About the Report
Base Year 2024Malaysia Branded Generics Market Overview
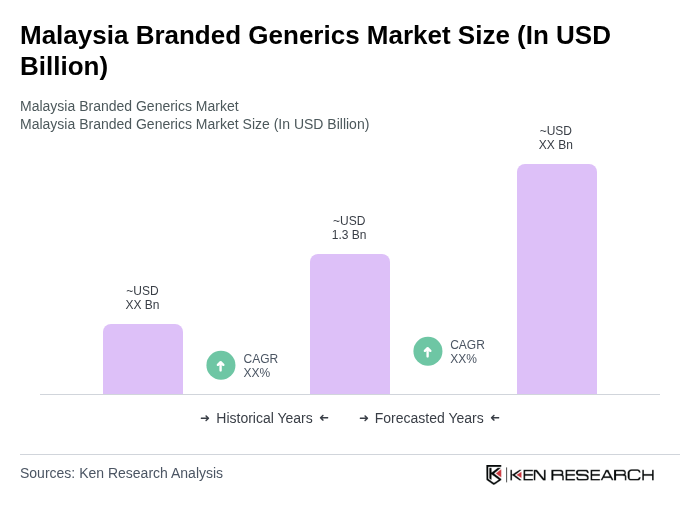
- The Malaysia Branded Generics Market is valued at USD 1.3 billion, based on a five-year historical analysis. This growth is primarily driven by the increasing demand for affordable healthcare solutions, rising prevalence of chronic diseases, government initiatives to promote generic medications, and growing healthcare investments that support local manufacturing and streamline regulatory processes. The market has seen a significant shift towards branded generics as consumers and healthcare providers seek cost-effective alternatives to branded pharmaceuticals.
- Key players in this market include Kuala Lumpur, Penang, and Johor Bahru, which dominate due to their robust healthcare infrastructure, presence of pharmaceutical companies, and strategic locations for distribution. These cities are also home to a large population, contributing to the demand for branded generics and fostering a competitive environment for manufacturers.
- The National Medicines Policy, 2014 issued by the Ministry of Health Malaysia, aims to enhance access to essential medicines, including branded generics. This policy encourages the production and use of generics through regulatory support, incentives for manufacturers, quality standards for registration and manufacturing, and promotion of rational use to improve public health outcomes and reduce healthcare costs.
Malaysia Branded Generics Market Segmentation
By Product Type:The product type segmentation includes Branded Generics, Unbranded Generics, Biosimilars, and Others. Among these, Branded Generics are currently leading the market due to their perceived quality and effectiveness, which appeals to both healthcare providers and patients. The growing acceptance of these products, coupled with aggressive marketing strategies by manufacturers, has significantly contributed to their dominance. Unbranded Generics also hold a substantial share, driven by cost-conscious consumers seeking affordable alternatives.

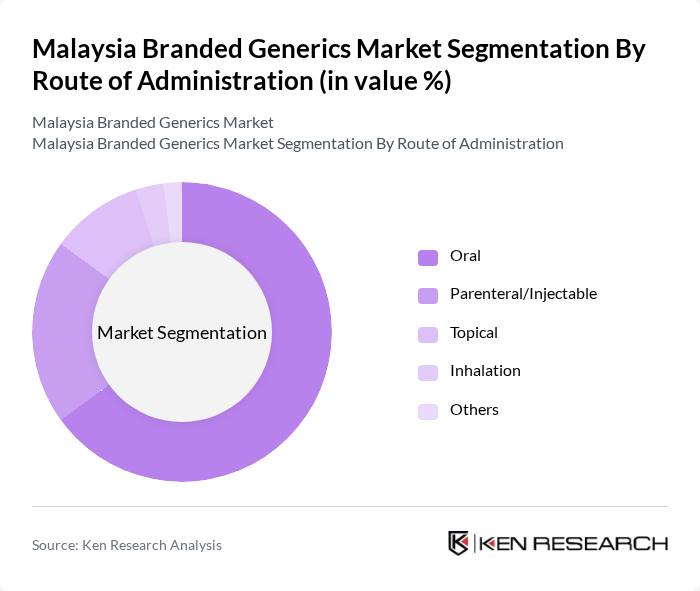
By Route of Administration:The route of administration includes Oral, Parenteral/Injectable, Topical, Inhalation, and Others. Oral administration dominates the market due to its convenience and ease of use, making it the preferred choice for both patients and healthcare providers. The increasing prevalence of chronic diseases requiring long-term medication has further solidified the demand for oral formulations. Parenteral/Injectable routes are also significant, particularly in hospital settings for acute care.
Malaysia Branded Generics Market Competitive Landscape
The Malaysia Branded Generics Market is characterized by a dynamic mix of regional and international players. Leading participants such as Pharmaniaga Berhad, Duopharma Biotech Berhad, Hovid Berhad, Kotra Pharma (M) Sdn Bhd, Apex Healthcare Berhad, CCM Duopharma Biotech Berhad, Viatris (Mylan) Malaysia Sdn Bhd, Sandoz (a Novartis division), Sun Pharmaceutical Industries Ltd., GlaxoSmithKline (GSK), Merck & Co., Inc., Pfizer Inc., Sanofi S.A., Teva Pharmaceutical Industries Ltd., Aurobindo Pharma contribute to innovation, geographic expansion, and service delivery in this space.
Malaysia Branded Generics Market Industry Analysis
Growth Drivers
- Increasing Demand for Affordable Medications:The Malaysian healthcare system is witnessing a significant shift towards affordable medications, driven by a growing population of approximately 34 million. In future, the total healthcare expenditure is projected to reach RM 75 billion, with a substantial portion allocated to generic drugs. This trend is fueled by rising out-of-pocket expenses, as 60% of healthcare costs are borne by consumers, prompting a preference for cost-effective alternatives.
- Government Initiatives to Promote Generic Drugs:The Malaysian government has implemented various initiatives to enhance the accessibility of generic medications. In future, the Ministry of Health plans to allocate RM 1.6 billion to support the production and distribution of generics. This includes tax incentives for local manufacturers and public awareness campaigns, which aim to increase the market share of generics from 40% to 50% in future, thereby improving public health outcomes.
- Rising Healthcare Expenditure:Malaysia's healthcare expenditure is on an upward trajectory, expected to grow from RM 75 billion in future to RM 85 billion by future. This increase is driven by an aging population and a higher prevalence of chronic diseases, which necessitate ongoing medication. As healthcare budgets expand, the demand for branded generics is anticipated to rise, as they offer a cost-effective solution for managing long-term health conditions, thus supporting market growth.
Market Challenges
- Intense Competition from Multinational Corporations:The Malaysian branded generics market faces fierce competition from multinational corporations, which dominate approximately 60% of the market share. These companies leverage extensive resources for marketing and distribution, making it challenging for local players to compete effectively. In future, the entry of new multinational brands is expected to further intensify this competition, potentially squeezing profit margins for local manufacturers.
- Regulatory Hurdles and Compliance Issues:Navigating the regulatory landscape in Malaysia poses significant challenges for branded generics manufacturers. The Drug Control Authority (DCA) enforces stringent regulations, requiring compliance with quality assurance standards. In future, the average time for drug approval is projected to be 18 months, which can delay market entry and increase costs for local companies. This regulatory burden can hinder innovation and limit the growth potential of the sector.
Malaysia Branded Generics Market Future Outlook
The future of the Malaysia branded generics market appears promising, driven by increasing healthcare demands and supportive government policies. As the population ages and chronic diseases rise, the need for affordable medications will intensify. Additionally, advancements in digital health solutions and e-commerce platforms are expected to enhance accessibility and distribution. By fostering partnerships with healthcare providers, local manufacturers can further penetrate the market, ensuring that generics become a staple in Malaysia's healthcare landscape.
Market Opportunities
- Growth in Chronic Disease Management:With chronic diseases affecting over 31% of the Malaysian population, there is a significant opportunity for branded generics to cater to this demographic. The government’s focus on managing these conditions through affordable medications can lead to increased sales and market penetration for local manufacturers, enhancing overall public health.
- Expansion into Rural Markets:Approximately 31% of Malaysia's population resides in rural areas, where access to affordable medications is limited. By targeting these underserved markets, branded generics can fill a critical gap. Initiatives to improve distribution networks in rural regions can significantly boost sales and improve healthcare access for these communities, creating a win-win scenario.
Scope of the Report
| Segment | Sub-Segments |
|---|---|
| By Product Type | Branded Generics Unbranded Generics Biosimilars Others |
| By Route of Administration | Oral Parenteral/Injectable Topical Inhalation Others |
| By Therapeutic Area | Cardiovascular Anti-infectives/Antibiotics Central Nervous System (CNS) Oncology Gastrointestinal Respiratory Endocrine & Metabolic (incl. Diabetes) Others |
| By Distribution Channel | Hospital Pharmacies Retail Pharmacies/Drugstores Online/E-pharmacies Government/Institutional Procurement Wholesalers & Distributors Others |
| By Manufacturer Type | Local Manufacturers Multinational Companies Contract Manufacturing Organizations (CMOs) Others |
| By Formulation | Tablets Capsules Liquids/Suspensions Ointments/Creams/Gels Others |
| By Region | Central Region Northern Region Southern Region Eastern Region |
Key Target Audience
Investors and Venture Capitalist Firms
Government and Regulatory Bodies (e.g., Ministry of Health Malaysia, National Pharmaceutical Regulatory Agency)
Manufacturers and Producers
Distributors and Retailers
Pharmaceutical Wholesalers
Healthcare Providers and Institutions
Industry Associations (e.g., Malaysian Pharmaceutical Society)
Financial Institutions
Players Mentioned in the Report:
Pharmaniaga Berhad
Duopharma Biotech Berhad
Hovid Berhad
Kotra Pharma (M) Sdn Bhd
Apex Healthcare Berhad
CCM Duopharma Biotech Berhad
Viatris (Mylan) Malaysia Sdn Bhd
Sandoz (a Novartis division)
Sun Pharmaceutical Industries Ltd.
GlaxoSmithKline (GSK)
Merck & Co., Inc.
Pfizer Inc.
Sanofi S.A.
Teva Pharmaceutical Industries Ltd.
Aurobindo Pharma
Table of Contents
Market Assessment Phase
1. Executive Summary and Approach
2. Malaysia Branded Generics Market Overview
2.1 Key Insights and Strategic Recommendations
2.2 Malaysia Branded Generics Market Overview
2.3 Definition and Scope
2.4 Evolution of Market Ecosystem
2.5 Timeline of Key Regulatory Milestones
2.6 Value Chain & Stakeholder Mapping
2.7 Business Cycle Analysis
2.8 Policy & Incentive Landscape
3. Malaysia Branded Generics Market Analysis
3.1 Growth Drivers
3.1.1 Increasing Demand for Affordable Medications
3.1.2 Government Initiatives to Promote Generic Drugs
3.1.3 Rising Healthcare Expenditure
3.1.4 Expanding Distribution Channels
3.2 Market Challenges
3.2.1 Intense Competition from Multinational Corporations
3.2.2 Regulatory Hurdles and Compliance Issues
3.2.3 Price Sensitivity Among Consumers
3.2.4 Limited Awareness of Generic Options
3.3 Market Opportunities
3.3.1 Growth in Chronic Disease Management
3.3.2 Expansion into Rural Markets
3.3.3 Partnerships with Healthcare Providers
3.3.4 Innovation in Drug Formulations
3.4 Market Trends
3.4.1 Shift Towards Digital Health Solutions
3.4.2 Increasing Focus on Sustainability
3.4.3 Rise of E-commerce in Pharmaceutical Sales
3.4.4 Growing Consumer Preference for Local Brands
3.5 Government Regulation
3.5.1 Implementation of Price Control Policies
3.5.2 Regulatory Framework for Drug Approval
3.5.3 Quality Assurance Standards for Generic Drugs
3.5.4 Incentives for Local Manufacturing
4. SWOT Analysis
5. Stakeholder Analysis
6. Porter's Five Forces Analysis
7. Malaysia Branded Generics Market Market Size, 2019-2024
7.1 By Value
7.2 By Volume
7.3 By Average Selling Price
8. Malaysia Branded Generics Market Segmentation
8.1 By Product Type
8.1.1 Branded Generics
8.1.2 Unbranded Generics
8.1.3 Biosimilars
8.1.4 Others
8.2 By Route of Administration
8.2.1 Oral
8.2.2 Parenteral/Injectable
8.2.3 Topical
8.2.4 Inhalation
8.2.5 Others
8.3 By Therapeutic Area
8.3.1 Cardiovascular
8.3.2 Anti-infectives/Antibiotics
8.3.3 Central Nervous System (CNS)
8.3.4 Oncology
8.3.5 Gastrointestinal
8.3.6 Respiratory
8.3.7 Endocrine & Metabolic (incl. Diabetes)
8.3.8 Others
8.4 By Distribution Channel
8.4.1 Hospital Pharmacies
8.4.2 Retail Pharmacies/Drugstores
8.4.3 Online/E-pharmacies
8.4.4 Government/Institutional Procurement
8.4.5 Wholesalers & Distributors
8.4.6 Others
8.5 By Manufacturer Type
8.5.1 Local Manufacturers
8.5.2 Multinational Companies
8.5.3 Contract Manufacturing Organizations (CMOs)
8.5.4 Others
8.6 By Formulation
8.6.1 Tablets
8.6.2 Capsules
8.6.3 Liquids/Suspensions
8.6.4 Ointments/Creams/Gels
8.6.5 Others
8.7 By Region
8.7.1 Central Region
8.7.2 Northern Region
8.7.3 Southern Region
8.7.4 Eastern Region
9. Malaysia Branded Generics Market Competitive Analysis
9.1 Market Share of Key Players
9.2 Cross Comparison of Key Players
9.2.1 Company Name
9.2.2 Group Size (Large, Medium, or Small as per industry convention)
9.2.3 Revenue Growth Rate (%)
9.2.4 Market Penetration Rate (%)
9.2.5 Customer Retention Rate (%)
9.2.6 Pricing Strategy (Premium/Mid-tier/Value)
9.2.7 Product Portfolio Breadth (Number of SKUs)
9.2.8 Distribution Network Coverage (%)
9.2.9 Brand Awareness Score (0-100)
9.2.10 Regulatory Compliance Rate (%)
9.3 SWOT Analysis of Top Players
9.4 Pricing Analysis
9.5 Detailed Profile of Major Companies
9.5.1 Pharmaniaga Berhad
9.5.2 Duopharma Biotech Berhad
9.5.3 Hovid Berhad
9.5.4 Kotra Pharma (M) Sdn Bhd
9.5.5 Apex Healthcare Berhad
9.5.6 CCM Duopharma Biotech Berhad
9.5.7 Viatris (Mylan) Malaysia Sdn Bhd
9.5.8 Sandoz (a Novartis division)
9.5.9 Sun Pharmaceutical Industries Ltd.
9.5.10 GlaxoSmithKline (GSK)
9.5.11 Merck & Co., Inc.
9.5.12 Pfizer Inc.
9.5.13 Sanofi S.A.
9.5.14 Teva Pharmaceutical Industries Ltd.
9.5.15 Aurobindo Pharma
10. Malaysia Branded Generics Market End-User Analysis
10.1 Procurement Behavior of Key Ministries
10.1.1 Budget Allocation Trends
10.1.2 Procurement Processes
10.1.3 Preferred Suppliers
10.1.4 Compliance Requirements
10.2 Corporate Spend on Infrastructure & Energy
10.2.1 Investment Trends
10.2.2 Key Projects
10.2.3 Funding Sources
10.2.4 Strategic Partnerships
10.3 Pain Point Analysis by End-User Category
10.3.1 Cost Management Issues
10.3.2 Quality Concerns
10.3.3 Accessibility Challenges
10.3.4 Regulatory Compliance Difficulties
10.4 User Readiness for Adoption
10.4.1 Training Needs
10.4.2 Technology Adoption Rates
10.4.3 Support Infrastructure
10.4.4 Feedback Mechanisms
10.5 Post-Deployment ROI and Use Case Expansion
10.5.1 Performance Metrics
10.5.2 Case Studies
10.5.3 User Satisfaction Levels
10.5.4 Future Expansion Plans
11. Malaysia Branded Generics Market Future Size, 2025-2030
11.1 By Value
11.2 By Volume
11.3 By Average Selling Price
12. Malaysia Branded Generics Market Future Size, 2031-2035
12.1 By Value
12.2 By Volume
12.3 By Average Selling Price
13. Market Dynamics and Forecast Assumptions
14. Scenario Analysis
15. Risk Assessment and Mitigation Strategies
Go-To-Market Strategy Phase
1. Whitespace Analysis + Business Model Canvas
1.1 Market Gaps Identification
1.2 Value Proposition Development
1.3 Revenue Streams
1.4 Cost Structure Analysis
1.5 Key Partnerships
1.6 Customer Segments
1.7 Channels
2. Marketing and Positioning Recommendations
2.1 Branding Strategies
2.2 Product USPs
3. Distribution Plan
3.1 Urban Retail vs Rural NGO Tie-ups
4. Channel & Pricing Gaps
4.1 Underserved Routes
4.2 Pricing Bands
5. Unmet Demand & Latent Needs
5.1 Category Gaps
5.2 Consumer Segments
6. Customer Relationship
6.1 Loyalty Programs
6.2 After-sales Service
7. Value Proposition
7.1 Sustainability
7.2 Integrated Supply Chains
8. Key Activities
8.1 Regulatory Compliance
8.2 Branding
8.3 Distribution Setup
9. Entry Strategy Evaluation
9.1 Domestic Market Entry Strategy
9.1.1 Product Mix
9.1.2 Pricing Band
9.1.3 Packaging
9.2 Export Entry Strategy
9.2.1 Target Countries
9.2.2 Compliance Roadmap
10. Entry Mode Assessment
10.1 JV
10.2 Greenfield
10.3 M&A
10.4 Distributor Model
11. Capital and Timeline Estimation
11.1 Capital Requirements
11.2 Timelines
12. Control vs Risk Trade-Off
12.1 Ownership vs Partnerships
13. Profitability Outlook
13.1 Breakeven Analysis
13.2 Long-term Sustainability
14. Potential Partner List
14.1 Distributors
14.2 JVs
14.3 Acquisition Targets
15. Execution Roadmap
15.1 Phased Plan for Market Entry
15.1.1 Market Setup
15.1.2 Market Entry
15.1.3 Growth Acceleration
15.1.4 Scale & Stabilize
15.2 Key Activities and Milestones
15.2.1 Milestone Planning
15.2.2 Activity Tracking
Research Methodology
Phase 1: Approach1
Desk Research
- Analysis of market reports from Malaysian pharmaceutical associations and health ministries
- Review of academic journals and publications on branded generics in Malaysia
- Examination of trade publications and industry news related to pharmaceutical regulations
Primary Research
- Interviews with key opinion leaders in the pharmaceutical industry, including regulatory officials
- Surveys with healthcare professionals and pharmacists regarding branded generics usage
- Focus groups with patients to understand perceptions and acceptance of branded generics
Validation & Triangulation
- Cross-validation of findings with data from healthcare expenditure reports and insurance claims
- Triangulation of insights from primary interviews with secondary data trends
- Sanity checks through expert panel discussions with industry veterans
Phase 2: Market Size Estimation1
Top-down Assessment
- Estimation of total pharmaceutical market size in Malaysia and share of branded generics
- Segmentation by therapeutic categories and distribution channels
- Incorporation of government healthcare initiatives promoting generic medications
Bottom-up Modeling
- Volume estimates based on sales data from major pharmacies and hospitals
- Cost analysis of branded generics versus originator brands
- Calculation of market share based on unit sales and revenue data from manufacturers
Forecasting & Scenario Analysis
- Multi-variable regression analysis incorporating demographic trends and healthcare policies
- Scenario modeling based on potential changes in regulatory frameworks and market dynamics
- Projections for market growth under baseline, optimistic, and pessimistic scenarios through 2030
Phase 3: CATI Sample Composition1
| Scope Item/Segment | Sample Size | Target Respondent Profiles |
|---|---|---|
| Pharmacists in Urban Areas | 120 | Community Pharmacists, Pharmacy Managers |
| Healthcare Professionals | 100 | Doctors, Nurses, Healthcare Administrators |
| Patients Using Branded Generics | 100 | Chronic Disease Patients, General Consumers |
| Pharmaceutical Distributors | 80 | Distribution Managers, Sales Representatives |
| Regulatory Officials | 40 | Government Health Officials, Policy Makers |
Frequently Asked Questions
What is the current value of the Malaysia Branded Generics Market?
The Malaysia Branded Generics Market is valued at approximately USD 1.3 billion, reflecting a significant growth trend driven by the demand for affordable healthcare solutions and government initiatives promoting generic medications.