Region:Middle East
Author(s):Geetanshi
Product Code:KRAD1156
Pages:99
Published On:November 2025
 Drugs Market.png)
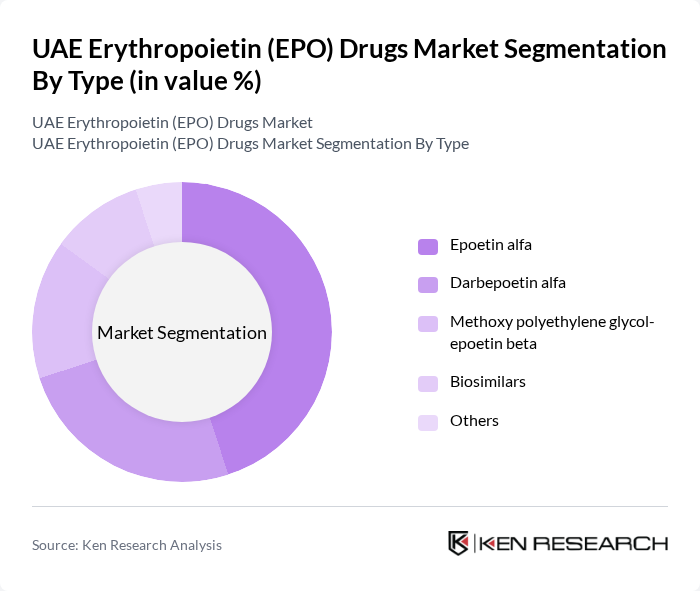
By Type:The market is segmented into various types of EPO drugs, including Epoetin alfa, Darbepoetin alfa, Methoxy polyethylene glycol-epoetin beta, Biosimilars, and Others. Among these, Epoetin alfa remains the most widely used due to its established efficacy and safety profile, making it the preferred choice for treating anemia in patients with chronic kidney disease and cancer. The adoption of biosimilars is also increasing, driven by cost-effectiveness and supportive regulatory pathways .
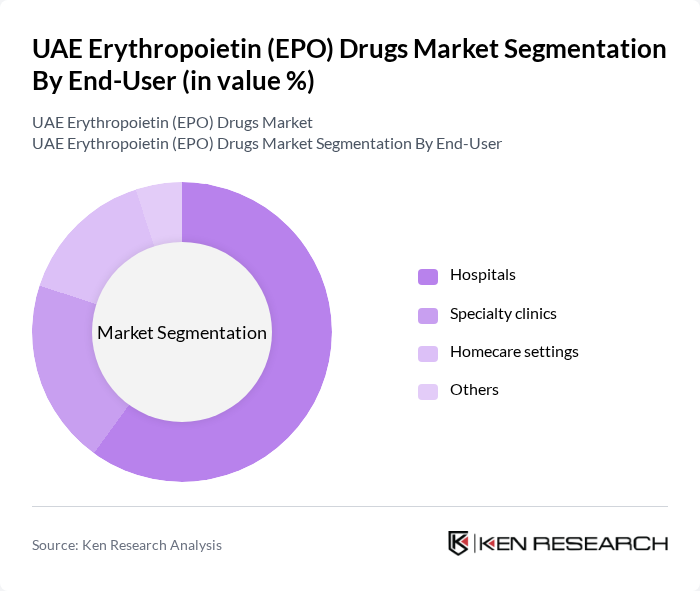
By End-User:The end-user segmentation includes Hospitals, Specialty clinics, Homecare settings, and Others. Hospitals are the leading end-users of EPO drugs, primarily due to their capacity to manage complex anemia cases and provide comprehensive care for patients with chronic kidney disease and cancer. Specialty clinics and homecare settings are witnessing gradual growth as outpatient management and home-based therapies become more prevalent .
The UAE Erythropoietin (EPO) Drugs Market is characterized by a dynamic mix of regional and international players. Leading participants such as Amgen Inc., Roche Holding AG, Johnson & Johnson, Pfizer Inc., Novartis AG, Sanofi S.A., Merck & Co., Inc., Teva Pharmaceutical Industries Ltd., AbbVie Inc., Hikma Pharmaceuticals PLC, GlaxoSmithKline plc (GSK), Astellas Pharma Inc., Sandoz (a Novartis division), Julphar (Gulf Pharmaceutical Industries), Cipla Ltd. contribute to innovation, geographic expansion, and service delivery in this space.
The UAE Erythropoietin (EPO) drugs market is poised for significant advancements, driven by the increasing focus on personalized medicine and patient-centric care. As healthcare providers adopt innovative treatment approaches, the integration of telemedicine and remote monitoring will enhance patient engagement and adherence. Furthermore, the growing emphasis on cost-effective treatments will encourage the development of affordable EPO alternatives, fostering a competitive landscape that benefits patients and healthcare systems alike.
| Segment | Sub-Segments |
|---|---|
| By Type | Epoetin alfa Darbepoetin alfa Methoxy polyethylene glycol-epoetin beta Biosimilars Others |
| By End-User | Hospitals Specialty clinics Homecare settings Others |
| By Patient Demographics | Pediatric patients Adult patients Geriatric patients Others |
| By Distribution Channel | Hospital pharmacies Retail pharmacies Online pharmacies Others |
| By Administration Route | Subcutaneous injection Intravenous injection Others |
| By Treatment Type | Anemia due to chronic kidney disease Anemia due to cancer Anemia due to other causes Others |
| By Region | Abu Dhabi Dubai Sharjah Others |
| Scope Item/Segment | Sample Size | Target Respondent Profiles |
|---|---|---|
| Oncology Treatment Centers | 60 | Oncologists, Treatment Coordinators |
| Nephrology Clinics | 50 | Nephrologists, Nurse Practitioners |
| Pharmacy Chains | 40 | Pharmacy Managers, Procurement Officers |
| Patient Advocacy Groups | 40 | Patient Representatives, Healthcare Advocates |
| Health Insurance Providers | 40 | Policy Analysts, Claims Managers |
The UAE Erythropoietin (EPO) Drugs Market is valued at approximately USD 95 million, reflecting a five-year historical analysis. This valuation is influenced by the increasing prevalence of anemia and advancements in biotechnology.