About the Report
Base Year 2024Australia Recombinant Protein Manufacturing Services Market Overview
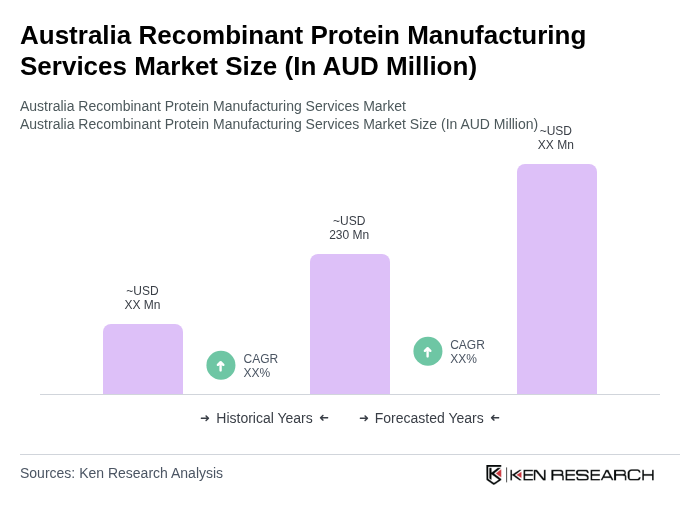
- The Australia Recombinant Protein Manufacturing Services Market is valued at AUD 230 million, based on a five-year historical analysis of the recombinant protein manufacturing services and therapeutic CDMO segments in the country. This growth is primarily driven by the increasing demand for biopharmaceuticals, expanding use of recombinant proteins in therapeutics and vaccines, rising R&D investments in biologics, and the growing burden of chronic and rare diseases that require advanced protein-based therapies.
- Key clusters in this market include major cities such as Sydney and Melbourne, which dominate due to their robust healthcare infrastructure, concentration of leading biotechnology and pharmaceutical firms, strong research universities, and co?location of medical research institutes and clinical trial sites. These cities foster innovation and collaboration across academia, industry, and healthcare providers, making them attractive hubs for recombinant protein development and manufacturing.
- In 2023, the Australian government continued to enforce the Therapeutic Goods (Manufacturing Principles) Determination 2020 issued by the Therapeutic Goods Administration (TGA), which adopts the PIC/S Guide to Good Manufacturing Practice for Medicinal Products and sets binding GMP requirements for biologics, including recombinant protein products. These regulations emphasize stringent quality control, validation, documentation, and compliance for recombinant protein manufacturing to ensure the safety, quality, and efficacy of biopharmaceuticals, thereby enhancing stakeholder confidence and supporting market growth.
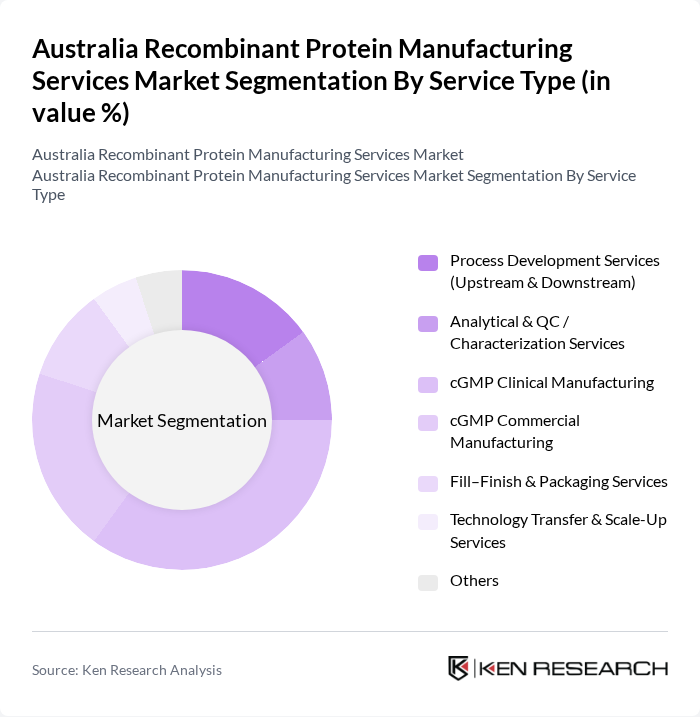
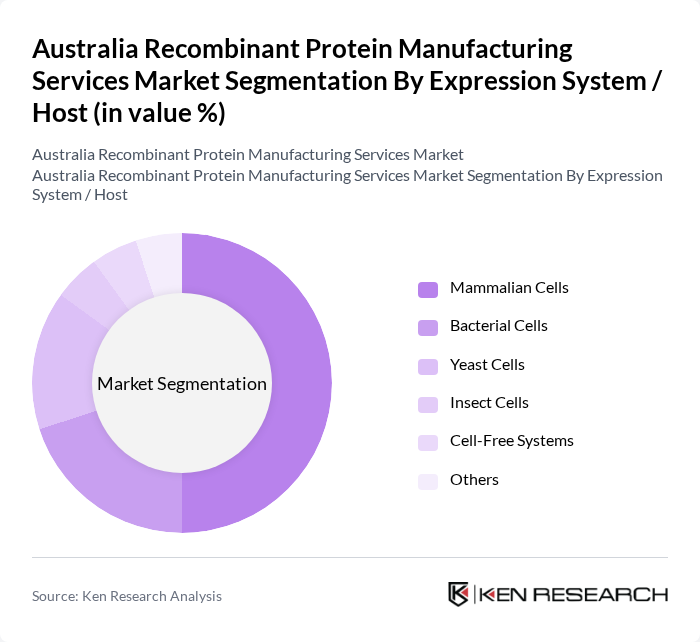
Australia Recombinant Protein Manufacturing Services Market Segmentation
By Service Type:The service type segmentation includes various offerings that support the end?to?end recombinant protein manufacturing workflow, from early process development to commercial supply. The dominant sub-segment is cGMP Clinical Manufacturing, which is crucial for producing clinical-grade proteins that meet TGA and international regulatory standards for use in human trials. This segment is driven by the increasing number of biologics and biosimilar clinical trials in Australia, greater outsourcing to CDMOs, and the demand for high-quality therapeutic proteins with robust comparability and documentation across trial phases.
By Expression System / Host:This segmentation focuses on the various expression systems used in recombinant protein production. The mammalian cells sub-segment is leading due to their ability to produce complex, human-like glycosylated proteins with appropriate post-translational modifications, which are essential for the safety and therapeutic efficacy of monoclonal antibodies, fusion proteins, and other biologics. The increasing preference for biologics and biosimilars over traditional small-molecule drugs, coupled with the expansion of antibody and recombinant enzyme pipelines, further drives the adoption of mammalian expression systems in Australia.
Australia Recombinant Protein Manufacturing Services Market Competitive Landscape
The Australia Recombinant Protein Manufacturing Services Market is characterized by a dynamic mix of regional and international players. Leading participants such as CSL Limited, Thermo Fisher Scientific, Merck KGaA (MilliporeSigma), Cytiva, Lonza Group, WuXi Biologics, Syngene International, Samsung Biologics, Catalent, Fujifilm Diosynth Biotechnologies, Patheon by Thermo Fisher, AGC Biologics, AbbVie Contract Manufacturing, Baxter BioPharma Solutions, and local and regional CDMOs in Australia contribute to innovation, geographic expansion, and service delivery in this space.
Australia Recombinant Protein Manufacturing Services Market Industry Analysis
Growth Drivers
- Increasing Demand for Biopharmaceuticals:The Australian biopharmaceutical market is projected to reach AUD 12 billion in the future, driven by a growing population and increased healthcare expenditure. The demand for innovative therapies, particularly monoclonal antibodies and vaccines, is surging, with the Therapeutic Goods Administration (TGA) approving over 60 new biopharmaceuticals in the future alone. This trend is expected to continue, propelling the recombinant protein manufacturing sector as companies seek to meet this rising demand.
- Advancements in Recombinant DNA Technology:The recombinant DNA technology landscape in Australia is evolving rapidly, with investments exceeding AUD 2 billion in the future. This includes funding for research and development initiatives aimed at enhancing production efficiency and product quality. The introduction of novel expression systems and purification techniques is expected to streamline processes, reduce time-to-market, and improve yield, thereby fostering growth in the recombinant protein manufacturing services market.
- Rising Investment in Biotechnology Research:Australia’s biotechnology sector attracted over AUD 3 billion in venture capital funding in the future, reflecting a robust interest in biopharmaceutical innovations. Government initiatives, such as the Biomedical Translation Fund, are further bolstering research efforts. This influx of capital is facilitating the development of new recombinant proteins, enhancing the capabilities of local manufacturers, and positioning Australia as a leader in biopharmaceutical production.
Market Challenges
- High Production Costs:The cost of producing recombinant proteins in Australia can exceed AUD 600 per gram, significantly impacting profitability. Factors contributing to these high costs include expensive raw materials, stringent quality control measures, and the need for advanced manufacturing facilities. As a result, many companies face challenges in maintaining competitive pricing while ensuring product quality, which can hinder market growth.
- Regulatory Hurdles:The regulatory landscape for recombinant protein manufacturing in Australia is complex, with compliance costs estimated at AUD 250 million annually for mid-sized firms. The Therapeutic Goods Administration (TGA) enforces rigorous guidelines that can delay product approvals and increase operational costs. Navigating these regulations requires significant resources, which can be a barrier for smaller companies looking to enter the market.
Australia Recombinant Protein Manufacturing Services Market Future Outlook
The future of the recombinant protein manufacturing services market in Australia appears promising, driven by technological advancements and increasing collaboration between industry and academia. The integration of automation and artificial intelligence in production processes is expected to enhance efficiency and reduce costs. Additionally, the growing focus on personalized medicine will likely create new avenues for innovation, allowing manufacturers to tailor products to specific patient needs, thereby expanding market potential.
Market Opportunities
- Expansion of Contract Manufacturing Organizations (CMOs):The rise of CMOs in Australia presents significant opportunities for recombinant protein manufacturers. With over 40 CMOs operating in the region, companies can leverage these partnerships to scale production without incurring heavy capital expenditures. This trend is expected to facilitate faster market entry for new biopharmaceuticals, enhancing overall industry growth.
- Development of Personalized Medicine:The shift towards personalized medicine is creating substantial opportunities for recombinant protein manufacturers. With the Australian government investing AUD 600 million in genomics research, companies can develop tailored therapies that meet individual patient needs. This focus on customization is anticipated to drive demand for specific recombinant proteins, fostering innovation and growth in the sector.
Scope of the Report
| Segment | Sub-Segments |
|---|---|
| By Service Type | Process Development Services (Upstream & Downstream) Analytical & QC / Characterization Services cGMP Clinical Manufacturing cGMP Commercial Manufacturing Fill–Finish & Packaging Services Technology Transfer & Scale-Up Services Others |
| By Expression System / Host | Mammalian Cells Bacterial Cells Yeast Cells Insect Cells Cell-Free Systems Others |
| By Molecule / Protein Type | Therapeutic Proteins (e.g., Cytokines, Growth Factors, Hormones) Monoclonal and Other Antibodies Enzymes Vaccine Antigens Research-Grade Recombinant Proteins Others |
| By Stage of Development | Pre?clinical Clinical (Phase I–III) Commercial |
| By Scale of Operation | Small / Pilot Scale Clinical Scale Large / Commercial Scale |
| By End-User | Pharmaceutical Companies Biotechnology Firms Academic and Research Institutions Contract Research Organizations (CROs) Contract Development & Manufacturing Organizations (CDMOs) Others |
| By Region | New South Wales Victoria Queensland Western Australia South Australia Others |
Key Target Audience
Investors and Venture Capitalist Firms
Government and Regulatory Bodies (e.g., Therapeutic Goods Administration, Department of Health)
Biopharmaceutical Companies
Contract Manufacturing Organizations (CMOs)
Healthcare Providers and Hospitals
Biotechnology Firms
Pharmaceutical Supply Chain Managers
Industry Associations (e.g., AusBiotech)
Players Mentioned in the Report:
CSL Limited
Thermo Fisher Scientific
Merck KGaA (MilliporeSigma)
Cytiva
Lonza Group
WuXi Biologics
Syngene International
Samsung Biologics
Catalent
Fujifilm Diosynth Biotechnologies
Patheon by Thermo Fisher
AGC Biologics
AbbVie Contract Manufacturing
Baxter BioPharma Solutions
Local and Regional CDMOs in Australia
Table of Contents
Market Assessment Phase
1. Executive Summary and Approach
2. Australia Recombinant Protein Manufacturing Services Market Overview
2.1 Key Insights and Strategic Recommendations
2.2 Australia Recombinant Protein Manufacturing Services Market Overview
2.3 Definition and Scope
2.4 Evolution of Market Ecosystem
2.5 Timeline of Key Regulatory Milestones
2.6 Value Chain & Stakeholder Mapping
2.7 Business Cycle Analysis
2.8 Policy & Incentive Landscape
3. Australia Recombinant Protein Manufacturing Services Market Analysis
3.1 Growth Drivers
3.1.1 Increasing demand for biopharmaceuticals
3.1.2 Advancements in recombinant DNA technology
3.1.3 Rising investment in biotechnology research
3.1.4 Growing prevalence of chronic diseases
3.2 Market Challenges
3.2.1 High production costs
3.2.2 Regulatory hurdles
3.2.3 Limited skilled workforce
3.2.4 Competition from alternative therapies
3.3 Market Opportunities
3.3.1 Expansion of contract manufacturing organizations (CMOs)
3.3.2 Collaborations with research institutions
3.3.3 Development of personalized medicine
3.3.4 Increasing focus on sustainable manufacturing practices
3.4 Market Trends
3.4.1 Shift towards automation in manufacturing
3.4.2 Growth of biosimilars
3.4.3 Integration of artificial intelligence in production
3.4.4 Emphasis on quality by design (QbD) principles
3.5 Government Regulation
3.5.1 Therapeutic Goods Administration (TGA) guidelines
3.5.2 National Health and Medical Research Council (NHMRC) regulations
3.5.3 Biosecurity Act compliance
3.5.4 Intellectual property protection laws
4. SWOT Analysis
5. Stakeholder Analysis
6. Porter's Five Forces Analysis
7. Australia Recombinant Protein Manufacturing Services Market Size, 2019-2024
7.1 By Value
7.2 By Volume
7.3 By Average Selling Price
8. Australia Recombinant Protein Manufacturing Services Market Segmentation
8.1 By Service Type
8.1.1 Process Development Services (Upstream & Downstream)
8.1.2 Analytical & QC / Characterization Services
8.1.3 cGMP Clinical Manufacturing
8.1.4 cGMP Commercial Manufacturing
8.1.5 Fill–Finish & Packaging Services
8.1.6 Technology Transfer & Scale-Up Services
8.1.7 Others
8.2 By Expression System / Host
8.2.1 Mammalian Cells
8.2.2 Bacterial Cells
8.2.3 Yeast Cells
8.2.4 Insect Cells
8.2.5 Cell-Free Systems
8.2.6 Others
8.3 By Molecule / Protein Type
8.3.1 Therapeutic Proteins (e.g., Cytokines, Growth Factors, Hormones)
8.3.2 Monoclonal and Other Antibodies
8.3.3 Enzymes
8.3.4 Vaccine Antigens
8.3.5 Research-Grade Recombinant Proteins
8.3.6 Others
8.4 By Stage of Development
8.4.1 Pre?clinical
8.4.2 Clinical (Phase I–III)
8.4.3 Commercial
8.5 By Scale of Operation
8.5.1 Small / Pilot Scale
8.5.2 Clinical Scale
8.5.3 Large / Commercial Scale
8.6 By End-User
8.6.1 Pharmaceutical Companies
8.6.2 Biotechnology Firms
8.6.3 Academic and Research Institutions
8.6.4 Contract Research Organizations (CROs)
8.6.5 Contract Development & Manufacturing Organizations (CDMOs)
8.6.6 Others
8.7 By Region
8.7.1 New South Wales
8.7.2 Victoria
8.7.3 Queensland
8.7.4 Western Australia
8.7.5 South Australia
8.7.6 Others
9. Australia Recombinant Protein Manufacturing Services Market Competitive Analysis
9.1 Market Share of Key Players
9.2 Cross Comparison of Key Players
9.2.1 Company Name
9.2.2 Ownership & Group Size (Global, Regional, Local)
9.2.3 Australia Recombinant Protein Manufacturing Services Revenue (AUD, Latest Year)
9.2.4 3?Year CAGR in Services Revenue
9.2.5 EBITDA Margin from Manufacturing Services
9.2.6 Capacity Utilization Rate (%)
9.2.7 Installed Bioreactor Capacity (L, Clinical & Commercial)
9.2.8 Number of Active Clients in Australia
9.2.9 Average Contract Value (ACV) / Deal Size
9.2.10 Win Rate in Competitive Tenders (%)
9.2.11 On?Time In?Full (OTIF) Delivery Rate
9.2.12 Batch Success / Release Rate (%)
9.2.13 R&D Intensity (% of Revenue Spent on R&D)
9.2.14 New Technology / Platform Launches (Annual Count)
9.2.15 Quality & Compliance Track Record (Audit Observations, Recalls)
9.3 SWOT Analysis of Top Players
9.4 Pricing Analysis
9.5 Detailed Profile of Major Companies
9.5.1 CSL Limited
9.5.2 Thermo Fisher Scientific
9.5.3 Merck KGaA (MilliporeSigma)
9.5.4 Cytiva
9.5.5 Lonza Group
9.5.6 WuXi Biologics
9.5.7 Syngene International
9.5.8 Samsung Biologics
9.5.9 Catalent
9.5.10 Fujifilm Diosynth Biotechnologies
9.5.11 Patheon by Thermo Fisher
9.5.12 AGC Biologics
9.5.13 AbbVie Contract Manufacturing
9.5.14 Baxter BioPharma Solutions
9.5.15 Local and Regional CDMOs in Australia
10. Australia Recombinant Protein Manufacturing Services Market End-User Analysis
10.1 Procurement Behavior of Key Ministries
10.1.1 Budget Allocation Trends
10.1.2 Decision-Making Processes
10.1.3 Supplier Selection Criteria
10.1.4 Contract Management Practices
10.2 Corporate Spend on Infrastructure & Energy
10.2.1 Investment Trends in Biotechnology
10.2.2 Infrastructure Development Initiatives
10.2.3 Energy Efficiency Programs
10.2.4 Corporate Social Responsibility (CSR) Spending
10.3 Pain Point Analysis by End-User Category
10.3.1 Cost Management Issues
10.3.2 Quality Control Challenges
10.3.3 Regulatory Compliance Difficulties
10.3.4 Supply Chain Disruptions
10.4 User Readiness for Adoption
10.4.1 Technology Adoption Rates
10.4.2 Training and Support Needs
10.4.3 Infrastructure Readiness
10.4.4 User Feedback Mechanisms
10.5 Post-Deployment ROI and Use Case Expansion
10.5.1 Performance Metrics Tracking
10.5.2 Case Studies of Successful Implementations
10.5.3 Future Use Case Identification
10.5.4 Long-term Value Assessment
11. Australia Recombinant Protein Manufacturing Services Market Future Size, 2025-2030
11.1 By Value
11.2 By Volume
11.3 By Average Selling Price
Go-To-Market Strategy Phase
1. Whitespace Analysis + Business Model Canvas
1.1 Market Gaps Identification
1.2 Value Proposition Development
1.3 Revenue Streams Analysis
1.4 Cost Structure Evaluation
1.5 Key Partnerships Exploration
1.6 Customer Segmentation
1.7 Channels of Distribution
2. Marketing and Positioning Recommendations
2.1 Branding Strategies
2.2 Product USPs
2.3 Target Market Identification
2.4 Communication Strategies
2.5 Digital Marketing Approaches
2.6 Customer Engagement Tactics
2.7 Performance Metrics
3. Distribution Plan
3.1 Urban Retail Strategies
3.2 Rural NGO Tie-ups
3.3 E-commerce Integration
3.4 Logistics and Supply Chain Management
3.5 Distribution Partnerships
3.6 Inventory Management
3.7 Performance Tracking
4. Channel & Pricing Gaps
4.1 Underserved Routes
4.2 Pricing Bands Analysis
4.3 Competitor Pricing Strategies
4.4 Customer Willingness to Pay
4.5 Value-Based Pricing Models
4.6 Discounting Strategies
4.7 Pricing Optimization Techniques
5. Unmet Demand & Latent Needs
5.1 Category Gaps Identification
5.2 Consumer Segments Analysis
5.3 Emerging Trends Exploration
5.4 Product Development Opportunities
5.5 Market Entry Strategies
5.6 Customer Feedback Mechanisms
5.7 Future Demand Forecasting
6. Customer Relationship
6.1 Loyalty Programs
6.2 After-sales Service
6.3 Customer Support Strategies
6.4 Feedback and Improvement Processes
6.5 Relationship Management Tools
6.6 Customer Retention Strategies
6.7 Performance Metrics
7. Value Proposition
7.1 Sustainability Initiatives
7.2 Integrated Supply Chains
7.3 Competitive Advantages
7.4 Customer-Centric Approaches
7.5 Innovation and R&D Focus
7.6 Market Differentiation Strategies
7.7 Performance Metrics
8. Key Activities
8.1 Regulatory Compliance
8.2 Branding Initiatives
8.3 Distribution Setup
8.4 Marketing Campaigns
8.5 Training and Development
8.6 Performance Monitoring
8.7 Continuous Improvement Processes
9. Entry Strategy Evaluation
9.1 Domestic Market Entry Strategy
9.1.1 Product Mix Considerations
9.1.2 Pricing Band Analysis
9.1.3 Packaging Strategies
9.2 Export Entry Strategy
9.2.1 Target Countries Identification
9.2.2 Compliance Roadmap Development
10. Entry Mode Assessment
10.1 Joint Ventures
10.2 Greenfield Investments
10.3 Mergers & Acquisitions
10.4 Distributor Model Evaluation
10.5 Risk Assessment
10.6 Strategic Fit Analysis
10.7 Performance Metrics
11. Capital and Timeline Estimation
11.1 Capital Requirements
11.2 Timelines for Implementation
11.3 Financial Projections
11.4 Funding Sources
11.5 Risk Management Strategies
11.6 Performance Metrics
12. Control vs Risk Trade-Off
12.1 Ownership vs Partnerships
12.2 Risk Mitigation Strategies
12.3 Control Mechanisms
12.4 Performance Metrics
13. Profitability Outlook
13.1 Breakeven Analysis
13.2 Long-term Sustainability
13.3 Financial Health Indicators
13.4 Performance Metrics
14. Potential Partner List
14.1 Distributors
14.2 Joint Ventures
14.3 Acquisition Targets
14.4 Strategic Alliances
14.5 Performance Metrics
15. Execution Roadmap
15.1 Phased Plan for Market Entry
15.1.1 Market Setup
15.1.2 Market Entry
15.1.3 Growth Acceleration
15.1.4 Scale & Stabilize
15.2 Key Activities and Milestones
15.2.1 Activity Planning
15.2.2 Milestone Tracking
15.2.3 Performance Metrics
Research Methodology
Phase 1: Approach1
Desk Research
- Analysis of industry reports from Australian biotechnology associations and government publications
- Review of scientific literature and patents related to recombinant protein technologies
- Examination of market trends and forecasts from reputable market research firms
Primary Research
- Interviews with executives from leading recombinant protein manufacturing companies
- Surveys targeting R&D managers in pharmaceutical and biotech firms
- Field interviews with regulatory affairs specialists to understand compliance challenges
Validation & Triangulation
- Cross-validation of findings through multiple data sources including trade publications and expert opinions
- Triangulation of market data with insights from industry conferences and seminars
- Sanity checks through feedback from a panel of industry experts and stakeholders
Phase 2: Market Size Estimation1
Top-down Assessment
- Estimation of market size based on national biotechnology spending and growth rates
- Segmentation by application areas such as therapeutics, diagnostics, and research
- Incorporation of government funding initiatives and grants for biotech research
Bottom-up Modeling
- Collection of production volume data from key recombinant protein manufacturers
- Operational cost analysis based on service pricing and production efficiency
- Estimation of market size using volume x average pricing for various protein products
Forecasting & Scenario Analysis
- Multi-variable regression analysis incorporating factors like healthcare expenditure and technological advancements
- Scenario modeling based on potential regulatory changes and market entry of new players
- Development of baseline, optimistic, and pessimistic forecasts through 2030
Phase 3: CATI Sample Composition1
| Scope Item/Segment | Sample Size | Target Respondent Profiles |
|---|---|---|
| Pharmaceutical Applications | 120 | Biotech Executives, Product Managers |
| Research Institutions | 90 | Research Scientists, Lab Managers |
| Diagnostics Sector | 80 | Clinical Lab Directors, Quality Assurance Managers |
| Regulatory Affairs | 60 | Regulatory Affairs Specialists, Compliance Officers |
| Contract Manufacturing Organizations (CMOs) | 100 | Operations Managers, Business Development Executives |
Frequently Asked Questions
What is the current value of the Australia Recombinant Protein Manufacturing Services Market?
The Australia Recombinant Protein Manufacturing Services Market is valued at approximately AUD 230 million, reflecting a robust growth trajectory driven by increasing demand for biopharmaceuticals and advancements in recombinant protein technologies.