Region:Middle East
Author(s):Dev
Product Code:KRAD6379
Pages:93
Published On:December 2025
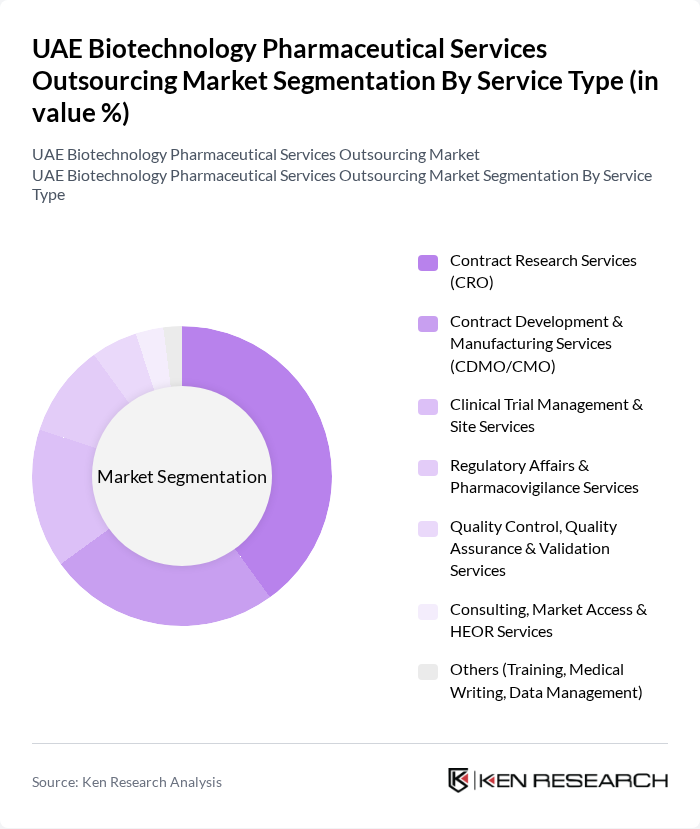
By Service Type:The service type segmentation includes various categories such as Contract Research Services (CRO), Contract Development & Manufacturing Services (CDMO/CMO), Clinical Trial Management & Site Services, Regulatory Affairs & Pharmacovigilance Services, Quality Control, Quality Assurance & Validation Services, Consulting, Market Access & HEOR Services, and Others (Training, Medical Writing, Data Management). Among these, Contract Research Services (CRO) is the leading sub-segment, driven by the increasing demand for clinical trials and research activities in the region. The growing number of pharmaceutical companies seeking to outsource their research functions has significantly contributed to the expansion of this segment.
By End-User:The end-user segmentation encompasses Pharmaceutical Companies, Biotechnology Firms, Medical Device & Diagnostics Companies, Academic & Research Institutions, Government & Public Health Agencies, and Others. Pharmaceutical Companies are the dominant end-user segment, primarily due to their extensive need for outsourcing services to enhance efficiency and reduce costs. The increasing complexity of drug development processes has led these companies to rely heavily on outsourcing to specialized service providers.

The UAE Biotechnology Pharmaceutical Services Outsourcing Market is characterized by a dynamic mix of regional and international players. Leading participants such as G42 Healthcare, PureHealth, Dubai Science Park, Dubai Healthcare City Authority, Neopharma, Gulf Pharmaceutical Industries (Julphar), Pharmax Pharmaceuticals, LIFEPharma FZE, Alliance Global Group (AGBL), M42 (including Abu Dhabi Health Data Services), Abu Dhabi Health Services Company (SEHA), Abu Dhabi Investment Office (ADIO) – Life Sciences Initiatives, Cleveland Clinic Abu Dhabi – Research & Innovation, Mohammed Bin Rashid University of Medicine and Health Sciences (MBRU), Dubai Academic Health Corporation & Dubai Clinical Research Center contribute to innovation, geographic expansion, and service delivery in this space.
The future of the UAE biotechnology pharmaceutical services outsourcing market appears promising, driven by increasing healthcare demands and technological advancements. The ongoing expansion of healthcare infrastructure and rising investments in R&D are expected to create a robust ecosystem for biopharmaceutical services. Additionally, the shift towards outsourcing will likely enhance operational efficiencies, allowing firms to focus on core competencies while leveraging external expertise. As the market evolves, collaboration with international firms will play a crucial role in driving innovation and competitiveness.
| Segment | Sub-Segments |
|---|---|
| By Service Type | Contract Research Services (CRO) Contract Development & Manufacturing Services (CDMO/CMO) Clinical Trial Management & Site Services Regulatory Affairs & Pharmacovigilance Services Quality Control, Quality Assurance & Validation Services Consulting, Market Access & HEOR Services Others (Training, Medical Writing, Data Management) |
| By End-User | Pharmaceutical Companies Biotechnology Firms Medical Device & Diagnostics Companies Academic & Research Institutions Government & Public Health Agencies Others |
| By Therapeutic Area | Oncology Cardiovascular & Metabolic Disorders Neurology & CNS Disorders Infectious & Communicable Diseases Immunology & Rare Diseases Others |
| By Outsourcing Model | Full-Service / End-to-End Outsourcing Functional Service Outsourcing (FSO) Project-Based Outsourcing Preferred Provider / Strategic Partnership Models Others |
| By Region | Abu Dhabi Dubai Sharjah Ras Al Khaimah, Ajman, Umm Al Quwain Fujairah & Northern Emirates (Others) |
| By Client Size | Large Multinational Enterprises Regional & Mid-Sized Enterprises Start-ups & Early-Stage Biotech Others |
| By Funding Source | Private & Corporate Investments Government Grants & Public Funding Venture Capital & Private Equity Others |
| Scope Item/Segment | Sample Size | Target Respondent Profiles |
|---|---|---|
| Clinical Trial Services | 45 | Clinical Research Coordinators, Project Managers |
| Regulatory Affairs Consulting | 40 | Regulatory Affairs Managers, Compliance Officers |
| Biomanufacturing Services | 35 | Production Managers, Quality Assurance Specialists |
| Market Access Strategies | 30 | Market Access Managers, Health Economists |
| Pharmacovigilance Services | 42 | Safety Officers, Pharmacovigilance Specialists |
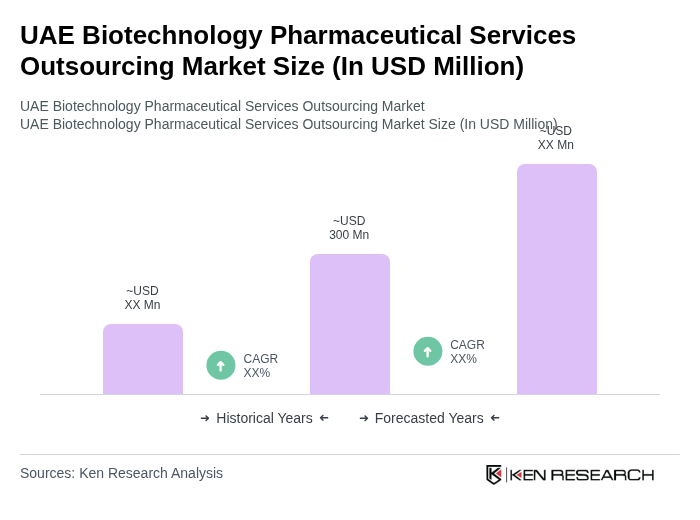
The UAE Biotechnology Pharmaceutical Services Outsourcing Market is valued at approximately USD 300 million, reflecting a five-year historical analysis. This growth is driven by factors such as innovative drug development and rising healthcare expenditures.