Region:Middle East
Author(s):Rebecca
Product Code:KRAD1351
Pages:95
Published On:November 2025
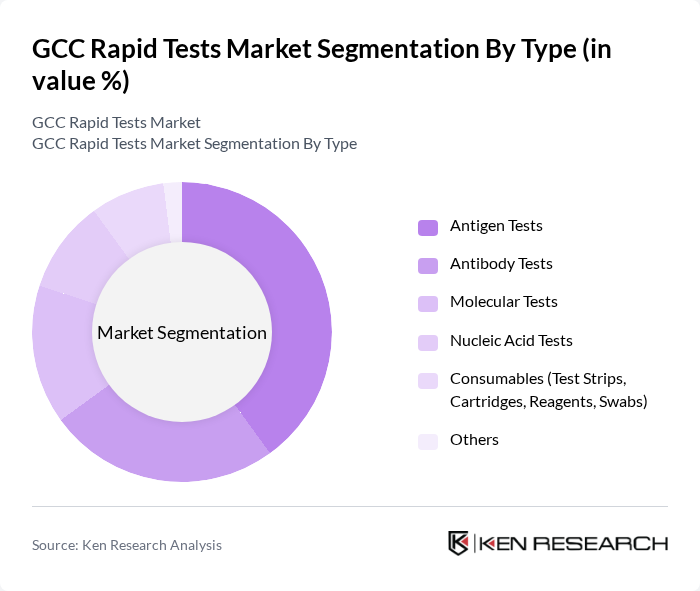
By Type:The market is segmented into various types of rapid tests, including Antigen Tests, Antibody Tests, Molecular Tests, Nucleic Acid Tests, Consumables (Test Strips, Cartridges, Reagents, Swabs), and Others. Among these, Antigen Tests are currently leading the market due to their rapid results, ease of use, and cost-effectiveness, making them highly preferred in both clinical and home settings. The demand for Antigen Tests surged during the pandemic, and their continued relevance in routine health checks and respiratory disease surveillance keeps them at the forefront of the market.
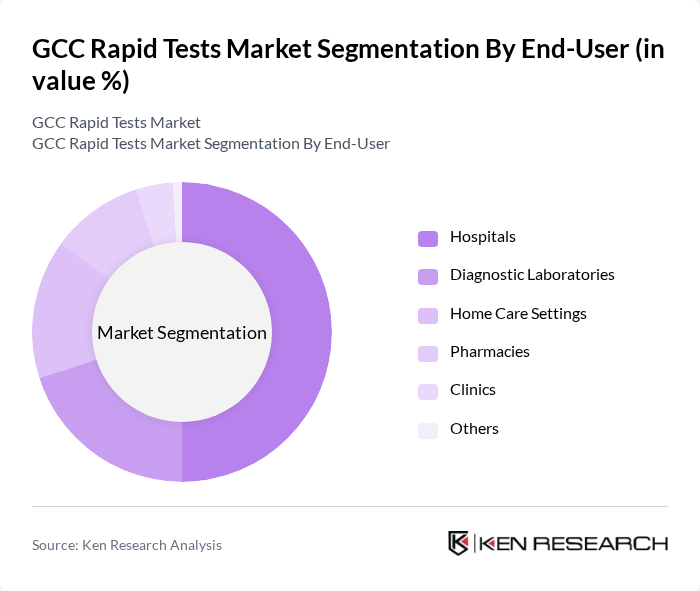
By End-User:The end-user segmentation includes Hospitals, Diagnostic Laboratories, Home Care Settings, Pharmacies, Clinics, and Others. Hospitals are the leading end-user segment, driven by the need for rapid diagnostics in emergency and outpatient settings. The increasing patient load, demand for timely clinical decisions, and integration of rapid tests into hospital workflows have made hospitals the primary consumers of rapid testing solutions. Diagnostic laboratories and home care settings are also seeing increased adoption due to expanded decentralized testing and self-diagnosis trends.
The GCC Rapid Tests Market is characterized by a dynamic mix of regional and international players. Leading participants such as Abbott Laboratories, Roche Diagnostics, Siemens Healthineers, BD (Becton, Dickinson and Company), Cepheid, Bio-Rad Laboratories, Quidel Corporation, Hologic, Inc., Thermo Fisher Scientific, Ortho Clinical Diagnostics, GenMark Diagnostics, DiaSorin S.p.A., Mylab Discovery Solutions, Eiken Chemical Co., Ltd., BioMérieux, PerkinElmer, The Binding Site Group, Fujifilm, ZyGene, Danaher Corporation contribute to innovation, geographic expansion, and service delivery in this space.
The future of the GCC rapid tests market appears promising, driven by ongoing technological advancements and increasing healthcare investments. With the GCC governments prioritizing healthcare infrastructure, the market is expected to see enhanced accessibility to rapid testing solutions. Additionally, the integration of artificial intelligence in testing processes is anticipated to improve accuracy and efficiency, further propelling market growth. The focus on personalized medicine will also shape the development of tailored rapid tests, meeting specific patient needs.
| Segment | Sub-Segments |
|---|---|
| By Type | Antigen Tests Antibody Tests Molecular Tests Nucleic Acid Tests Consumables (Test Strips, Cartridges, Reagents, Swabs) Others |
| By End-User | Hospitals Diagnostic Laboratories Home Care Settings Pharmacies Clinics Others |
| By Application | Infectious Disease Testing (COVID-19, Influenza, HIV, STDs) Drug Testing Blood Glucose Monitoring Pregnancy Testing Food Safety Testing Others |
| By Distribution Channel | Direct Sales Online Retail Distributors Pharmacies Others |
| By Region | Saudi Arabia UAE Qatar Kuwait Oman Bahrain |
| By Technology | Lateral Flow Assays Enzyme-Linked Immunosorbent Assay (ELISA) Polymerase Chain Reaction (PCR) Chromatography-Based Immunoassay-Based Others |
| By Policy Support | Government Subsidies Tax Incentives Research Grants Others |
| Scope Item/Segment | Sample Size | Target Respondent Profiles |
|---|---|---|
| Healthcare Providers | 100 | Doctors, Nurses, Laboratory Technicians |
| Distributors of Rapid Test Kits | 60 | Sales Managers, Product Managers |
| Regulatory Bodies | 40 | Policy Makers, Compliance Officers |
| End-users (Patients) | 50 | General Public, Patients with Chronic Conditions |
| Healthcare Institutions | 50 | Hospital Administrators, Procurement Officers |
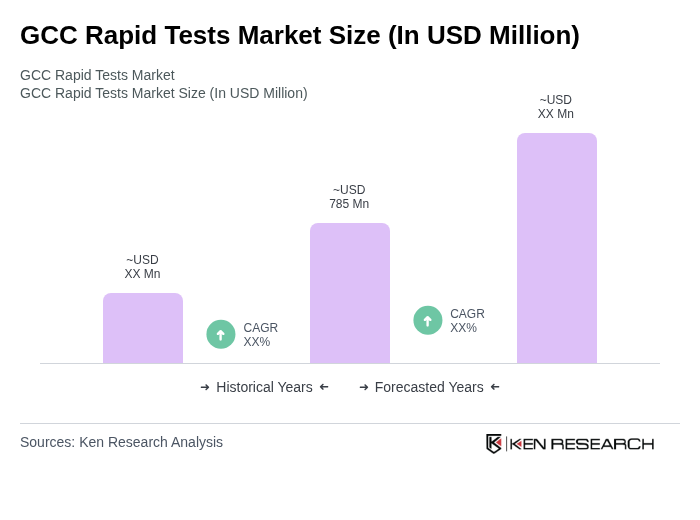
The GCC Rapid Tests Market is valued at approximately USD 785 million, reflecting significant growth driven by the demand for quick and accurate diagnostic solutions, particularly in the wake of the COVID-19 pandemic.