Region:Global
Author(s):Dev
Product Code:KRAA3043
Pages:93
Published On:August 2025
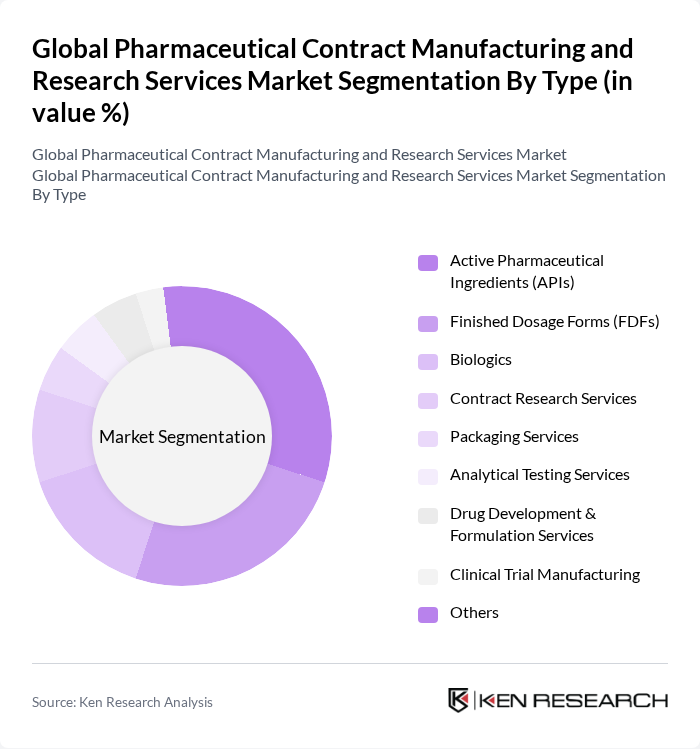
By Type:The market is segmented into various types, including Active Pharmaceutical Ingredients (APIs), Finished Dosage Forms (FDFs), Biologics, Contract Research Services, Packaging Services, Analytical Testing Services, Drug Development & Formulation Services, Clinical Trial Manufacturing, and Others. Each of these subsegments plays a vital role in the overall market dynamics. APIs and FDFs constitute the largest shares, reflecting the core outsourcing needs of the industry, while biologics and specialized services such as analytical testing and clinical trial manufacturing are rapidly growing due to the complexity and regulatory requirements of modern drug development .
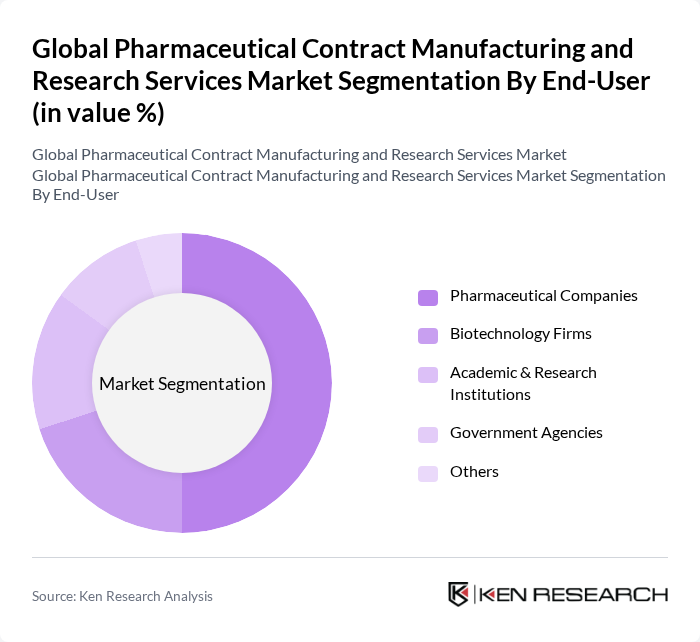
By End-User:The end-user segmentation includes Pharmaceutical Companies, Biotechnology Firms, Academic & Research Institutions, Government Agencies, and Others. Each of these segments has unique requirements and contributes differently to the market. Pharmaceutical companies remain the dominant end-users, leveraging contract manufacturing and research services to optimize operational efficiency and focus on core R&D, while biotechnology firms and research institutions increasingly utilize these services for specialized and early-stage development needs .
The Global Pharmaceutical Contract Manufacturing and Research Services Market is characterized by a dynamic mix of regional and international players. Leading participants such as Lonza Group AG, Catalent, Inc., Samsung Biologics, WuXi AppTec, Patheon (Thermo Fisher Scientific), Recipharm AB, Aenova Group, Siegfried Holding AG, Famar Health, Piramal Pharma Solutions, Vetter Pharma International GmbH, Jubilant HollisterStier, Almac Group, Boehringer Ingelheim BioXcellence, Fujifilm Diosynth Biotechnologies, AbbVie Contract Manufacturing, Pfizer CentreOne, Fareva, CordenPharma, Aesica Pharmaceuticals, Dr. Reddy's Laboratories, Prakruti Life Science contribute to innovation, geographic expansion, and service delivery in this space.
The future of the pharmaceutical contract manufacturing and research services market appears promising, driven by technological advancements and increasing collaboration among industry players. As companies adopt continuous manufacturing processes and integrate AI into operations, efficiency and quality are expected to improve significantly. Furthermore, the growing emphasis on personalized medicine will likely lead to tailored manufacturing solutions, enhancing the market's adaptability and responsiveness to evolving healthcare needs in the None region.
| Segment | Sub-Segments |
|---|---|
| By Type | Active Pharmaceutical Ingredients (APIs) Finished Dosage Forms (FDFs) Biologics Contract Research Services Packaging Services Analytical Testing Services Drug Development & Formulation Services Clinical Trial Manufacturing Others |
| By End-User | Pharmaceutical Companies Biotechnology Firms Academic & Research Institutions Government Agencies Others |
| By Service Type | Manufacturing Services Research and Development Services Quality Control & Analytical Services Regulatory Affairs Services Supply Chain & Logistics Services Others |
| By Region | North America Europe Asia-Pacific Latin America Middle East & Africa Others |
| By Client Type | Large Pharmaceutical Companies Small and Medium Enterprises (SMEs) Virtual/Biotech Startups Others |
| By Contract Duration | Short-term Contracts Long-term Contracts Project-based Contracts Others |
| By Pricing Model | Fixed Pricing Cost-plus Pricing Performance-based Pricing Milestone-based Payments Others |
| Scope Item/Segment | Sample Size | Target Respondent Profiles |
|---|---|---|
| Pharmaceutical Contract Manufacturing | 100 | Manufacturing Directors, Quality Assurance Managers |
| Research Services in Biopharma | 60 | R&D Managers, Clinical Trial Managers |
| API Production and Supply Chain | 50 | Procurement Managers, Supply Chain Analysts |
| Regulatory Affairs and Compliance | 40 | Regulatory Affairs Specialists, Compliance Officers |
| Market Access and Commercialization | 50 | Market Access Managers, Business Development Executives |
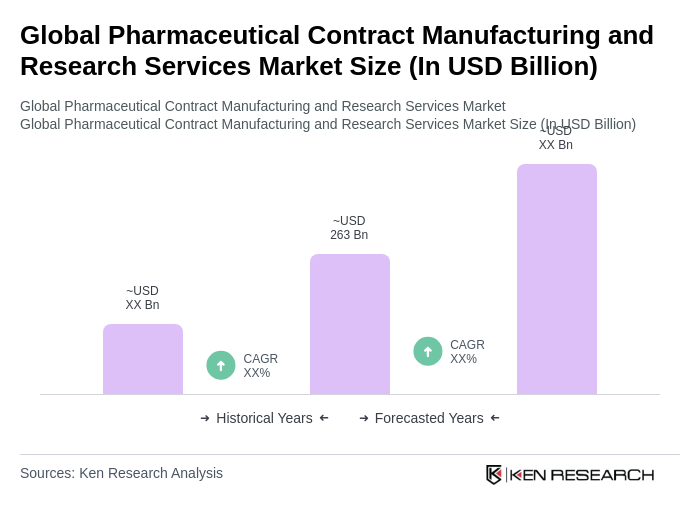
The Global Pharmaceutical Contract Manufacturing and Research Services Market is valued at approximately USD 263 billion, reflecting significant growth driven by the increasing demand for outsourcing manufacturing and research services in the pharmaceutical industry.