Target Respondent Profiles |
|---|---|---|
| Cardiology Diagnostic Tools | 100 | Cardiologists, Hospital Administrators |
| Neurology Diagnostic Equipment | 80 | Neurologists, Lab Managers |
| Healthcare Procurement Practices | 70 | Procurement Officers, Supply Chain Managers |
| Market Trends in In Vitro Diagnostics | 90 | Healthcare Analysts, Medical Device Experts |
| Regulatory Impact on Diagnostics | 60 | Regulatory Affairs Specialists, Compliance Officers |
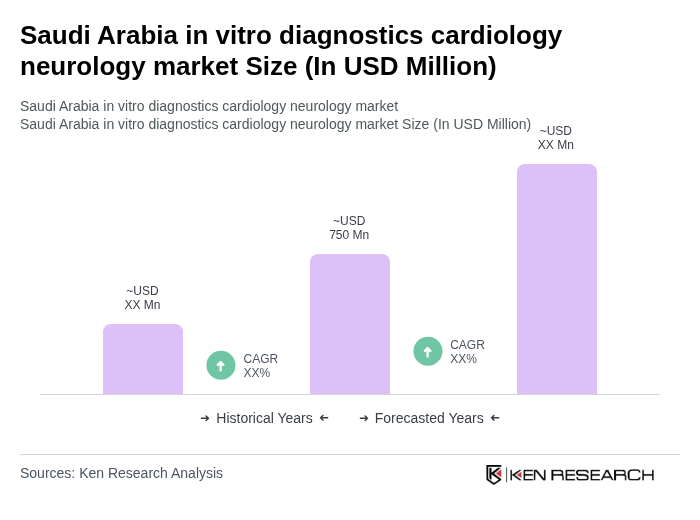
The Saudi Arabia in vitro diagnostics cardiology neurology market is valued at approximately USD 1.1 billion, reflecting significant growth driven by the rising prevalence of cardiovascular and neurological disorders and advancements in diagnostic technologies.