Region:Middle East
Author(s):Rebecca
Product Code:KRAA0656
Pages:98
Published On:January 2026
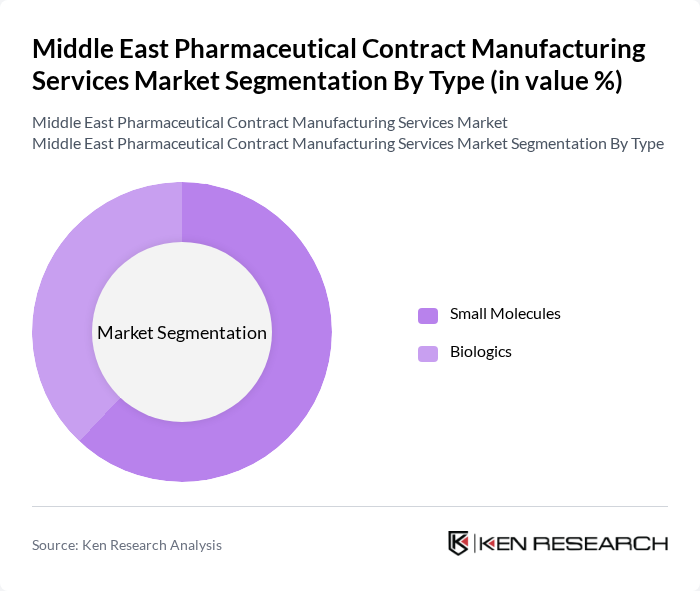
By Type:The market is segmented into Small Molecules and Biologics. Small molecules are widely used in various therapeutic areas due to their established efficacy and lower production costs. Biologics, on the other hand, are gaining traction due to their targeted therapeutic effects and the increasing prevalence of chronic diseases. The demand for biologics is expected to rise as more companies invest in innovative therapies.
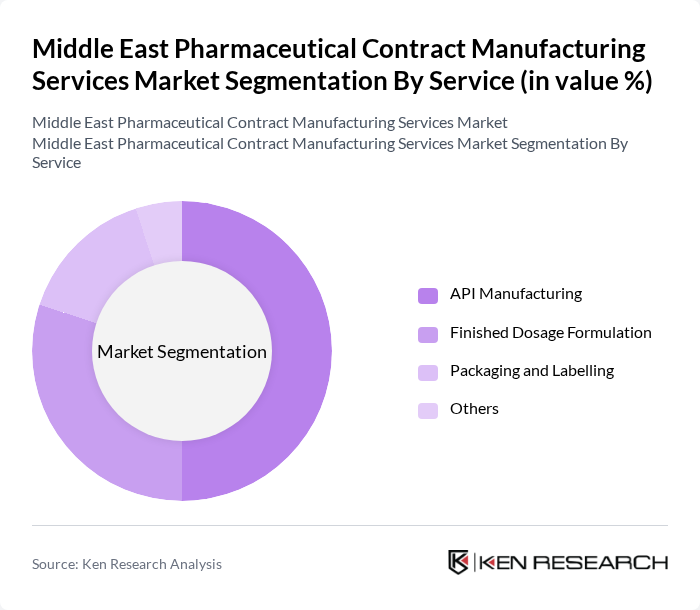
By Service:The market is categorized into API Manufacturing, Finished Dosage Formulation, Packaging and Labelling, and Others. API Manufacturing is the leading service segment, driven by the increasing demand for active pharmaceutical ingredients in various therapeutic applications. Finished Dosage Formulation is also significant, as companies focus on delivering ready-to-use products to healthcare providers and patients.
The Middle East Pharmaceutical Contract Manufacturing Services Market is characterized by a dynamic mix of regional and international players. Leading participants such as Julphar, Hikma Pharmaceuticals, Neopharma, Gulf Pharmaceutical Industries (Julphar), Pharma International, Al Haramain Pharmaceutical, Tabuk Pharmaceuticals, Dar Al Dawa, United Pharmacies, Aster DM Healthcare, Al Ain Pharmaceuticals, Globalpharma, Medpharma, Siham Pharmaceutical, Al Jazeera Pharmaceutical contribute to innovation, geographic expansion, and service delivery in this space.
The future of the Middle East pharmaceutical contract manufacturing services market appears promising, driven by technological advancements and a growing emphasis on outsourcing. As companies increasingly seek to optimize production costs and focus on core competencies, the trend towards outsourcing manufacturing is expected to gain momentum. Additionally, the integration of digital technologies, such as automation and data analytics, will enhance operational efficiency, enabling manufacturers to respond swiftly to market demands and regulatory changes.
| Segment | Sub-Segments |
|---|---|
| By Type | Small Molecules Biologics |
| By Service | API Manufacturing Finished Dosage Formulation Packaging and Labelling Others |
| By Workflow | Preclinical Clinical Commercial |
| By End-User | Large Pharmaceutical Companies Medium Pharmaceutical Companies Small Pharmaceutical Companies |
| By Therapeutic Area | Oncology Cardiovascular Neurology Others |
| By Region | GCC Countries Levant Region North Africa Others |
| Scope Item/Segment | Sample Size | Target Respondent Profiles |
|---|---|---|
| Pharmaceutical Contract Manufacturing | 120 | Operations Managers, Business Development Executives |
| Regulatory Compliance in Pharma | 85 | Regulatory Affairs Managers, Quality Assurance Officers |
| Market Trends in Biopharmaceuticals | 75 | Market Analysts, Product Managers |
| Supply Chain Management in Pharma | 65 | Supply Chain Directors, Procurement Specialists |
| Emerging Markets for Pharma Products | 80 | Market Researchers, Strategic Planners |
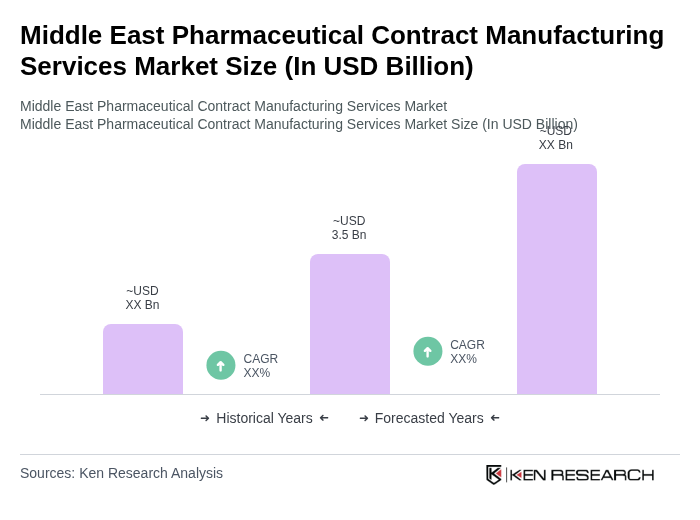
The Middle East Pharmaceutical Contract Manufacturing Services Market is valued at approximately USD 3.5 billion, driven by factors such as rising healthcare spending, increasing demand for cost-effective manufacturing solutions, and government investments in healthcare infrastructure.