Region:Middle East
Author(s):Rebecca
Product Code:KRAE0842
Pages:94
Published On:December 2025
By Type:The market is segmented into various types of drug-device combination products, including injectable, implantable, inhalable, transdermal, and others. Among these, injectable combination products dominate the market due to their widespread use in chronic disease management, particularly in diabetes and pain management. The convenience and effectiveness of these products have led to increased consumer adoption, making them a preferred choice for both healthcare providers and patients.
By End-User:The end-user segmentation includes hospitals, clinics, home healthcare, research institutions, and others. Hospitals are the leading end-users of drug-device combination products, driven by the increasing number of patients requiring advanced treatment options. The integration of these products into hospital settings enhances patient care and treatment outcomes, making them essential in modern healthcare delivery.
The GCC Drug Device Combination Products Market is characterized by a dynamic mix of regional and international players. Leading participants such as Medtronic, Abbott Laboratories, Boston Scientific, Johnson & Johnson, Becton, Dickinson and Company, Stryker Corporation, Siemens Healthineers, Philips Healthcare, Terumo Corporation, Fresenius Kabi, Cook Medical, Baxter International, 3M Health Care, and GE Healthcare contribute to innovation, geographic expansion, and service delivery in this space.
The future of the GCC drug-device combination products market appears promising, driven by ongoing technological innovations and a growing emphasis on patient-centric healthcare solutions. As healthcare providers increasingly adopt integrated approaches to treatment, the demand for combination products is expected to rise. Furthermore, the expansion of telemedicine and digital health solutions will likely enhance accessibility and efficiency in healthcare delivery, fostering a conducive environment for market growth and innovation in the coming years.
| Segment | Sub-Segments |
|---|---|
| By Type | Injectable Combination Products Implantable Combination Products Inhalable Combination Products Transdermal Combination Products Others |
| By End-User | Hospitals Clinics Home Healthcare Research Institutions Others |
| By Distribution Channel | Direct Sales Distributors Online Sales Retail Pharmacies Others |
| By Application | Diabetes Management Cardiovascular Treatment Cancer Treatment Pain Management Others |
| By Material | Polymer-based Products Metal-based Products Biodegradable Materials Others |
| By Region | Saudi Arabia UAE Qatar Kuwait Oman |
| By Regulatory Approval Status | Approved Products Products Under Review Products in Development Others |
| Scope Item/Segment | Sample Size | Target Respondent Profiles |
|---|---|---|
| Pharmaceutical Manufacturers | 100 | Product Managers, R&D Directors |
| Medical Device Companies | 80 | Regulatory Affairs Specialists, Marketing Managers |
| Healthcare Providers | 120 | Doctors, Pharmacists, Nurses |
| Patients Using Combination Products | 150 | Chronic Disease Patients, Caregivers |
| Regulatory Bodies | 50 | Policy Makers, Compliance Officers |
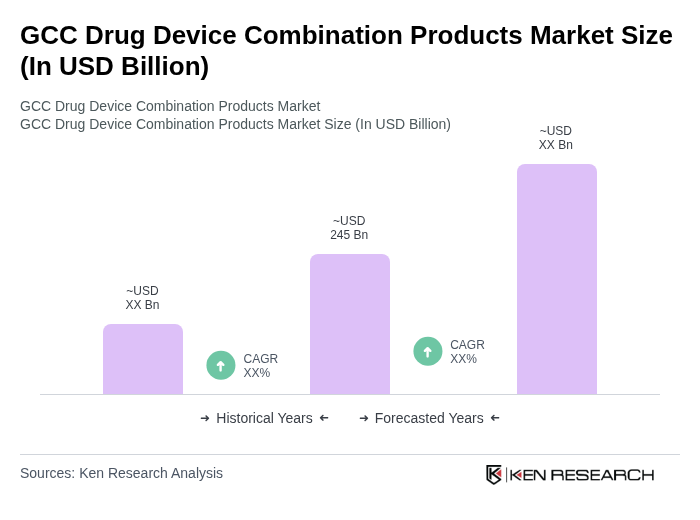
The GCC Drug Device Combination Products Market is valued at approximately USD 245 billion, reflecting significant growth driven by the rising prevalence of chronic diseases and advancements in drug delivery technologies.