Region:Middle East
Author(s):Geetanshi
Product Code:KRAA5983
Pages:93
Published On:January 2026
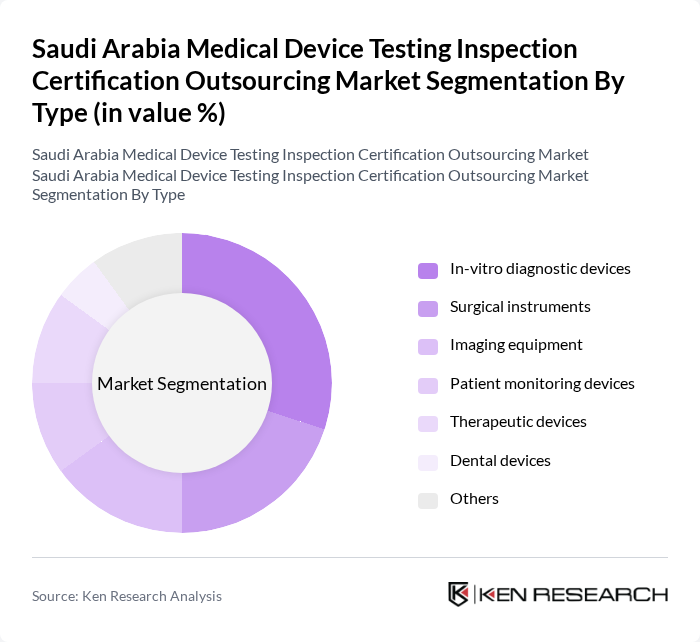
By Type:The market is segmented into various types of medical devices, including in-vitro diagnostic devices, surgical instruments, imaging equipment, patient monitoring devices, therapeutic devices, dental devices, and others. Among these, in-vitro diagnostic devices are leading due to their critical role in disease detection and management, especially in the context of rising chronic diseases and the need for rapid testing solutions.
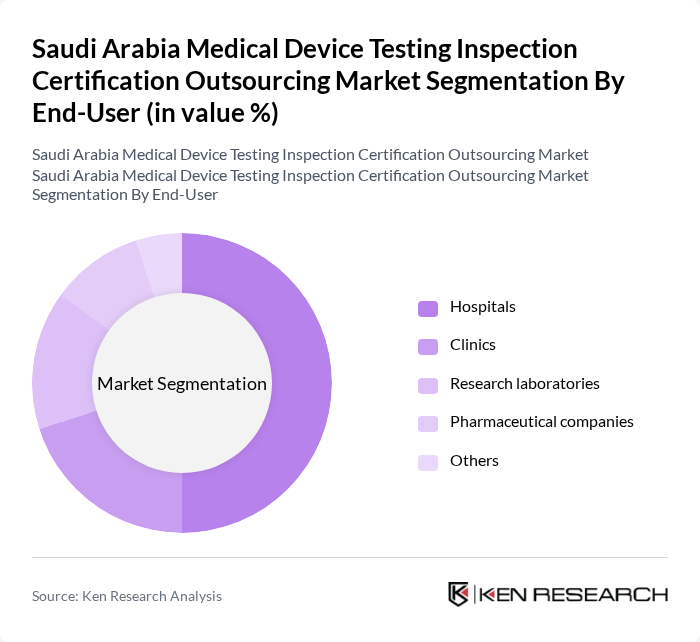
By End-User:The end-user segmentation includes hospitals, clinics, research laboratories, pharmaceutical companies, and others. Hospitals dominate this segment due to their extensive need for a variety of medical devices for patient care, diagnostics, and treatment, which drives the demand for testing and certification services.
The Saudi Arabia Medical Device Testing Inspection Certification Outsourcing Market is characterized by a dynamic mix of regional and international players. Leading participants such as SGS SA, TÜV SÜD, Intertek Group plc, Bureau Veritas, UL LLC, DNV GL, Eurofins Scientific, BSI Group, Medpace, Qserve Group, Emergo by UL, KPMG, Deloitte, PwC, RINA contribute to innovation, geographic expansion, and service delivery in this space.
The future of the Saudi Arabia medical device testing and certification outsourcing market appears promising, driven by ongoing healthcare reforms and regulatory advancements. The shift towards value-based healthcare models is expected to enhance the demand for rigorous testing and compliance systems, potentially leading to annual savings of SAR 40–65 billion by 2035. Additionally, the SFDA's status as a WHO-benchmarking authority positions Saudi Arabia as a regional leader, further increasing the need for outsourced inspection and certification services.
| Segment | Sub-Segments |
|---|---|
| By Type | In-vitro diagnostic devices Surgical instruments Imaging equipment Patient monitoring devices Therapeutic devices Dental devices Others |
| By End-User | Hospitals Clinics Research laboratories Pharmaceutical companies Others |
| By Application | Diagnostic applications Therapeutic applications Monitoring applications Surgical applications Others |
| By Certification Type | ISO Certification CE Marking FDA Approval Others |
| By Testing Method | Mechanical testing Electrical safety testing Biocompatibility testing Performance testing Others |
| By Region | Central Region Eastern Region Western Region Southern Region Others |
| By Market Maturity | Emerging market Established market Others |
| Scope Item/Segment | Sample Size | Target Respondent Profiles |
|---|---|---|
| Medical Device Manufacturers | 100 | Product Managers, Regulatory Affairs Specialists |
| Healthcare Providers | 80 | Clinical Directors, Procurement Managers |
| Testing and Certification Bodies | 60 | Quality Assurance Officers, Compliance Managers |
| Distributors and Wholesalers | 70 | Sales Managers, Supply Chain Coordinators |
| Regulatory Agencies | 50 | Policy Makers, Regulatory Compliance Experts |
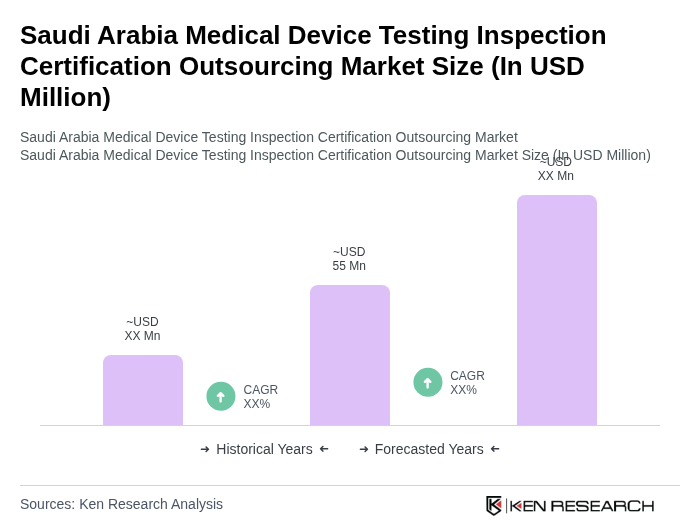
The Saudi Arabia Medical Device Testing Inspection Certification Outsourcing Market is valued at approximately USD 55 million, driven by investments in digital health, AI integration, and healthcare infrastructure improvements under Vision 2030.