Region:Asia
Author(s):Rebecca
Product Code:KRAA9384
Pages:90
Published On:November 2025
By Type:This segmentation includes various service types that cater to the needs of pharmaceutical and biotechnology companies. The subsegments are Contract Research Organizations (CROs), Contract Manufacturing Organizations (CMOs), Contract Development and Manufacturing Organizations (CDMOs), Regulatory Affairs Outsourcing, and Pharmacovigilance Services. Each of these plays a crucial role in the drug development lifecycle, from research to market entry.
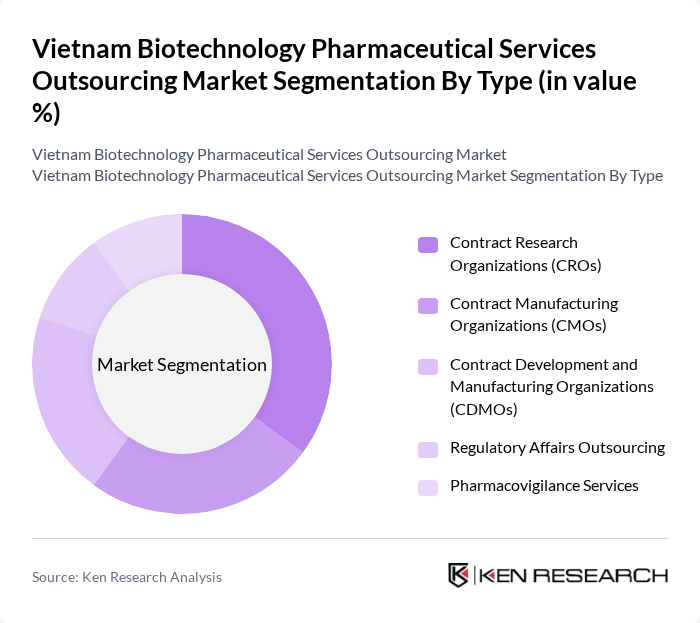
The Contract Research Organizations (CROs) segment is currently dominating the market due to the increasing number of clinical trials and the demand for specialized research services. Pharmaceutical companies are increasingly outsourcing their research activities to CROs to reduce costs and improve efficiency. This trend is driven by the need for faster drug development timelines and the growing complexity of clinical trials, which require specialized expertise and resources.
By End-User:This segmentation focuses on the various entities that utilize biotechnology pharmaceutical services. The subsegments include Pharmaceutical Companies, Biotechnology Firms, Academic & Research Institutes, Hospitals & Healthcare Providers, and Others. Each end-user has distinct needs and requirements that drive their engagement with outsourcing services.
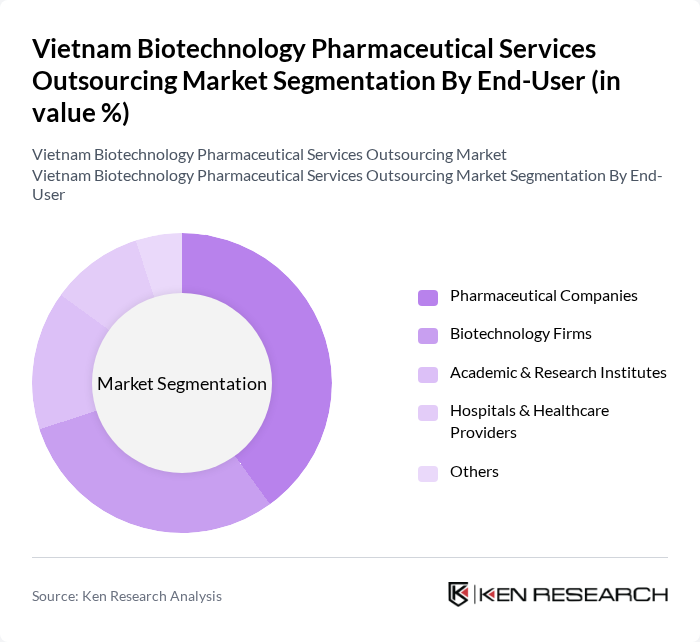
Pharmaceutical Companies represent the largest end-user segment, driven by their need for comprehensive outsourcing solutions to manage the complexities of drug development. These companies often seek to leverage the expertise of outsourcing partners to enhance their research capabilities, reduce operational costs, and expedite the time-to-market for new drugs. The increasing trend of collaboration between pharmaceutical companies and outsourcing service providers is expected to continue, further solidifying this segment's dominance.
The Vietnam Biotechnology Pharmaceutical Services Outsourcing Market is characterized by a dynamic mix of regional and international players. Leading participants such as VinBioCare, Vabiotech, Medlatec Group, Ho Chi Minh City Biotechnology Center, Saigon Pharmaceutical Company (Sapharco), Vietnam Pharmaceutical Corporation (Vinapharm), Biomedic JSC, Imexpharm Corporation, DHG Pharmaceutical JSC, Binh Dinh Pharmaceutical and Medical Equipment JSC (Bidiphar), Hanoi Medical University, FPT Long Chau Pharma JSC, An Phuoc Pharmaceutical, Dong Nai Pharmaceutical JSC (Donapharco), Binh Duong Pharmaceutical JSC (Bidipharco) contribute to innovation, geographic expansion, and service delivery in this space.
The future of the Vietnam biotechnology pharmaceutical services outsourcing market appears promising, driven by increasing investments in research and development, alongside a growing emphasis on personalized medicine. As the government continues to enhance regulatory frameworks and support for biotechnology initiatives, the market is expected to attract more international collaborations. Additionally, advancements in digital health technologies and telemedicine are likely to reshape service delivery, making healthcare more accessible and efficient for the Vietnamese population.
| Segment | Sub-Segments |
|---|---|
| By Type | Contract Research Organizations (CROs) Contract Manufacturing Organizations (CMOs) Contract Development and Manufacturing Organizations (CDMOs) Regulatory Affairs Outsourcing Pharmacovigilance Services |
| By End-User | Pharmaceutical Companies Biotechnology Firms Academic & Research Institutes Hospitals & Healthcare Providers Others |
| By Therapeutic Area | Oncology Infectious Diseases Cardiovascular Neurology Metabolic Disorders Others |
| By Service Type | Preclinical Services Clinical Trial Management Regulatory Affairs Data Management & Biostatistics Pharmacovigilance & Safety Services Manufacturing & Supply Chain Services Others |
| By Region | Northern Vietnam Southern Vietnam Central Vietnam |
| By Client Type | Startups Established Companies Government Agencies Multinational Corporations Others |
| By Funding Source | Private Investments Government Grants International Funding Public-Private Partnerships Others |
| Scope Item/Segment | Sample Size | Target Respondent Profiles |
|---|---|---|
| Clinical Trial Outsourcing | 100 | Clinical Research Managers, Biotech Executives |
| Manufacturing Services | 70 | Production Managers, Quality Assurance Officers |
| Regulatory Affairs Consulting | 60 | Regulatory Affairs Specialists, Compliance Managers |
| Market Access Strategies | 50 | Market Access Managers, Business Development Directors |
| Pharmaceutical Supply Chain Management | 80 | Supply Chain Managers, Logistics Coordinators |
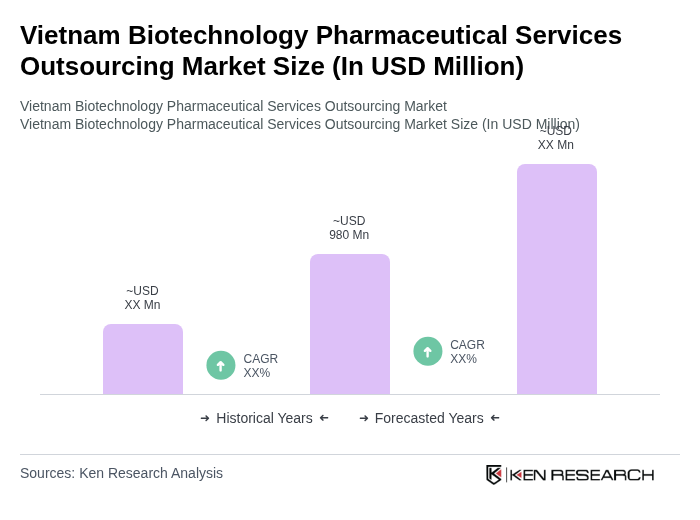
The Vietnam Biotechnology Pharmaceutical Services Outsourcing Market is valued at approximately USD 980 million, reflecting significant growth driven by increased demand for innovative drug development and rising healthcare expenditures.