Region:Middle East
Author(s):Rebecca
Product Code:KRAC2924
Pages:83
Published On:January 2026
By Device Type:
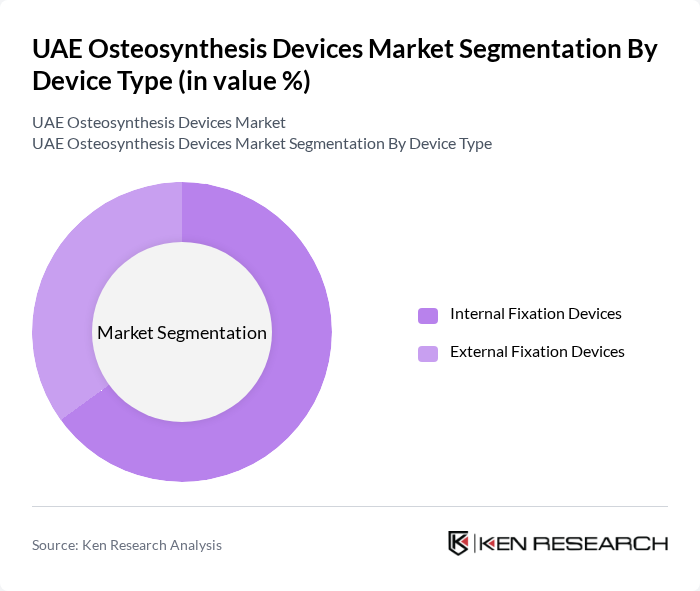
The market is segmented into Internal Fixation Devices and External Fixation Devices. Internal Fixation Devices dominate the market due to their effectiveness in stabilizing fractures and promoting faster healing. These devices, such as plates and screws, are widely used in various orthopedic surgeries, making them a preferred choice among surgeons. External Fixation Devices, while also important, are typically used in more complex cases or for temporary stabilization, which limits their market share compared to internal devices.
By Fracture Type:
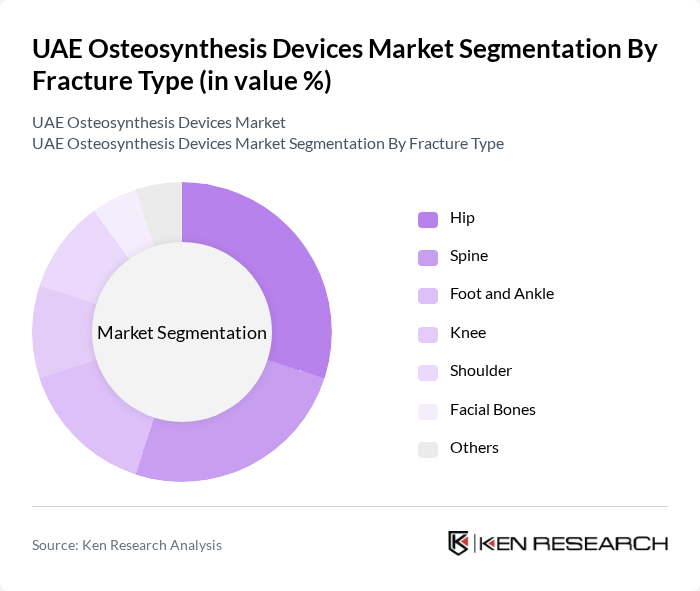
The market is segmented by fracture type into Hip, Spine, Foot and Ankle, Knee, Shoulder, Facial Bones, and Others. The Hip fracture segment leads the market due to the high prevalence of hip fractures among the elderly population, driven by factors such as osteoporosis and falls. The Spine and Knee segments also show significant demand, attributed to the increasing number of sports-related injuries and degenerative conditions. Other fracture types, while important, do not match the volume of these leading segments.
The UAE Osteosynthesis Devices Market is characterized by a dynamic mix of regional and international players. Leading participants such as Stryker Corporation, DePuy Synthes, Zimmer Biomet, Medtronic, Smith & Nephew, B. Braun Melsungen AG, Orthofix Medical Inc., NuVasive, Inc., Aesculap Implant Systems, LLC, Conmed Corporation, Arthrex, Inc., KLS Martin Group, Medartis AG, Acumed LLC, Orthofix International N.V. contribute to innovation, geographic expansion, and service delivery in this space.
The future of the UAE osteosynthesis devices market appears promising, driven by ongoing advancements in technology and an increasing focus on patient-centered care. As healthcare infrastructure expands, particularly in rural areas, access to orthopedic services is expected to improve. Additionally, the integration of smart technologies into devices will likely enhance surgical outcomes, while the growing emphasis on minimally invasive procedures will further stimulate market growth, ensuring that patients receive effective and efficient treatment options.
| Segment | Sub-Segments |
|---|---|
| By Device Type | Internal Fixation Devices External Fixation Devices |
| By Fracture Type | Hip Spine Foot and Ankle Knee Shoulder Facial Bones Others |
| By Material | Stainless Steel Titanium Bioresorbable Materials Polymers Others |
| By End-User | Hospitals Orthopaedic Clinics Ambulatory Surgical Centers Others |
| Scope Item/Segment | Sample Size | Target Respondent Profiles |
|---|---|---|
| Orthopedic Surgeons | 100 | Consultant Surgeons, Orthopedic Specialists |
| Hospital Procurement Managers | 80 | Supply Chain Managers, Purchasing Directors |
| Medical Device Distributors | 60 | Sales Managers, Distribution Executives |
| Healthcare Facility Administrators | 70 | Operations Managers, Facility Directors |
| Regulatory Affairs Professionals | 50 | Compliance Officers, Regulatory Managers |
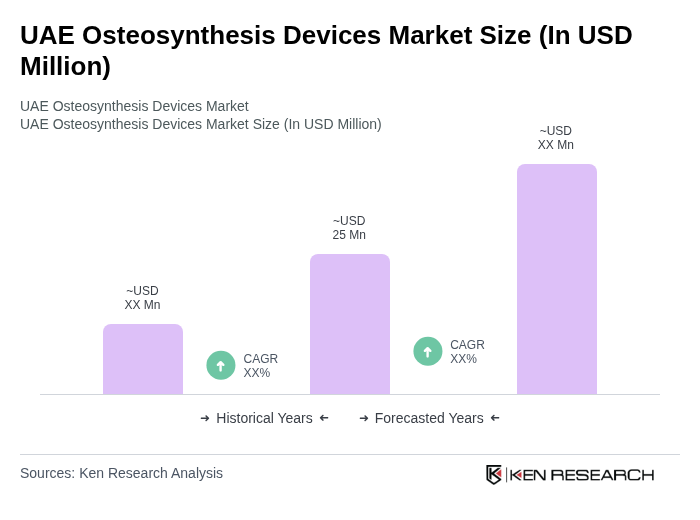
The UAE Osteosynthesis Devices Market is valued at approximately USD 30 million, reflecting a five-year historical analysis. This growth is driven by increasing orthopedic injuries, advancements in surgical techniques, and a rising geriatric population requiring surgical interventions.