Region:Asia
Author(s):Dev
Product Code:KRAB2354
Pages:96
Published On:January 2026
By Test Type:The market is segmented into various test types, including Cell-free DNA (cfDNA) in maternal plasma tests, biochemical screening tests, ultrasound-based screening, and others. cfDNA-based NIPT leverages next-generation sequencing and advanced bioinformatics to analyze fetal cell-free DNA in maternal blood, providing high sensitivity and specificity for common aneuploidies compared with conventional first-trimester biochemical and ultrasound screening. Among these, cfDNA tests are gaining significant traction due to their high accuracy, early testing window, and noninvasive nature, making them the preferred choice for many expectant mothers and clinicians, particularly in high-risk pregnancies. The increasing adoption of advanced sequencing platforms, partnerships between global NIPT providers and Thai laboratories, and ongoing price reductions in genomic testing are also contributing to the growth of this segment.

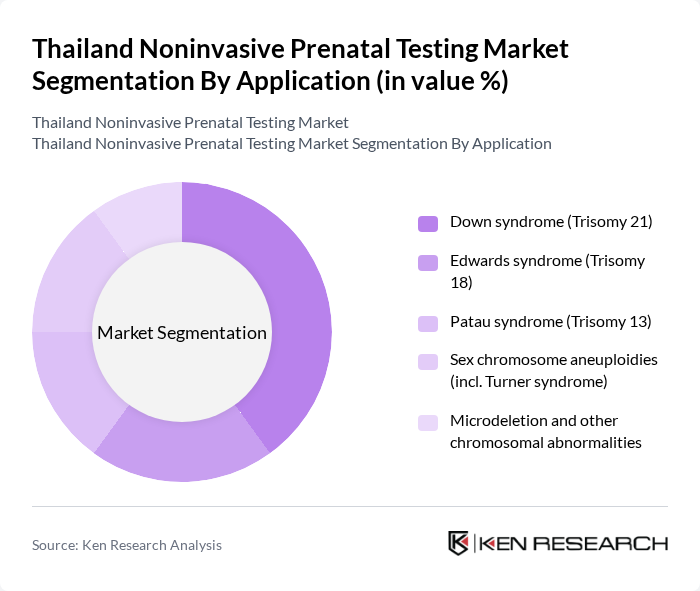
By Application:The applications of noninvasive prenatal testing include screening for Down syndrome (Trisomy 21), Edwards syndrome (Trisomy 18), Patau syndrome (Trisomy 13), sex chromosome aneuploidies (including Turner syndrome), and microdeletion and other chromosomal abnormalities. The Down syndrome screening is the most prevalent application, driven by heightened awareness of trisomy 21 risk, the relatively higher incidence compared with other aneuploidies, and the importance of early detection to support informed decision-making and perinatal care planning.
The Thailand Noninvasive Prenatal Testing Market is characterized by a dynamic mix of regional and international players. Leading participants such as Bangkok Hospital Group, Bumrungrad International Hospital, King Chulalongkorn Memorial Hospital, Samitivej Hospital, Phyathai Hospital, Vejthani Hospital, Bangkok Hospital Medical Center, Thonburi Hospital, Ramkhamhaeng Hospital, Siriraj Hospital, BNH Hospital, Piyavate Hospital, MedPark Hospital, Bangkok Nursing Home Hospital, Theptarin Hospital contribute to innovation, geographic expansion, and service delivery in this space.
The future of the NIPT market in Thailand appears promising, driven by ongoing advancements in technology and increasing public health initiatives. As healthcare infrastructure continues to expand, particularly in underserved regions, access to NIPT is expected to improve significantly. Additionally, the integration of telemedicine and digital health solutions will facilitate remote consultations, further enhancing the reach of prenatal testing services. These trends indicate a robust growth trajectory for the NIPT market, aligning with the country's commitment to improving maternal health outcomes.
| Segment | Sub-Segments |
|---|---|
| By Test Type | Cell-free DNA (cfDNA) in maternal plasma tests Biochemical screening tests Ultrasound-based screening Others |
| By Application | Down syndrome (Trisomy 21) Edwards syndrome (Trisomy 18) Patau syndrome (Trisomy 13) Sex chromosome aneuploidies (incl. Turner syndrome) Microdeletion and other chromosomal abnormalities |
| By Gestational Age | –12 weeks –24 weeks Above 24 weeks |
| By Sample Type | Maternal plasma cfDNA Other blood-based samples |
| By End-User | Hospitals and birthing centers Diagnostic laboratories IVF and fertility clinics Others |
| By Component | Kits and reagents Instruments and platforms Services |
| By Distribution Channel | Physician-referral Direct-to-consumer (DTC) Others |
| Scope Item/Segment | Sample Size | Target Respondent Profiles |
|---|---|---|
| Obstetricians and Gynecologists | 60 | Healthcare Providers, Specialists in Prenatal Care |
| Expectant Parents | 120 | Parents-to-be, Couples Considering NIPT |
| Genetic Counselors | 50 | Healthcare Professionals, Genetic Testing Experts |
| Healthcare Administrators | 40 | Hospital Managers, Clinic Directors |
| Insurance Providers | 40 | Policy Makers, Health Insurance Analysts |
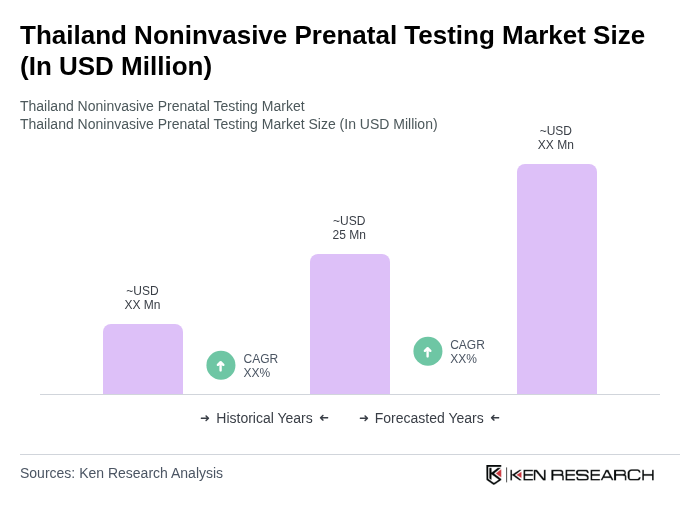
The Thailand Noninvasive Prenatal Testing Market is valued at approximately USD 25 million, reflecting a growing trend towards noninvasive procedures among expectant mothers, driven by increased awareness of prenatal health and advancements in genetic testing technologies.