Region:Asia
Author(s):Shubham
Product Code:KRAD2452
Pages:86
Published On:January 2026
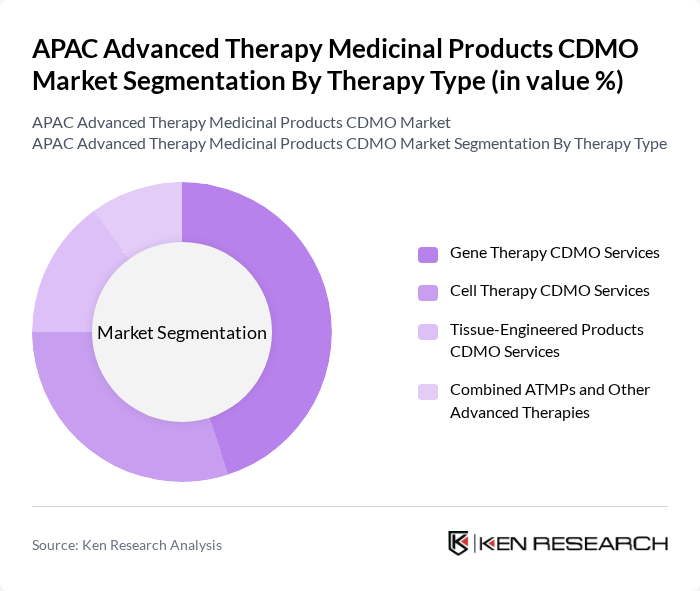
By Therapy Type:The market is segmented into four key therapy types: Gene Therapy CDMO Services, Cell Therapy CDMO Services, Tissue-Engineered Products CDMO Services, and Combined ATMPs and Other Advanced Therapies. Gene Therapy CDMO Services is currently a leading segment globally and is gaining strong traction in APAC, driven by the increasing number of viral-vector and non-viral gene-based therapies entering clinical trials and the growing demand for precision and personalized medicine. Advancements in gene editing technologies, such as CRISPR and other targeted editing platforms, along with rising oncology and rare disease pipelines, have contributed to the expansion of this segment and to higher outsourcing of complex vector development and manufacturing to specialized CDMOs in the region.
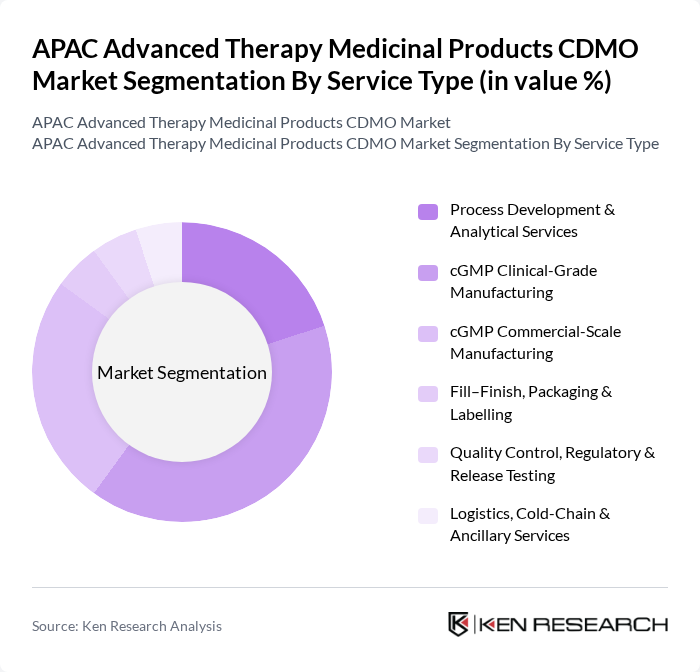
By Service Type:The service type segmentation includes Process Development & Analytical Services, cGMP Clinical-Grade Manufacturing, cGMP Commercial-Scale Manufacturing, Fill–Finish, Packaging & Labelling, Quality Control, Regulatory & Release Testing, and Logistics, Cold-Chain & Ancillary Services. The cGMP Clinical-Grade Manufacturing segment is currently the most significant, as sponsors increasingly outsource early- and mid-stage GMP production for cell and gene therapies to access specialized facilities, single-use bioprocessing platforms, and experienced technical teams that can meet stringent regulatory standards for safety and efficacy, which is critical for clinical progression and market approval. In parallel, demand is rising for integrated process development, analytical characterization, and cold-chain logistics services to support complex ATMP supply chains and minimize time-to-clinic in the APAC region.
The APAC Advanced Therapy Medicinal Products CDMO Market is characterized by a dynamic mix of regional and international players. Leading participants such as Lonza Group AG, WuXi Advanced Therapies (WuXi AppTec Group), Catalent, Inc., Samsung Biologics Co., Ltd., FUJIFILM Diosynth Biotechnologies, AGC Biologics, Minaris Regenerative Medicine, Celonic Group, Rentschler Biopharma SE, GenScript Biotech Corporation, Charles River Laboratories International, Inc., Thermo Fisher Scientific (Patheon), Novartis AG (Cell & Gene Therapies Manufacturing), Bio Elpida, and other emerging APAC-focused ATMP CDMOs contribute to innovation, geographic expansion, and service delivery in this space.
The future of the APAC Advanced Therapy Medicinal Products CDMO market appears promising, driven by ongoing innovations and increasing collaborations between industry players and research institutions. As the demand for personalized medicine continues to rise, CDMOs are expected to enhance their capabilities to meet evolving market needs. Additionally, the integration of automation and AI technologies in manufacturing processes will likely streamline operations, reduce costs, and improve product quality, positioning the market for sustained growth in the coming years.
| Segment | Sub-Segments |
|---|---|
| By Therapy Type | Gene Therapy CDMO Services Cell Therapy CDMO Services Tissue-Engineered Products CDMO Services Combined ATMPs and Other Advanced Therapies |
| By Service Type | Process Development & Analytical Services cGMP Clinical-Grade Manufacturing cGMP Commercial-Scale Manufacturing Fill–Finish, Packaging & Labelling Quality Control, Regulatory & Release Testing Logistics, Cold-Chain & Ancillary Services |
| By Development Phase | Pre-clinical & Early Development Phase I Phase II Phase III Phase IV / Post-marketing |
| By Indication Area | Oncology Cardiology Neurology & CNS Disorders Musculoskeletal & Orthopedic Infectious Diseases Immunology, Inflammation & Autoimmune Endocrine, Metabolic & Genetic Disorders Other Indications |
| By Manufacturing Modality | Autologous Therapies Allogeneic Therapies Hybrid / Other Manufacturing Approaches |
| By Technology Platform | Viral Vector Platforms (AAV, Lentiviral, Retroviral, Others) Non-viral Vector & Gene Editing Platforms (mRNA, Plasmid DNA, CRISPR, Others) Cell Processing & Expansion Platforms Other Enabling Technologies |
| By Country / Sub-region (APAC) | China Japan South Korea India Australia & New Zealand Southeast Asia (incl. Singapore, Malaysia, Thailand, Indonesia, Vietnam, Others) Rest of APAC |
| Scope Item/Segment | Sample Size | Target Respondent Profiles |
|---|---|---|
| Gene Therapy Development | 95 | R&D Directors, Clinical Trial Managers |
| Cell Therapy Manufacturing | 85 | Production Managers, Quality Assurance Officers |
| Tissue Engineering Applications | 75 | Biomedical Engineers, Regulatory Affairs Specialists |
| Regulatory Compliance in ATMP | 65 | Compliance Officers, Legal Advisors |
| Market Access Strategies for ATMP | 105 | Market Access Managers, Health Economists |
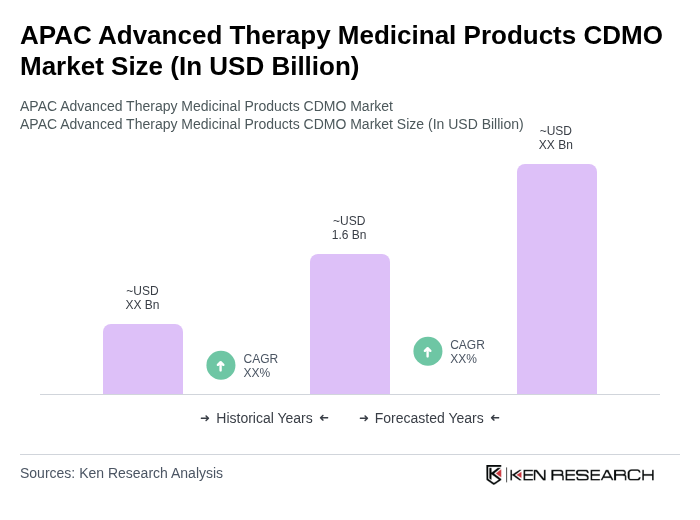
The APAC Advanced Therapy Medicinal Products CDMO Market is valued at approximately USD 1.6 billion, reflecting its significant growth compared to North America and Europe, driven by increasing demand for personalized medicine and advancements in cell and gene therapy.