About the Report
Base Year 2024UAE Sterile Injectable Contract Manufacturing Market Overview
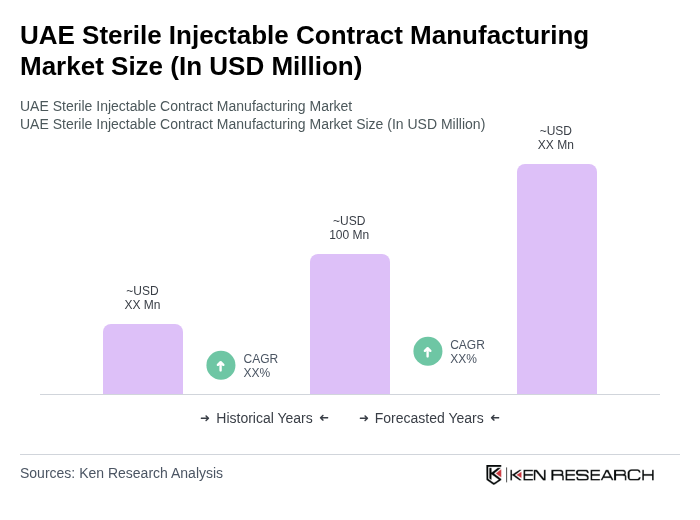
- The UAE Sterile Injectable Contract Manufacturing Market is valued at USD 100 million, based on a five-year historical analysis. This growth is primarily driven by the increasing demand for biologics and biosimilars, the rising prevalence of chronic diseases, and the expansion of healthcare infrastructure. The market is further supported by the growing trend of outsourcing manufacturing processes by pharmaceutical and biopharmaceutical companies, as well as investments in advanced fill-finish technologies and automation to ensure product safety and efficiency .
- Key cities dominating this market include Dubai and Abu Dhabi, which serve as major healthcare hubs in the region. Their dominance is attributed to the presence of advanced healthcare facilities, a robust regulatory framework, and significant investments in biotechnology and pharmaceutical sectors. These cities also benefit from strategic geographic locations that facilitate trade and access to international markets .
- The "Ministerial Resolution No. 404 of 2015 Concerning the Controls and Requirements for Manufacturing and Importing of Pharmaceutical Products," issued by the UAE Ministry of Health and Prevention, governs the approval process for sterile injectables. This regulation sets operational requirements for local and international manufacturers, including facility licensing, GMP compliance, and product registration, thereby streamlining market entry and encouraging investment in the UAE sterile injectables sector.
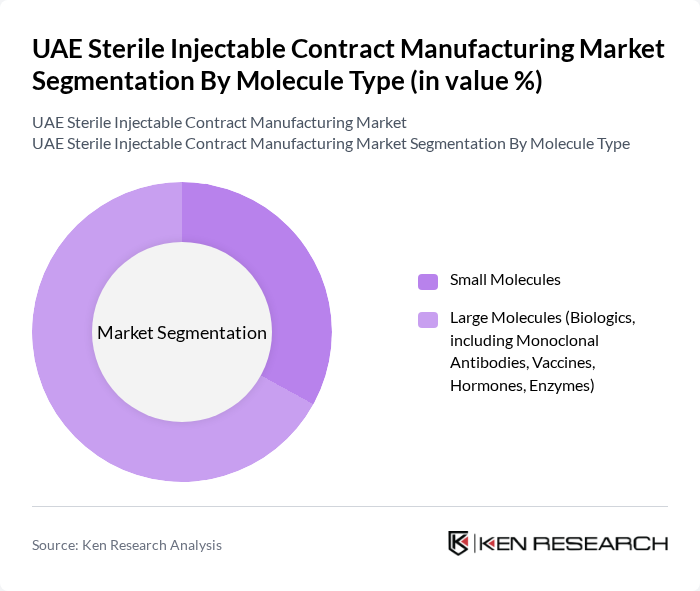
UAE Sterile Injectable Contract Manufacturing Market Segmentation
By Molecule Type:The market is segmented into two primary categories: Small Molecules and Large Molecules. Small Molecules are typically low molecular weight compounds that are easier to manufacture and have a long-standing presence in the pharmaceutical industry. In contrast, Large Molecules, which include biologics such as monoclonal antibodies, vaccines, hormones, and enzymes, are gaining traction due to their effectiveness in treating complex diseases. The demand for Large Molecules is particularly high, driven by advancements in biotechnology, the increasing focus on personalized medicine, and the rising number of biopharmaceutical companies outsourcing production .
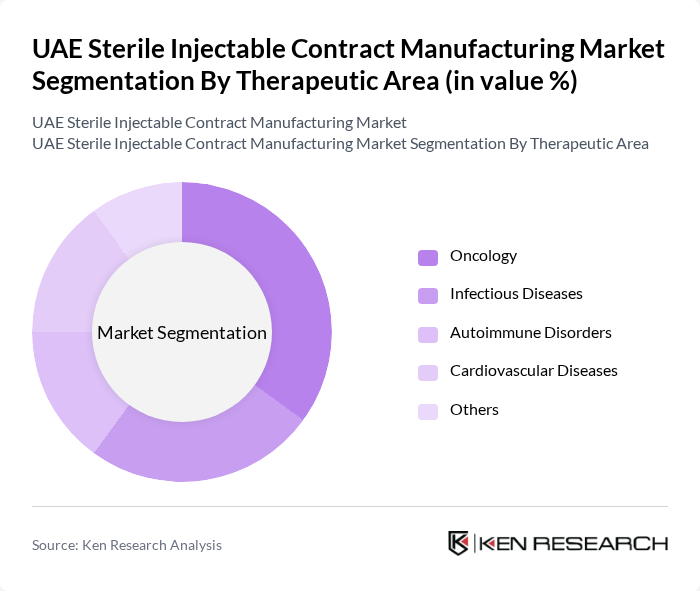
By Therapeutic Area:The therapeutic areas covered in this market include Oncology, Infectious Diseases, Autoimmune Disorders, Cardiovascular Diseases, and Others. Oncology is currently the leading therapeutic area, driven by the rising incidence of cancer and the increasing availability of innovative therapies. The demand for injectable treatments in oncology is particularly strong, as they often provide targeted and effective treatment options for patients. Infectious diseases also represent a significant segment, especially in light of recent global health challenges and the increased focus on vaccine production .
UAE Sterile Injectable Contract Manufacturing Market Competitive Landscape
The UAE Sterile Injectable Contract Manufacturing Market is characterized by a dynamic mix of regional and international players. Leading participants such as Julphar (Gulf Pharmaceutical Industries), Neopharma, Globalpharma (Sanofi Company), Medpharma (Julphar Subsidiary), Pharmax Pharmaceuticals, LifePharma FZE, Aster DM Healthcare, Tabuk Pharmaceuticals Manufacturing Co., United Pharmaceuticals, Biogenix Lab, Amana Healthcare, Mediserv, Al Hayat Pharmaceuticals, Al Jazeera Pharmaceutical Industries, Al Mufeed Pharmaceutical Industries contribute to innovation, geographic expansion, and service delivery in this space.
UAE Sterile Injectable Contract Manufacturing Market Industry Analysis
Growth Drivers
- Increasing Demand for Biologics:The UAE's healthcare sector is witnessing a significant rise in the demand for biologics, driven by a growing population and increased healthcare spending. In future, the UAE's healthcare expenditure is projected to reach approximately AED 66 billion, reflecting a 10% increase from the previous year. This surge is largely attributed to the rising prevalence of diseases that require biologic treatments, such as cancer and autoimmune disorders, which are expected to drive the sterile injectable market significantly.
- Rising Prevalence of Chronic Diseases:Chronic diseases are becoming increasingly prevalent in the UAE, with diabetes and cardiovascular diseases affecting over 35% of the adult population. According to the World Health Organization, the number of diabetes cases in the UAE is expected to rise to 1.7 million in future. This alarming trend is propelling the demand for sterile injectables, as these treatments are essential for managing chronic conditions effectively and improving patient outcomes.
- Advancements in Manufacturing Technologies:The sterile injectable market in the UAE is benefiting from rapid advancements in manufacturing technologies, including automation and process optimization. In future, investments in advanced manufacturing technologies are expected to exceed AED 1.1 billion, enhancing production efficiency and product quality. These innovations are crucial for meeting the stringent regulatory standards and increasing the capacity to produce complex biologics, thereby driving market growth.
Market Challenges
- Stringent Regulatory Requirements:The UAE's sterile injectable market faces significant challenges due to stringent regulatory requirements imposed by health authorities. Compliance with the UAE's Ministry of Health and Prevention regulations necessitates extensive documentation and quality assurance processes, which can delay product launches. In future, the cost of compliance is projected to rise by 15%, impacting the operational efficiency of manufacturers and potentially hindering market growth.
- High Operational Costs:The operational costs associated with sterile injectable manufacturing in the UAE are notably high, primarily due to the need for specialized facilities and equipment. In future, the average operational cost for manufacturers is expected to reach AED 550 million, driven by the need for advanced technology and skilled labor. These high costs can deter new entrants and limit the ability of existing companies to expand their production capabilities.
UAE Sterile Injectable Contract Manufacturing Market Future Outlook
The future of the UAE sterile injectable contract manufacturing market appears promising, driven by ongoing investments in healthcare infrastructure and technological advancements. As the demand for personalized medicine grows, manufacturers are likely to adapt their production processes to meet these evolving needs. Additionally, the increasing trend of outsourcing production to specialized contract manufacturing organizations (CMOs) will further enhance operational efficiencies and allow companies to focus on core competencies, ultimately benefiting the market landscape.
Market Opportunities
- Expansion of Healthcare Infrastructure:The UAE government is investing heavily in healthcare infrastructure, with plans to increase the number of hospitals and clinics by 25% in future. This expansion will create a greater demand for sterile injectables, providing manufacturers with opportunities to establish partnerships and expand their market presence significantly.
- Growth in Outsourcing Trends:The trend of outsourcing manufacturing processes is gaining traction in the UAE, with an estimated 35% of pharmaceutical companies expected to outsource their sterile injectable production in future. This shift presents a lucrative opportunity for contract manufacturers to capture a larger share of the market, as companies seek to reduce costs and improve efficiency.
Scope of the Report
| Segment | Sub-Segments |
|---|---|
| By Molecule Type | Small Molecules Large Molecules (Biologics, including Monoclonal Antibodies, Vaccines, Hormones, Enzymes) |
| By Therapeutic Area | Oncology Infectious Diseases Autoimmune Disorders Cardiovascular Diseases Others |
| By End-User | Pharmaceutical Companies Biopharmaceutical Companies Research Institutions Others |
| By Packaging Type | Vials Ampoules Pre-filled Syringes Cartridges Others |
| By Distribution Channel | Direct Sales Distributors Online Sales Others |
| By Region | Abu Dhabi Dubai Sharjah Others |
| By Regulatory Compliance | GMP Compliance UAE Ministry of Health & Prevention (MOHAP) Approval US FDA/EMA Compliance Others |
Key Target Audience
Investors and Venture Capitalist Firms
Government and Regulatory Bodies (e.g., Ministry of Health and Prevention, Emirates Authority for Standardization and Metrology)
Pharmaceutical Manufacturers
Biotechnology Companies
Contract Research Organizations (CROs)
Healthcare Providers and Hospitals
Logistics and Supply Chain Companies
Pharmaceutical Distributors
Players Mentioned in the Report:
Julphar (Gulf Pharmaceutical Industries)
Neopharma
Globalpharma (Sanofi Company)
Medpharma (Julphar Subsidiary)
Pharmax Pharmaceuticals
LifePharma FZE
Aster DM Healthcare
Tabuk Pharmaceuticals Manufacturing Co.
United Pharmaceuticals
Biogenix Lab
Amana Healthcare
Mediserv
Al Hayat Pharmaceuticals
Al Jazeera Pharmaceutical Industries
Al Mufeed Pharmaceutical Industries
Table of Contents
Market Assessment Phase
1. Executive Summary and Approach
2. UAE Sterile Injectable Contract Manufacturing Market Overview
2.1 Key Insights and Strategic Recommendations
2.2 UAE Sterile Injectable Contract Manufacturing Market Overview
2.3 Definition and Scope
2.4 Evolution of Market Ecosystem
2.5 Timeline of Key Regulatory Milestones
2.6 Value Chain & Stakeholder Mapping
2.7 Business Cycle Analysis
2.8 Policy & Incentive Landscape
3. UAE Sterile Injectable Contract Manufacturing Market Analysis
3.1 Growth Drivers
3.1.1 Increasing demand for biologics
3.1.2 Rising prevalence of chronic diseases
3.1.3 Advancements in manufacturing technologies
3.1.4 Favorable government initiatives
3.2 Market Challenges
3.2.1 Stringent regulatory requirements
3.2.2 High operational costs
3.2.3 Limited skilled workforce
3.2.4 Supply chain disruptions
3.3 Market Opportunities
3.3.1 Expansion of healthcare infrastructure
3.3.2 Growth in outsourcing trends
3.3.3 Increasing investment in R&D
3.3.4 Emergence of personalized medicine
3.4 Market Trends
3.4.1 Shift towards sustainable manufacturing practices
3.4.2 Integration of digital technologies
3.4.3 Rise of contract manufacturing organizations (CMOs)
3.4.4 Focus on quality assurance and compliance
3.5 Government Regulation
3.5.1 Regulatory frameworks for sterile products
3.5.2 Compliance with international standards
3.5.3 Licensing requirements for manufacturers
3.5.4 Monitoring and inspection protocols
4. SWOT Analysis
5. Stakeholder Analysis
6. Porter's Five Forces Analysis
7. UAE Sterile Injectable Contract Manufacturing Market Size, 2019-2024
7.1 By Value
7.2 By Volume
7.3 By Average Selling Price
8. UAE Sterile Injectable Contract Manufacturing Market Segmentation
8.1 By Molecule Type
8.1.1 Small Molecules
8.1.2 Large Molecules (Biologics, including Monoclonal Antibodies, Vaccines, Hormones, Enzymes)
8.2 By Therapeutic Area
8.2.1 Oncology
8.2.2 Infectious Diseases
8.2.3 Autoimmune Disorders
8.2.4 Cardiovascular Diseases
8.2.5 Others
8.3 By End-User
8.3.1 Pharmaceutical Companies
8.3.2 Biopharmaceutical Companies
8.3.3 Research Institutions
8.3.4 Others
8.4 By Packaging Type
8.4.1 Vials
8.4.2 Ampoules
8.4.3 Pre-filled Syringes
8.4.4 Cartridges
8.4.5 Others
8.5 By Distribution Channel
8.5.1 Direct Sales
8.5.2 Distributors
8.5.3 Online Sales
8.5.4 Others
8.6 By Region
8.6.1 Abu Dhabi
8.6.2 Dubai
8.6.3 Sharjah
8.6.4 Others
8.7 By Regulatory Compliance
8.7.1 GMP Compliance
8.7.2 UAE Ministry of Health & Prevention (MOHAP) Approval
8.7.3 US FDA/EMA Compliance
8.7.4 Others
9. UAE Sterile Injectable Contract Manufacturing Market Competitive Analysis
9.1 Market Share of Key Players
9.2 Cross Comparison of Key Players
9.2.1 Company Name
9.2.2 Group Size (Large, Medium, or Small as per industry convention)
9.2.3 Revenue (USD Million, Latest Year)
9.2.4 Revenue Growth Rate (%)
9.2.5 Market Share (%)
9.2.6 Production Capacity (Million Units/Year)
9.2.7 Capacity Utilization Rate (%)
9.2.8 Number of Sterile Injectable Facilities (UAE)
9.2.9 Regulatory Approvals (GMP, MOHAP, US FDA, EMA)
9.2.10 R&D Investment as % of Revenue
9.2.11 Number of Active Clients/Projects
9.2.12 On-Time Delivery Rate (%)
9.2.13 Quality Assurance Metrics (Batch Failure Rate, Audit Findings)
9.2.14 Supply Chain Lead Time (Days)
9.2.15 Customer Retention Rate (%)
9.2.16 Pricing Strategy (Premium, Value, Cost-Plus)
9.3 SWOT Analysis of Top Players
9.4 Pricing Analysis
9.5 Detailed Profile of Major Companies
9.5.1 Julphar (Gulf Pharmaceutical Industries)
9.5.2 Neopharma
9.5.3 Globalpharma (Sanofi Company)
9.5.4 Medpharma (Julphar Subsidiary)
9.5.5 Pharmax Pharmaceuticals
9.5.6 LifePharma FZE
9.5.7 Aster DM Healthcare
9.5.8 Tabuk Pharmaceuticals Manufacturing Co.
9.5.9 United Pharmaceuticals
9.5.10 Biogenix Lab
9.5.11 Amana Healthcare
9.5.12 Mediserv
9.5.13 Al Hayat Pharmaceuticals
9.5.14 Al Jazeera Pharmaceutical Industries
9.5.15 Al Mufeed Pharmaceutical Industries
10. UAE Sterile Injectable Contract Manufacturing Market End-User Analysis
10.1 Procurement Behavior of Key Ministries
10.1.1 Ministry of Health and Prevention
10.1.2 Ministry of Industry and Advanced Technology
10.1.3 Ministry of Finance
10.1.4 Others
10.2 Corporate Spend on Infrastructure & Energy
10.2.1 Investment in Healthcare Facilities
10.2.2 Budget Allocation for R&D
10.2.3 Spending on Technology Upgrades
10.2.4 Others
10.3 Pain Point Analysis by End-User Category
10.3.1 Pharmaceutical Companies
10.3.2 Biotechnology Firms
10.3.3 Research Institutions
10.3.4 Others
10.4 User Readiness for Adoption
10.4.1 Awareness of Contract Manufacturing Benefits
10.4.2 Training and Skill Development Needs
10.4.3 Infrastructure Readiness
10.4.4 Others
10.5 Post-Deployment ROI and Use Case Expansion
10.5.1 Measurement of ROI
10.5.2 Case Studies of Successful Implementations
10.5.3 Future Use Case Scenarios
10.5.4 Others
11. UAE Sterile Injectable Contract Manufacturing Market Future Size, 2025-2030
11.1 By Value
11.2 By Volume
11.3 By Average Selling Price
Go-To-Market Strategy Phase
1. Whitespace Analysis + Business Model Canvas
1.1 Market Gaps Identification
1.2 Business Model Development
2. Marketing and Positioning Recommendations
2.1 Branding Strategies
2.2 Product USPs
3. Distribution Plan
3.1 Urban Retail vs Rural NGO Tie-ups
4. Channel & Pricing Gaps
4.1 Underserved Routes
4.2 Pricing Bands
5. Unmet Demand & Latent Needs
5.1 Category Gaps
5.2 Consumer Segments
6. Customer Relationship
6.1 Loyalty Programs
6.2 After-sales Service
7. Value Proposition
7.1 Sustainability
7.2 Integrated Supply Chains
8. Key Activities
8.1 Regulatory Compliance
8.2 Branding
8.3 Distribution Setup
9. Entry Strategy Evaluation
9.1 Domestic Market Entry Strategy
9.1.1 Product Mix
9.1.2 Pricing Band
9.1.3 Packaging
9.2 Export Entry Strategy
9.2.1 Target Countries
9.2.2 Compliance Roadmap
10. Entry Mode Assessment
10.1 JV
10.2 Greenfield
10.3 M&A
10.4 Distributor Model
11. Capital and Timeline Estimation
11.1 Capital Requirements
11.2 Timelines
12. Control vs Risk Trade-Off
12.1 Ownership vs Partnerships
13. Profitability Outlook
13.1 Breakeven Analysis
13.2 Long-term Sustainability
14. Potential Partner List
14.1 Distributors
14.2 JVs
14.3 Acquisition Targets
15. Execution Roadmap
15.1 Phased Plan for Market Entry
15.1.1 Market Setup
15.1.2 Market Entry
15.1.3 Growth Acceleration
15.1.4 Scale & Stabilize
15.2 Key Activities and Milestones
15.2.1 Milestone Planning
15.2.2 Activity Tracking
Research Methodology
Phase 1: Approach1
Desk Research
- Analysis of industry reports from regulatory bodies such as the UAE Ministry of Health and Prevention
- Review of market studies and white papers published by pharmaceutical associations in the UAE
- Examination of trade publications and journals focusing on sterile injectable manufacturing trends
Primary Research
- Interviews with key stakeholders in the pharmaceutical sector, including manufacturing executives
- Surveys conducted with quality assurance managers in sterile injectable facilities
- Field interviews with regulatory affairs specialists to understand compliance challenges
Validation & Triangulation
- Cross-validation of data through multiple sources, including industry reports and expert opinions
- Triangulation of findings from primary interviews with secondary data insights
- Sanity checks through feedback from a panel of industry experts and consultants
Phase 2: Market Size Estimation1
Top-down Assessment
- Estimation of market size based on national healthcare expenditure and pharmaceutical sales data
- Segmentation of the market by therapeutic areas and types of sterile injectables
- Incorporation of growth rates from government healthcare initiatives and policies
Bottom-up Modeling
- Collection of production data from leading contract manufacturers in the UAE
- Operational cost analysis based on pricing models of sterile injectable products
- Volume estimates derived from historical sales data and projected demand trends
Forecasting & Scenario Analysis
- Utilization of time-series analysis to project future market growth based on historical data
- Scenario modeling based on potential regulatory changes and market entry of new players
- Development of baseline, optimistic, and pessimistic forecasts through 2030
Phase 3: CATI Sample Composition1
| Scope Item/Segment | Sample Size | Target Respondent Profiles |
|---|---|---|
| Pharmaceutical Manufacturing Facilities | 80 | Production Managers, Quality Control Supervisors |
| Regulatory Compliance in Sterile Injectables | 50 | Regulatory Affairs Managers, Compliance Officers |
| Market Trends in Contract Manufacturing | 60 | Business Development Managers, Market Analysts |
| Healthcare Providers' Perspectives | 70 | Pharmacists, Hospital Procurement Officers |
| End-user Feedback on Sterile Injectables | 40 | Healthcare Practitioners, Clinical Researchers |
Frequently Asked Questions
What is the current value of the UAE Sterile Injectable Contract Manufacturing Market?
The UAE Sterile Injectable Contract Manufacturing Market is valued at approximately USD 100 million, reflecting a five-year historical analysis. This growth is driven by increasing demand for biologics, chronic disease prevalence, and advancements in healthcare infrastructure.