Region:Asia
Author(s):Dev
Product Code:KRAD1651
Pages:84
Published On:November 2025
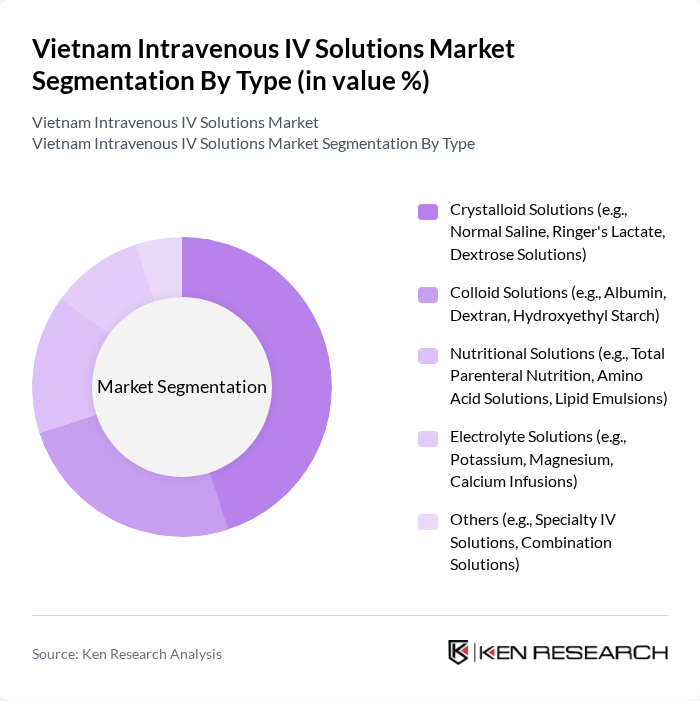
By Type:The market is segmented into various types of intravenous solutions, including Crystalloid Solutions, Colloid Solutions, Nutritional Solutions, Electrolyte Solutions, and Others. Among these, Crystalloid Solutions, which include Normal Saline and Ringer's Lactate, dominate the market due to their widespread use in hydration and electrolyte balance during medical treatments. The increasing number of surgical procedures and critical care admissions has further propelled the demand for these solutions.
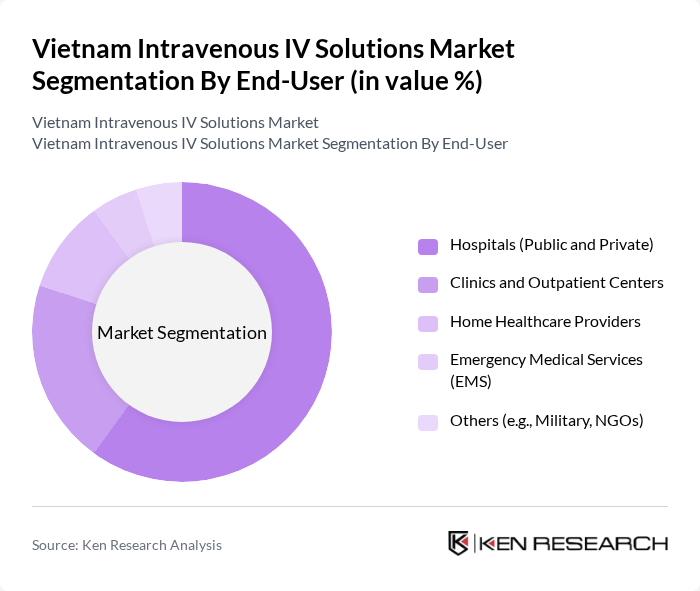
By End-User:The end-user segmentation includes Hospitals (Public and Private), Clinics and Outpatient Centers, Home Healthcare Providers, Emergency Medical Services (EMS), and Others. Hospitals, particularly public ones, are the leading end-users of intravenous solutions due to their extensive patient care requirements. The increasing number of patients requiring IV therapy in hospitals, especially in critical care units, has significantly contributed to the growth of this segment.
The Vietnam Intravenous IV Solutions Market is characterized by a dynamic mix of regional and international players. Leading participants such as Fresenius Kabi AG, Baxter International Inc., B. Braun Melsungen AG, Otsuka Pharmaceutical Co., Ltd., JW Life Science (JW Pharmaceutical Corporation), Grifols, S.A., Vimedimex Medi-Pharma JSC, Imexpharm Pharmaceutical JSC, Bidiphar Pharmaceutical JSC (Binh Dinh Pharmaceutical and Medical Equipment JSC), Traphaco JSC, Hau Giang Pharmaceutical JSC (DHG Pharma), Mekophar Chemical Pharmaceutical JSC, Domesco Medical Import-Export JSC, Central Pharmaceutical Joint Stock Company No. 1 (CPC1), SPM Pharmaceutical JSC contribute to innovation, geographic expansion, and service delivery in this space.
The future of the intravenous solutions market in Vietnam appears promising, driven by increasing healthcare investments and a growing focus on patient-centered care. As the government continues to enhance healthcare infrastructure, the demand for innovative IV solutions is expected to rise. Additionally, the integration of digital health technologies will likely streamline operations and improve patient outcomes, further propelling market growth. The emphasis on personalized medicine will also shape product development, catering to specific patient needs and preferences.
| Segment | Sub-Segments |
|---|---|
| By Type | Crystalloid Solutions (e.g., Normal Saline, Ringer's Lactate, Dextrose Solutions) Colloid Solutions (e.g., Albumin, Dextran, Hydroxyethyl Starch) Nutritional Solutions (e.g., Total Parenteral Nutrition, Amino Acid Solutions, Lipid Emulsions) Electrolyte Solutions (e.g., Potassium, Magnesium, Calcium Infusions) Others (e.g., Specialty IV Solutions, Combination Solutions) |
| By End-User | Hospitals (Public and Private) Clinics and Outpatient Centers Home Healthcare Providers Emergency Medical Services (EMS) Others (e.g., Military, NGOs) |
| By Distribution Channel | Direct Sales (to Hospitals/Institutions) Distributors/Wholesalers Online B2B Sales Retail Pharmacies Others (e.g., Government Tenders) |
| By Packaging Type | Bottles (Glass/Plastic) Flexible Bags (PVC/Non-PVC) Vials and Ampoules Others (e.g., Prefilled Syringes) |
| By Application | Surgical Procedures Critical Care and Intensive Care Hydration and Fluid Replacement Therapy Drug Delivery (IV Antibiotics, Chemotherapy, etc.) Others (e.g., Nutritional Support, Emergency Resuscitation) |
| By Region | Northern Vietnam Southern Vietnam Central Vietnam Others |
| By Policy Support | Government Subsidies Tax Incentives Regulatory Support Others |
| Scope Item/Segment | Sample Size | Target Respondent Profiles |
|---|---|---|
| Hospital Procurement Departments | 60 | Procurement Managers, Supply Chain Coordinators |
| Pharmacy Operations | 40 | Pharmacy Directors, Clinical Pharmacists |
| Healthcare Providers | 50 | Doctors, Nurses, Healthcare Administrators |
| Manufacturers of IV Solutions | 40 | Product Managers, Sales Executives |
| Regulatory Bodies | 40 | Regulatory Affairs Specialists, Compliance Officers |
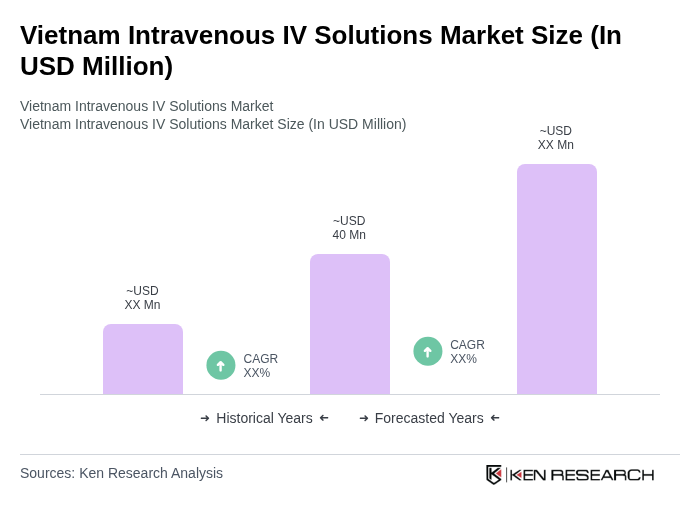
The Vietnam Intravenous IV Solutions Market is valued at approximately USD 40 million, reflecting a significant growth driven by factors such as the increasing prevalence of chronic diseases and advancements in healthcare infrastructure.