Region:Asia
Author(s):Dev
Product Code:KRAA2494
Pages:81
Published On:January 2026
By Type:
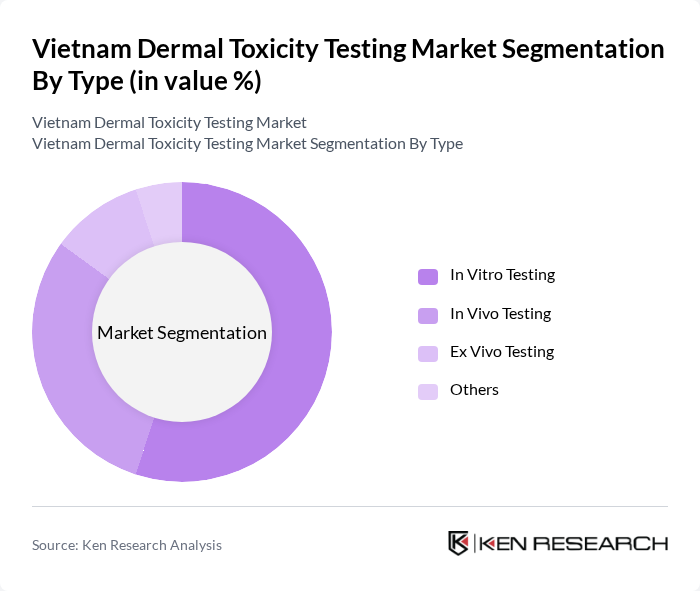
The market is segmented into four types: In Vitro Testing, In Vivo Testing, Ex Vivo Testing, and Others. Among these, In Vitro Testing is the leading sub-segment due to its cost-effectiveness and ethical advantages over animal testing, including reconstructed human epidermis models validated by OECD guidelines. The increasing adoption of alternative testing methods, driven by regulatory pressures and consumer preferences for cruelty-free products, has further solidified its dominance. In Vivo Testing, while still significant, is gradually being overshadowed by advancements in In Vitro methodologies. Ex Vivo Testing and Others contribute to niche applications but do not hold substantial market shares.
By End-User:
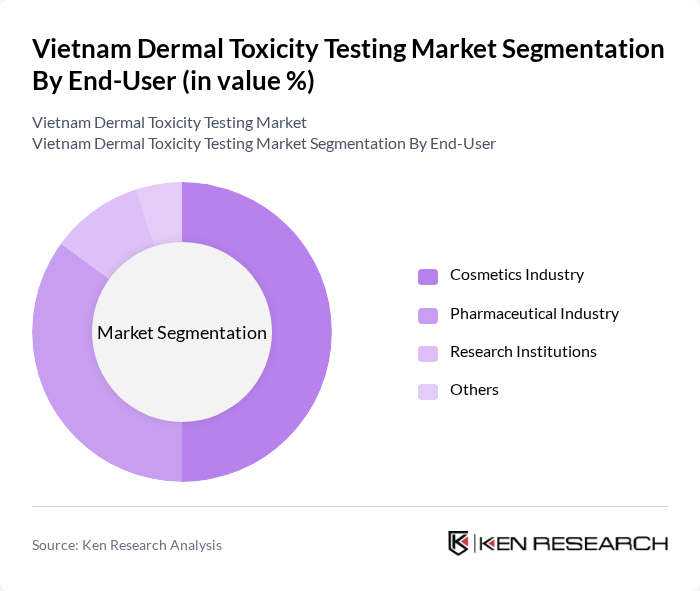
The end-user segmentation includes the Cosmetics Industry, Pharmaceutical Industry, Research Institutions, and Others. The Cosmetics Industry is the dominant segment, driven by stringent regulations and a growing consumer base that demands safe and effective products. The Pharmaceutical Industry follows closely, as drug safety assessments are critical for regulatory approvals. Research Institutions play a vital role in advancing testing methodologies, while the Others category includes various sectors that utilize dermal toxicity testing but contribute less significantly to overall market revenue.
The Vietnam Dermal Toxicity Testing Market is characterized by a dynamic mix of regional and international players. Leading participants such as Eurofins Scientific, SGS S.A., Intertek Group plc, Charles River Laboratories, Covance Inc., BioReliance, Toxikon Corporation, Wuxi AppTec, Medpace, Labcorp Drug Development, Q2 Solutions, Envigo, AccuBioTech, Atox Bio, ToxServices LLC contribute to innovation, geographic expansion, and service delivery in this space.
The Vietnam dermal toxicity testing market is poised for significant transformation, driven by evolving consumer preferences and regulatory landscapes. As the demand for safe and effective cosmetic and pharmaceutical products continues to rise, companies are likely to adopt innovative testing methods, including in vitro techniques. Furthermore, collaboration with international testing laboratories will enhance local capabilities, ensuring compliance with global standards. This dynamic environment presents opportunities for growth, particularly in the realm of non-animal testing methods and advanced technological integration.
| Segment | Sub-Segments |
|---|---|
| By Type | In Vitro Testing In Vivo Testing Ex Vivo Testing Others |
| By End-User | Cosmetics Industry Pharmaceutical Industry Research Institutions Others |
| By Application | Skin Irritation Testing Skin Sensitization Testing Phototoxicity Testing Others |
| By Methodology | Cell Culture Techniques Organ-on-a-Chip Technologies Computational Models Others |
| By Region | Northern Vietnam Southern Vietnam Central Vietnam |
| By Regulatory Compliance | GLP Compliance ISO Standards Others |
| By Market Maturity | Emerging Market Established Market Others |
| Scope Item/Segment | Sample Size | Target Respondent Profiles |
|---|---|---|
| Pharmaceutical Testing Facilities | 100 | Laboratory Directors, Quality Assurance Managers |
| Cosmetic Product Testing Labs | 80 | Regulatory Affairs Specialists, R&D Managers |
| Industrial Chemical Testing Centers | 70 | Compliance Officers, Toxicology Experts |
| Academic Research Institutions | 60 | Research Scientists, Professors in Toxicology |
| Government Regulatory Bodies | 50 | Policy Makers, Health Inspectors |
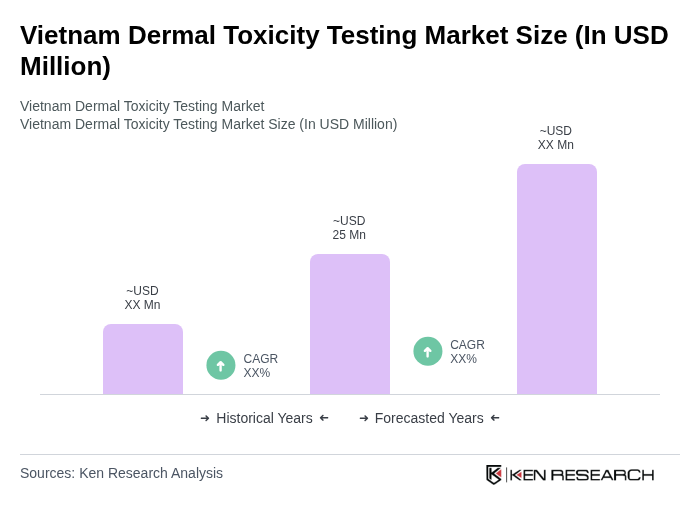
The Vietnam Dermal Toxicity Testing Market is valued at approximately USD 25 million, reflecting a five-year historical analysis. This growth is driven by increasing demand for safety assessments in the cosmetics and pharmaceutical sectors.