Region:North America
Author(s):Rebecca
Product Code:KRAC9710
Pages:81
Published On:November 2025
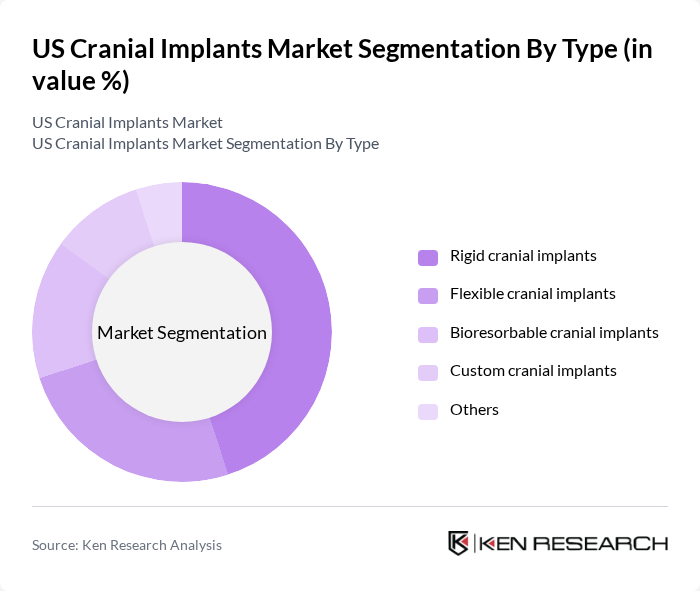
By Type:The cranial implants market is segmented into rigid cranial implants, flexible cranial implants, bioresorbable cranial implants, custom cranial implants, and others. Rigid cranial implants, typically made from titanium or PEEK, are the most widely used due to their strength, durability, and suitability for a range of neurosurgical applications. Flexible cranial implants, often constructed from polymeric materials, are increasingly adopted in cosmetic and reconstructive procedures for their adaptability to patient anatomy. Bioresorbable implants are gaining traction as they gradually dissolve in the body, reducing the need for secondary surgeries and minimizing long-term complications. Custom cranial implants, enabled by 3D printing and advanced imaging, are preferred for complex cranial defects requiring precise anatomical fit .
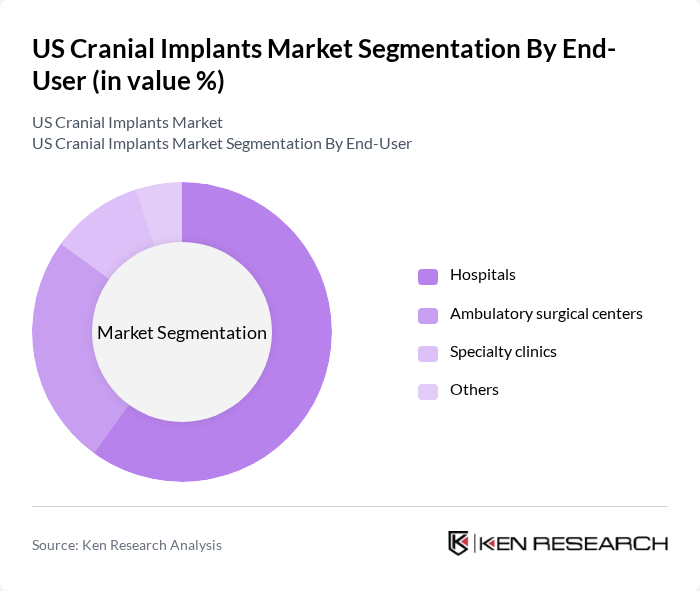
By End-User:The end-user segmentation includes hospitals, ambulatory surgical centers, specialty clinics, and others. Hospitals represent the primary end-users of cranial implants, accounting for the majority of procedures due to their comprehensive neurosurgical facilities and multidisciplinary care teams. Ambulatory surgical centers are experiencing increased adoption of cranial implants, reflecting the shift toward minimally invasive and outpatient neurosurgical procedures. Specialty clinics, particularly those focused on cosmetic and reconstructive surgery, are emerging as important end-users, leveraging advanced implant technologies for personalized patient care .
The US Cranial Implants Market is characterized by a dynamic mix of regional and international players. Leading participants such as Medtronic, Stryker Corporation, Johnson & Johnson (DePuy Synthes), Zimmer Biomet, Integra LifeSciences, B. Braun Melsungen AG, KLS Martin Group, NuVasive, Aesculap, Inc. (B. Braun), Boston Scientific, Amedica Corporation, Medline Industries, Orthofix Medical Inc., Acumed LLC, and Biometrix contribute to innovation, geographic expansion, and service delivery in this space.
The future of the U.S. cranial implants market appears promising, driven by ongoing advancements in surgical techniques and materials. As the healthcare infrastructure expands, particularly in underserved areas, access to cranial implant procedures is expected to improve. Additionally, the integration of digital technologies, such as 3D printing, will likely enhance customization and reduce production costs, further stimulating market growth. The focus on patient-specific solutions will also drive innovation and improve surgical outcomes in the coming years.
| Segment | Sub-Segments |
|---|---|
| By Type | Rigid cranial implants Flexible cranial implants Bioresorbable cranial implants Custom cranial implants Others |
| By End-User | Hospitals Ambulatory surgical centers Specialty clinics Others |
| By Material | Titanium Polyether ether ketone (PEEK) Polymethyl methacrylate (PMMA) Others |
| By Application | Neurosurgery Trauma surgery Cosmetic surgery Others |
| By Distribution Channel | Direct sales Distributors Online sales Others |
| By Region | Northeast Midwest South West |
| By Patient Demographics | Pediatric patients Adult patients Geriatric patients Others |
| Scope Item/Segment | Sample Size | Target Respondent Profiles |
|---|---|---|
| Neurosurgeons | 60 | Surgeons specializing in cranial procedures |
| Hospital Procurement Managers | 50 | Decision-makers in hospital supply chains |
| Patients with Cranial Implants | 40 | Individuals who have undergone cranial implant surgeries |
| Medical Device Distributors | 40 | Professionals involved in the distribution of cranial implants |
| Healthcare Policy Experts | 40 | Advisors on healthcare regulations and reimbursement policies |
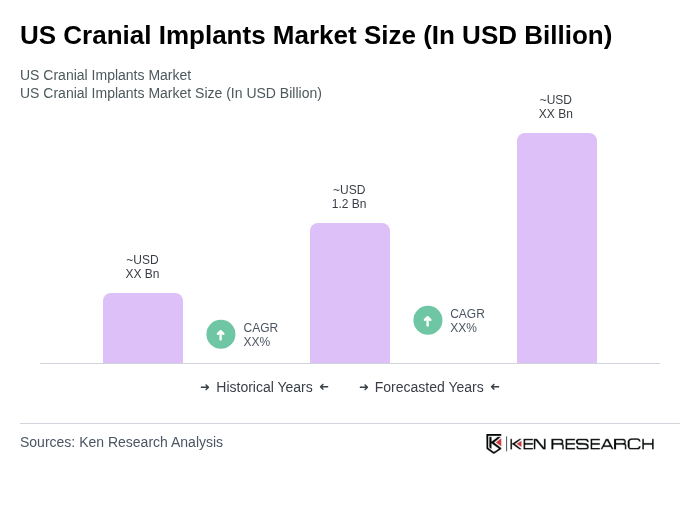
The US Cranial Implants Market is valued at approximately USD 1.2 billion, driven by factors such as the increasing prevalence of traumatic brain injuries and advancements in implant materials, including patient-specific 3D-printed options.