Region:Europe
Author(s):Shubham
Product Code:KRAB3279
Pages:95
Published On:October 2025
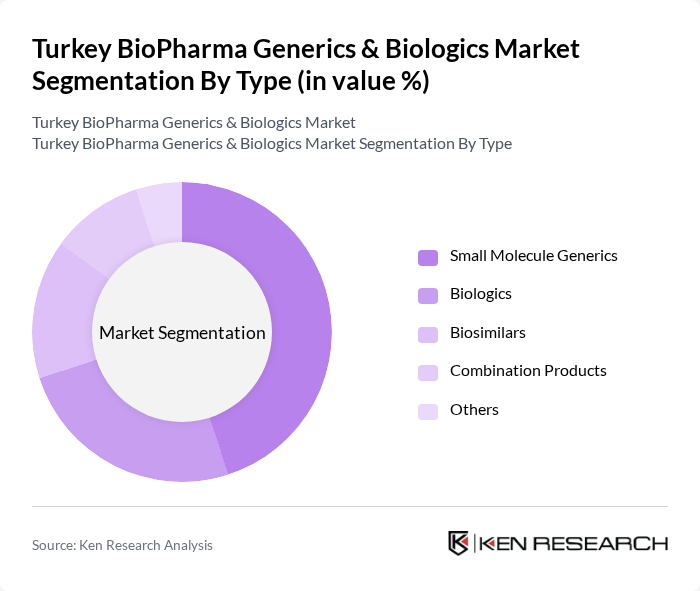
By Type:The market is segmented into various types, including Small Molecule Generics, Biologics, Biosimilars, Combination Products, and Others. Among these, Small Molecule Generics dominate the market due to their widespread use in treating common ailments and their cost-effectiveness. Biologics and Biosimilars are also gaining traction, driven by the increasing demand for innovative therapies and the need for affordable alternatives to expensive biologic drugs.
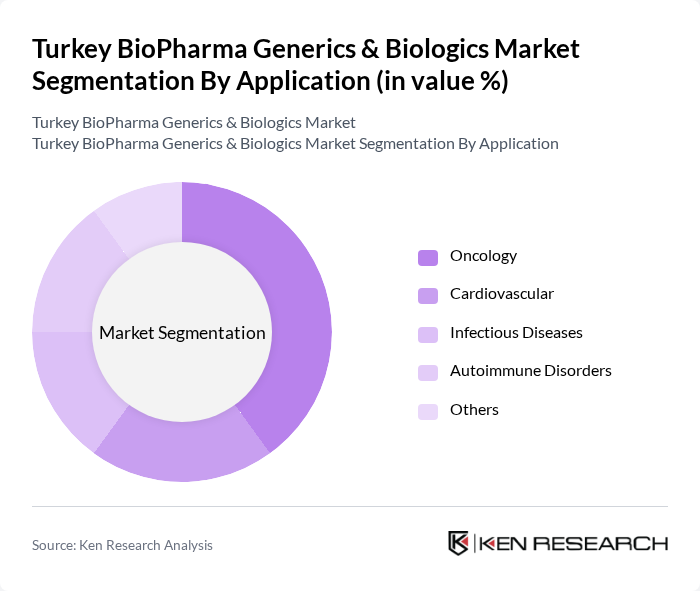
By Application:The applications of bio-pharmaceuticals include Oncology, Cardiovascular, Infectious Diseases, Autoimmune Disorders, and Others. Oncology is the leading application area, driven by the rising incidence of cancer and the demand for effective treatment options. The increasing focus on personalized medicine and targeted therapies further propels the growth of this segment, while other applications are also expanding due to advancements in treatment methodologies.
The Turkey BioPharma Generics & Biologics Market is characterized by a dynamic mix of regional and international players. Leading participants such as Abdi Ibrahim Pharmaceuticals, Deva Holding, Nobel Pharmaceuticals, ?laç Sanayi ve Ticaret A.?., Sandoz Turkey, Sanofi Turkey, Pfizer Turkey, Roche Turkey, MSD Turkey, GSK Turkey, Novartis Turkey, Teva Turkey, Amgen Turkey, Merck Turkey, Eli Lilly Turkey contribute to innovation, geographic expansion, and service delivery in this space.
The Turkey BioPharma Generics and Biologics market is poised for significant transformation driven by technological advancements and increasing consumer awareness. As the healthcare infrastructure expands, the focus on personalized medicine and biosimilars is expected to grow, creating new avenues for innovation. Additionally, the government's commitment to enhancing local production capabilities will likely foster a more competitive environment, encouraging partnerships and collaborations that can lead to breakthroughs in drug development and distribution.
| Segment | Sub-Segments |
|---|---|
| By Type | Small Molecule Generics Biologics Biosimilars Combination Products Others |
| By Application | Oncology Cardiovascular Infectious Diseases Autoimmune Disorders Others |
| By Distribution Channel | Hospital Pharmacies Retail Pharmacies Online Pharmacies Wholesalers Others |
| By End-User | Hospitals Clinics Homecare Research Institutions Others |
| By Pricing Strategy | Cost-Plus Pricing Competitive Pricing Value-Based Pricing Others |
| By Regulatory Compliance | Local Regulations International Standards Quality Certifications Others |
| By Market Segment | Established Markets Emerging Markets Niche Markets Others |
| Scope Item/Segment | Sample Size | Target Respondent Profiles |
|---|---|---|
| Generics Market Insights | 150 | Product Managers, Market Analysts |
| Biologics Development Trends | 100 | R&D Directors, Regulatory Affairs Specialists |
| Healthcare Provider Perspectives | 80 | Pharmacists, Physicians |
| Market Access Strategies | 70 | Market Access Managers, Health Economists |
| Patient Access and Affordability | 90 | Patient Advocacy Representatives, Healthcare Policy Experts |
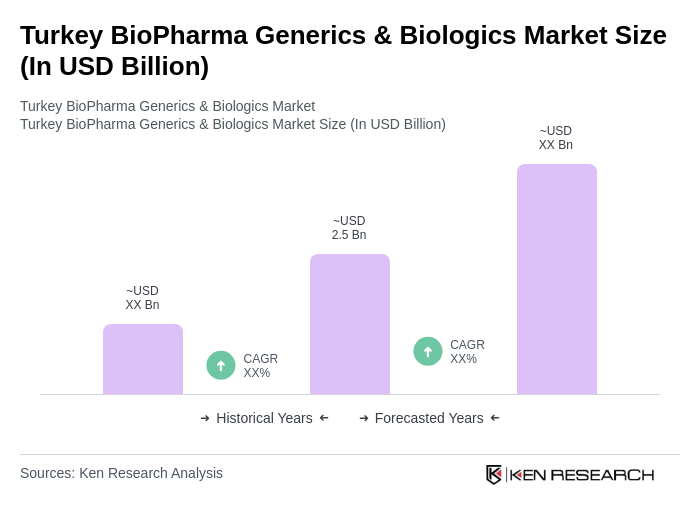
The Turkey BioPharma Generics & Biologics Market is valued at approximately USD 2.5 billion, reflecting a significant growth driven by the increasing prevalence of chronic diseases and rising healthcare expenditures.