Region:Middle East
Author(s):Geetanshi
Product Code:KRAA3492
Pages:96
Published On:January 2026
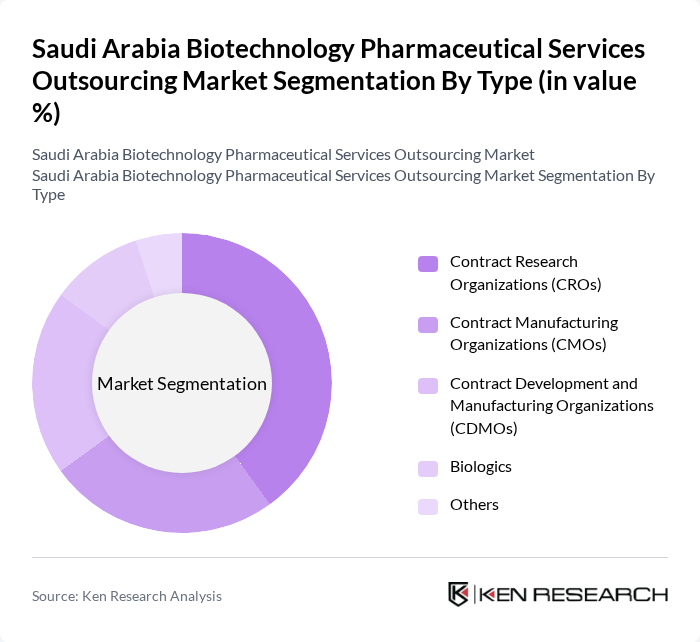
By Type:The market is segmented into various types, including Contract Research Organizations (CROs), Contract Manufacturing Organizations (CMOs), Contract Development and Manufacturing Organizations (CDMOs), Biologics, and Others. Among these, CROs are leading the market due to the increasing demand for clinical trials and research services from pharmaceutical companies.
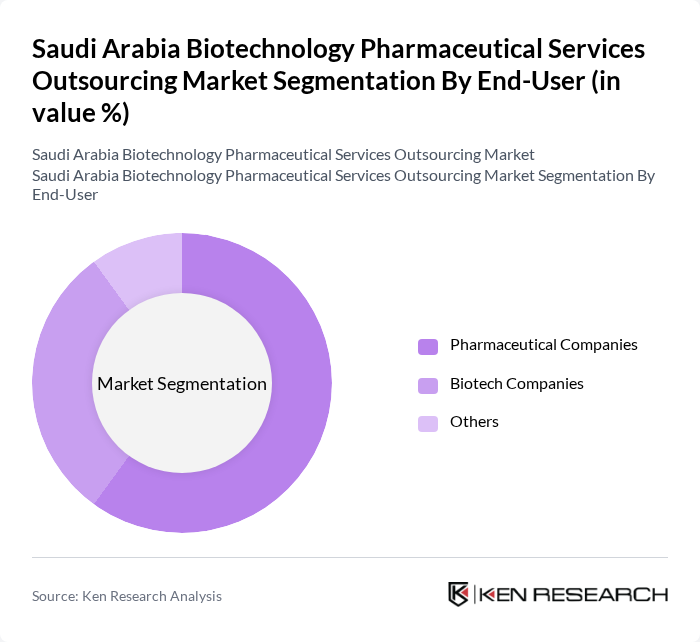
By End-User:The end-user segmentation includes Pharmaceutical Companies, Biotechnology Firms, Academic Institutions, Government Agencies, and Others. Pharmaceutical companies are the dominant end-users, driven by their need for outsourcing research and development to enhance efficiency and reduce costs.
The Saudi Arabia Biotechnology Pharmaceutical Services Outsourcing Market is characterized by a dynamic mix of regional and international players. Leading participants such as Saudi Pharmaceutical Industries and Medical Appliances Corporation (SPIMACO), Al-Dawaa Pharmacies, Saudi Biotech, Gulf Pharmaceutical Industries (Julphar), Advanced Pharmaceutical Industries, Al-Hokair Group, United Pharmacies, Al-Muhaidib Group, Tabuk Pharmaceuticals, Aster DM Healthcare, Hikma Pharmaceuticals, Novartis Pharmaceuticals, Pfizer Saudi Arabia, Sanofi Saudi Arabia, Merck Sharp & Dohme (MSD) Saudi Arabia contribute to innovation, geographic expansion, and service delivery in this space.
The future of the Saudi Arabian biotechnology pharmaceutical services outsourcing market appears promising, driven by increasing investments in healthcare infrastructure and a growing emphasis on innovative drug development. As the government continues to support biotechnology initiatives, the sector is likely to attract more international collaborations. Furthermore, advancements in artificial intelligence and machine learning are expected to streamline drug development processes, enhancing efficiency and reducing time-to-market for new therapies, thereby fostering market growth.
| Segment | Sub-Segments |
|---|---|
| By Type | Contract Research Organizations (CROs) Contract Manufacturing Organizations (CMOs) Contract Development and Manufacturing Organizations (CDMOs) Biologics Others |
| By End-User | Pharmaceutical Companies Biotechnology Firms Academic Institutions Government Agencies Others |
| By Service Type | Preclinical Services Clinical Trials Regulatory Affairs Pharmacovigilance Others |
| By Therapeutic Area | Oncology Cardiovascular Neurology Infectious Diseases Others |
| By Region | Central Region Eastern Region Western Region Southern Region |
| By Client Type | Large Enterprises Small and Medium Enterprises (SMEs) Startups Others |
| By Project Size | Small Projects Medium Projects Large Projects Others |
| Scope Item/Segment | Sample Size | Target Respondent Profiles |
|---|---|---|
| Clinical Trial Services | 45 | Clinical Research Managers, Biostatisticians |
| Regulatory Affairs Outsourcing | 40 | Regulatory Affairs Specialists, Compliance Officers |
| Biomanufacturing Services | 50 | Production Managers, Quality Assurance Leads |
| Market Access Consulting | 40 | Market Access Managers, Health Economists |
| Pharmaceutical Supply Chain Management | 45 | Supply Chain Directors, Procurement Managers |
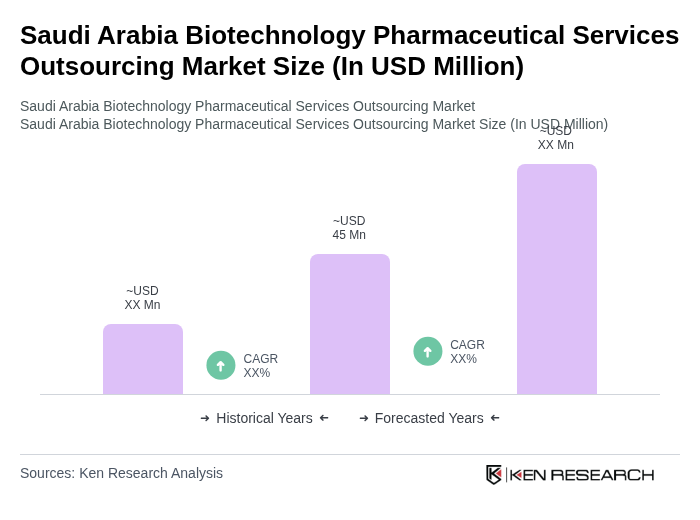
The Saudi Arabia Biotechnology Pharmaceutical Services Outsourcing Market is valued at approximately USD 45 million, reflecting a five-year historical analysis driven by increased healthcare investments and demand for innovative therapies.