Region:Asia
Author(s):Shubham
Product Code:KRAA4997
Pages:80
Published On:September 2025
By Type:The market is segmented into various types of diagnostic services, including Clinical Chemistry, Microbiology, Hematology, Immunology, Molecular Diagnostics, Pathology, and Others. Each of these segments plays a crucial role in the overall diagnostics landscape, catering to different healthcare needs and patient demographics.
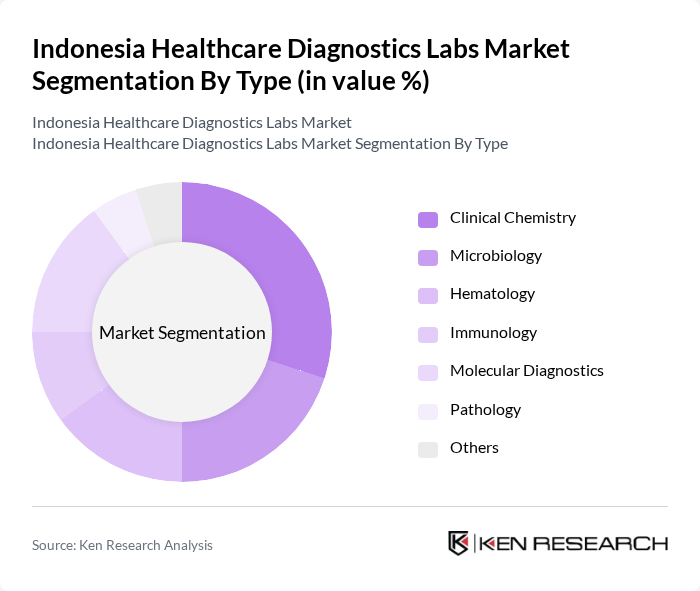
The Clinical Chemistry segment is currently dominating the market due to its extensive application in disease diagnosis and monitoring. This segment encompasses a wide range of tests that are essential for assessing metabolic and organ function, making it a critical component of patient care. The increasing incidence of lifestyle-related diseases, such as diabetes and cardiovascular conditions, has further propelled the demand for clinical chemistry tests. Additionally, advancements in technology and automation in laboratories have enhanced the efficiency and accuracy of these tests, solidifying their market leadership.
By End-User:The market is segmented based on end-users, including Hospitals, Diagnostic Laboratories, Research Institutions, and Home Care Settings. Each segment serves distinct functions within the healthcare ecosystem, catering to various patient needs and diagnostic requirements.
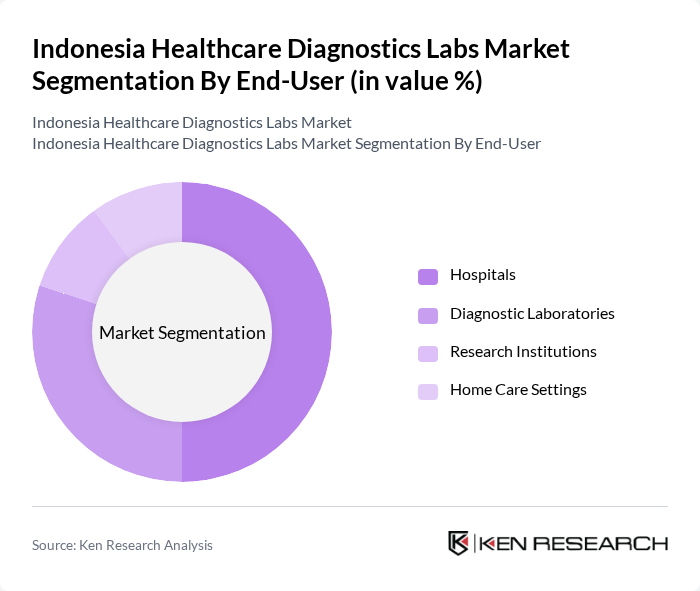
Hospitals are the leading end-user segment in the market, accounting for a significant share due to their comprehensive healthcare services and the necessity for in-house diagnostic capabilities. The increasing patient volume in hospitals, driven by the rising prevalence of diseases and the need for timely diagnosis, has led to a higher demand for diagnostic services. Furthermore, hospitals often invest in advanced diagnostic technologies to enhance patient care and streamline operations, reinforcing their dominant position in the market.
The Indonesia Healthcare Diagnostics Labs Market is characterized by a dynamic mix of regional and international players. Leading participants such as PT. Kimia Farma Tbk, PT. Bio Farma, PT. Prodia Widyahusada Tbk, PT. Laboratorium Klinik Bunda, PT. Siloam International Hospitals Tbk, PT. RSUP Dr. Cipto Mangunkusumo, PT. Medisafe Technologies, PT. SehatQ, PT. Alodokter, PT. Halodoc, PT. Klinik Pratama, PT. Mitra Keluarga Karyasehat Tbk, PT. RSUP Dr. Hasan Sadikin, PT. Medika Bina Sehat, PT. Citra Medika contribute to innovation, geographic expansion, and service delivery in this space.
The future of Indonesia's healthcare diagnostics labs market appears promising, driven by increasing investments in healthcare infrastructure and technology. The government’s commitment to enhancing healthcare access, particularly in rural areas, is expected to facilitate growth. Additionally, the rising trend of personalized medicine and home-based diagnostics will likely reshape service delivery models. As telemedicine continues to expand, diagnostics labs will play a crucial role in providing timely and accurate testing, further solidifying their importance in the healthcare ecosystem.
| Segment | Sub-Segments |
|---|---|
| By Type | Clinical Chemistry Microbiology Hematology Immunology Molecular Diagnostics Pathology Others |
| By End-User | Hospitals Diagnostic Laboratories Research Institutions Home Care Settings |
| By Application | Disease Diagnosis Health Screening Drug Development Clinical Trials |
| By Distribution Channel | Direct Sales Online Sales Distributors |
| By Region | Java Sumatra Bali and Nusa Tenggara Kalimantan Sulawesi Maluku and Papua |
| By Pricing Strategy | Premium Pricing Competitive Pricing Value-Based Pricing |
| By Technology Adoption | Traditional Methods Automated Systems Digital Solutions |
| Scope Item/Segment | Sample Size | Target Respondent Profiles |
|---|---|---|
| Diagnostic Imaging Services | 100 | Radiologists, Imaging Technicians |
| Pathology Labs | 80 | Pathologists, Lab Managers |
| Molecular Diagnostics | 70 | Geneticists, Laboratory Scientists |
| Patient Experience in Diagnostics | 90 | Patients, Healthcare Advocates |
| Healthcare Provider Insights | 110 | General Practitioners, Specialists |
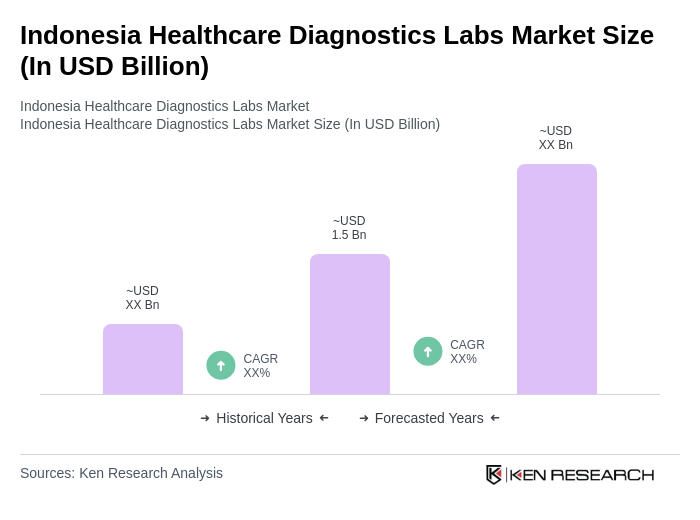
The Indonesia Healthcare Diagnostics Labs Market is valued at approximately USD 1.5 billion, reflecting significant growth driven by the rising prevalence of chronic diseases, increased healthcare expenditure, and advancements in diagnostic technologies.