About the Report
Base Year 2024Middle East Virtual Clinical Trials Market Overview
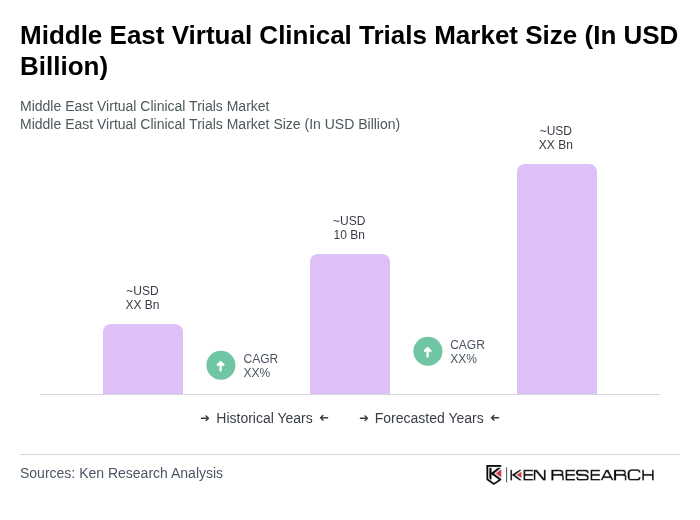
- The Middle East Virtual Clinical Trials Market is valued at USD 10 billion, based on a five-year historical analysis. This growth is primarily driven by advancements in digital health infrastructure and the increasing prevalence of chronic and complex diseases, which propel the adoption of decentralized trial models. Enhanced regulatory frameworks and investments in telemedicine, AI-powered analytics, and real-world data capabilities are also key growth enablers, supporting more efficient and patient-centric virtual trial conduct.
- Key players in this market include Saudi Arabia and the UAE, where robust healthcare infrastructure, strategic investments, and streamlined digital regulatory pathways foster dynamic market development. These countries are leading the way in virtual clinical trials due to their commitment to healthcare innovation and the integration of technology in clinical research.
- In 2024, the Middle East government announced a strategic initiative to implement harmonized digital regulatory systems across GCC countries, featuring e-submission portals and AI-enabled review tools. This initiative aims to reduce approval timelines by up to 40 percent, accelerating trial initiation and ensuring data integrity.
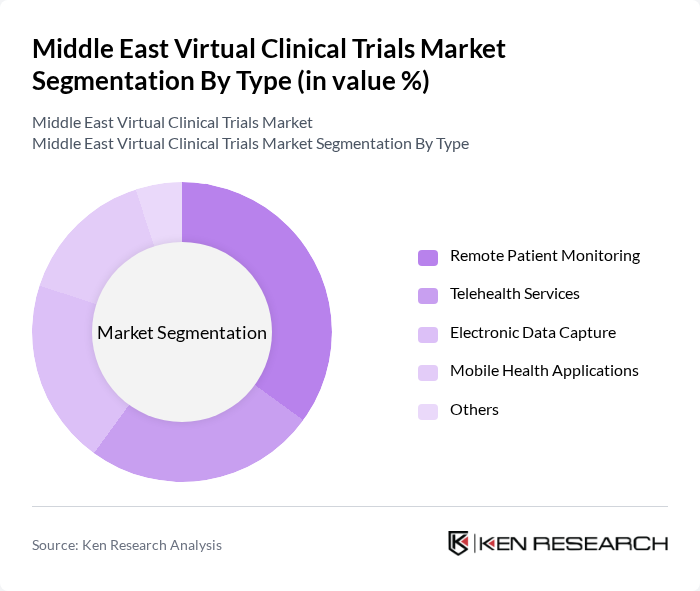
Middle East Virtual Clinical Trials Market Segmentation
By Type:The market is segmented into various types, including Remote Patient Monitoring, Telehealth Services, Electronic Data Capture, Mobile Health Applications, and Others. Among these, Remote Patient Monitoring is currently the leading sub-segment, driven by the increasing demand for continuous health monitoring and the convenience it offers to patients. The rise in chronic diseases necessitates ongoing patient engagement, making this segment crucial for virtual clinical trials.
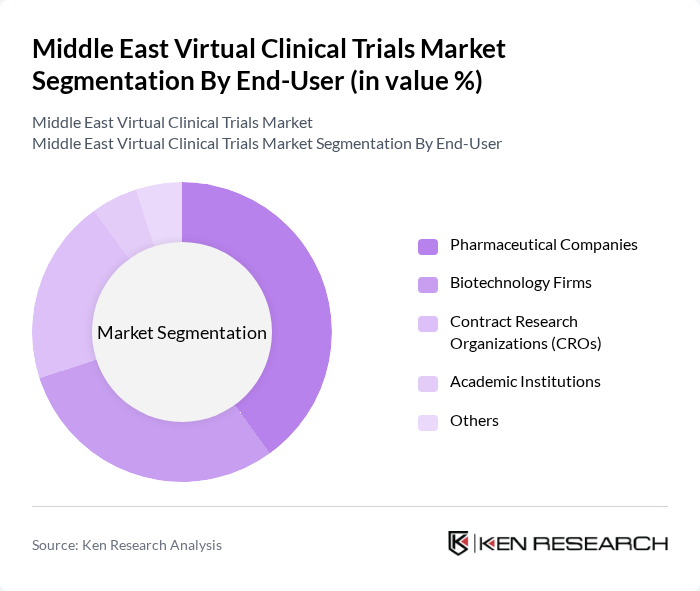
By End-User:The end-user segmentation includes Pharmaceutical Companies, Biotechnology Firms, Contract Research Organizations (CROs), Academic Institutions, and Others. Pharmaceutical Companies dominate this segment, as they are increasingly adopting virtual trials to enhance efficiency and reduce costs associated with traditional clinical trials. The shift towards patient-centric approaches in drug development is also a significant factor contributing to their leadership in this market.
Middle East Virtual Clinical Trials Market Competitive Landscape
The Middle East Virtual Clinical Trials Market is characterized by a dynamic mix of regional and international players. Leading participants such as Medable, Science 37, Medidata Solutions, Oracle Health Sciences, Parexel International, Covance, CRF Health, eClinical Solutions, Veeva Systems, Syneos Health, PPD, ICON plc, WCG Clinical, KCR, Clinipace contribute to innovation, geographic expansion, and service delivery in this space.
Middle East Virtual Clinical Trials Market Industry Analysis
Growth Drivers
- Increasing Adoption of Digital Health Technologies:The Middle East is witnessing a surge in digital health technology adoption, with investments reaching approximately $1.5 billion in the future. This growth is driven by a 30% increase in telehealth services, as healthcare providers leverage technology to enhance patient access and streamline clinical trials. The World Health Organization reported that 70% of healthcare facilities in the region are integrating digital solutions, facilitating more efficient virtual clinical trials.
- Rising Demand for Patient-Centric Trial Designs:Patient-centric trial designs are becoming increasingly popular, with 60% of clinical trial sponsors in the Middle East prioritizing patient engagement strategies. This shift is supported by a 25% increase in patient recruitment through digital platforms, allowing for more diverse participant pools. The region's healthcare expenditure is projected to reach $200 billion in the future, further emphasizing the need for innovative trial methodologies that cater to patient preferences and improve retention rates.
- Enhanced Data Collection and Analytics Capabilities:The Middle East is investing heavily in data analytics, with the market for healthcare analytics expected to grow to $1.2 billion in the future. This investment is driven by a 40% increase in the use of electronic health records (EHRs) across the region, enabling more accurate data collection for virtual trials. Enhanced analytics capabilities allow for real-time monitoring and improved decision-making, ultimately leading to more efficient trial outcomes and better patient safety.
Market Challenges
- Data Privacy and Security Concerns:Data privacy remains a significant challenge in the Middle East, with 75% of healthcare organizations expressing concerns over data breaches. The region's regulatory landscape is evolving, but only 50% of healthcare providers have implemented robust data protection measures. The lack of standardized protocols increases the risk of non-compliance, potentially jeopardizing patient trust and participation in virtual clinical trials.
- Limited Patient Engagement in Virtual Trials:Despite advancements, patient engagement in virtual trials is limited, with only 40% of eligible patients participating in such studies. Barriers include technological literacy and access to devices, particularly among older populations. The World Bank reported that 20% of the Middle East's population lacks reliable internet access, which hampers the ability to conduct effective virtual trials and limits the diversity of trial participants.
Middle East Virtual Clinical Trials Market Future Outlook
The future of virtual clinical trials in the Middle East appears promising, driven by technological advancements and a growing emphasis on patient-centric approaches. As healthcare systems increasingly adopt hybrid trial designs, the integration of artificial intelligence and real-world evidence generation will enhance trial efficiency. Furthermore, the expansion of telehealth services and collaborations with technology providers will likely create a more robust ecosystem, fostering innovation and improving patient recruitment and retention in clinical trials.
Market Opportunities
- Expansion of Telehealth Services:The ongoing expansion of telehealth services presents a significant opportunity for virtual clinical trials. With telehealth usage increasing by 50% in the future, researchers can leverage these platforms to enhance patient recruitment and streamline trial processes, ultimately improving trial outcomes and participant satisfaction.
- Collaborations with Technology Providers:Collaborations with technology providers can drive innovation in virtual clinical trials. By partnering with tech firms specializing in data analytics and patient engagement tools, clinical trial sponsors can enhance their capabilities, leading to more efficient trial designs and improved patient experiences, thereby increasing overall trial success rates.
Scope of the Report
| Segment | Sub-Segments |
|---|---|
| By Type | Remote Patient Monitoring Telehealth Services Electronic Data Capture Mobile Health Applications Others |
| By End-User | Pharmaceutical Companies Biotechnology Firms Contract Research Organizations (CROs) Academic Institutions Others |
| By Patient Demographics | Age Groups (Pediatric, Adult, Geriatric) Gender Chronic Disease Prevalence Others |
| By Therapeutic Area | Oncology Cardiovascular Diseases Neurological Disorders Infectious Diseases Others |
| By Technology Used | Cloud-Based Solutions Mobile Applications Wearable Devices Artificial Intelligence Tools Others |
| By Geographic Coverage | GCC Countries Levant Region North Africa Others |
| By Funding Source | Government Grants Private Investments Public-Private Partnerships Others |
Key Target Audience
Investors and Venture Capitalist Firms
Government and Regulatory Bodies (e.g., Ministry of Health, Food and Drug Administration)
Pharmaceutical Companies
Biotechnology Firms
Clinical Research Organizations (CROs)
Healthcare Technology Companies
Insurance Providers
Health Data Management Firms
Players Mentioned in the Report:
Medable
Science 37
Medidata Solutions
Oracle Health Sciences
Parexel International
Covance
CRF Health
eClinical Solutions
Veeva Systems
Syneos Health
PPD
ICON plc
WCG Clinical
KCR
Clinipace
Table of Contents
Market Assessment Phase
1. Executive Summary and Approach
2. Middle East Virtual Clinical Trials Market Overview
2.1 Key Insights and Strategic Recommendations
2.2 Middle East Virtual Clinical Trials Market Overview
2.3 Definition and Scope
2.4 Evolution of Market Ecosystem
2.5 Timeline of Key Regulatory Milestones
2.6 Value Chain & Stakeholder Mapping
2.7 Business Cycle Analysis
2.8 Policy & Incentive Landscape
3. Middle East Virtual Clinical Trials Market Analysis
3.1 Growth Drivers
3.1.1 Increasing adoption of digital health technologies
3.1.2 Rising demand for patient-centric trial designs
3.1.3 Enhanced data collection and analytics capabilities
3.1.4 Supportive regulatory frameworks
3.2 Market Challenges
3.2.1 Data privacy and security concerns
3.2.2 Limited patient engagement in virtual trials
3.2.3 Variability in regulatory standards across countries
3.2.4 Technological barriers for older populations
3.3 Market Opportunities
3.3.1 Expansion of telehealth services
3.3.2 Collaborations with technology providers
3.3.3 Growth in decentralized clinical trial models
3.3.4 Increasing investment in healthcare innovation
3.4 Market Trends
3.4.1 Shift towards hybrid clinical trial designs
3.4.2 Integration of artificial intelligence in trial processes
3.4.3 Focus on real-world evidence generation
3.4.4 Rise of patient recruitment platforms
3.5 Government Regulation
3.5.1 Streamlined approval processes for virtual trials
3.5.2 Guidelines for data management and security
3.5.3 Support for telemedicine in clinical research
3.5.4 Incentives for innovation in trial methodologies
4. SWOT Analysis
5. Stakeholder Analysis
6. Porter's Five Forces Analysis
7. Middle East Virtual Clinical Trials Market Size, 2019-2024
7.1 By Value
7.2 By Volume
7.3 By Average Selling Price
8. Middle East Virtual Clinical Trials Market Segmentation
8.1 By Type
8.1.1 Remote Patient Monitoring
8.1.2 Telehealth Services
8.1.3 Electronic Data Capture
8.1.4 Mobile Health Applications
8.1.5 Others
8.2 By End-User
8.2.1 Pharmaceutical Companies
8.2.2 Biotechnology Firms
8.2.3 Contract Research Organizations (CROs)
8.2.4 Academic Institutions
8.2.5 Others
8.3 By Patient Demographics
8.3.1 Age Groups (Pediatric, Adult, Geriatric)
8.3.2 Gender
8.3.3 Chronic Disease Prevalence
8.3.4 Others
8.4 By Therapeutic Area
8.4.1 Oncology
8.4.2 Cardiovascular Diseases
8.4.3 Neurological Disorders
8.4.4 Infectious Diseases
8.4.5 Others
8.5 By Technology Used
8.5.1 Cloud-Based Solutions
8.5.2 Mobile Applications
8.5.3 Wearable Devices
8.5.4 Artificial Intelligence Tools
8.5.5 Others
8.6 By Geographic Coverage
8.6.1 GCC Countries
8.6.2 Levant Region
8.6.3 North Africa
8.6.4 Others
8.7 By Funding Source
8.7.1 Government Grants
8.7.2 Private Investments
8.7.3 Public-Private Partnerships
8.7.4 Others
9. Middle East Virtual Clinical Trials Market Competitive Analysis
9.1 Market Share of Key Players
9.2 Cross Comparison of Key Players
9.2.1 Company Name
9.2.2 Group Size (Large, Medium, or Small as per industry convention)
9.2.3 Revenue Growth Rate
9.2.4 Customer Acquisition Cost
9.2.5 Market Penetration Rate
9.2.6 Customer Retention Rate
9.2.7 Pricing Strategy
9.2.8 Average Trial Duration
9.2.9 Patient Enrollment Rate
9.2.10 Regulatory Compliance Rate
9.3 SWOT Analysis of Top Players
9.4 Pricing Analysis
9.5 Detailed Profile of Major Companies
9.5.1 Medable
9.5.2 Science 37
9.5.3 Medidata Solutions
9.5.4 Oracle Health Sciences
9.5.5 Parexel International
9.5.6 Covance
9.5.7 CRF Health
9.5.8 eClinical Solutions
9.5.9 Veeva Systems
9.5.10 Syneos Health
9.5.11 PPD
9.5.12 ICON plc
9.5.13 WCG Clinical
9.5.14 KCR
9.5.15 Clinipace
10. Middle East Virtual Clinical Trials Market End-User Analysis
10.1 Procurement Behavior of Key Ministries
10.1.1 Ministry of Health Initiatives
10.1.2 Budget Allocation Trends
10.1.3 Collaboration with Private Sector
10.1.4 Others
10.2 Corporate Spend on Infrastructure & Energy
10.2.1 Investment in Digital Health Infrastructure
10.2.2 Funding for Clinical Research
10.2.3 Partnerships with Technology Firms
10.2.4 Others
10.3 Pain Point Analysis by End-User Category
10.3.1 Challenges in Patient Recruitment
10.3.2 Data Management Issues
10.3.3 Regulatory Compliance Difficulties
10.3.4 Others
10.4 User Readiness for Adoption
10.4.1 Awareness of Virtual Trials
10.4.2 Training and Support Needs
10.4.3 Technology Accessibility
10.4.4 Others
10.5 Post-Deployment ROI and Use Case Expansion
10.5.1 Measurement of Trial Success
10.5.2 Expansion into New Therapeutic Areas
10.5.3 Long-term Partnerships with Stakeholders
10.5.4 Others
11. Middle East Virtual Clinical Trials Market Future Size, 2025-2030
11.1 By Value
11.2 By Volume
11.3 By Average Selling Price
Go-To-Market Strategy Phase
1. Whitespace Analysis + Business Model Canvas
1.1 Market Gaps Identification
1.2 Business Model Development
2. Marketing and Positioning Recommendations
2.1 Branding Strategies
2.2 Product USPs
3. Distribution Plan
3.1 Urban Retail vs Rural NGO Tie-ups
4. Channel & Pricing Gaps
4.1 Underserved Routes
4.2 Pricing Bands
5. Unmet Demand & Latent Needs
5.1 Category Gaps
5.2 Consumer Segments
6. Customer Relationship
6.1 Loyalty Programs
6.2 After-sales Service
7. Value Proposition
7.1 Sustainability
7.2 Integrated Supply Chains
8. Key Activities
8.1 Regulatory Compliance
8.2 Branding
8.3 Distribution Setup
9. Entry Strategy Evaluation
9.1 Domestic Market Entry Strategy
9.1.1 Product Mix
9.1.2 Pricing Band
9.1.3 Packaging
9.2 Export Entry Strategy
9.2.1 Target Countries
9.2.2 Compliance Roadmap
10. Entry Mode Assessment
10.1 JV
10.2 Greenfield
10.3 M&A
10.4 Distributor Model
11. Capital and Timeline Estimation
11.1 Capital Requirements
11.2 Timelines
12. Control vs Risk Trade-Off
12.1 Ownership vs Partnerships
13. Profitability Outlook
13.1 Breakeven Analysis
13.2 Long-term Sustainability
14. Potential Partner List
14.1 Distributors
14.2 JVs
14.3 Acquisition Targets
15. Execution Roadmap
15.1 Phased Plan for Market Entry
15.1.1 Market Setup
15.1.2 Market Entry
15.1.3 Growth Acceleration
15.1.4 Scale & Stabilize
15.2 Key Activities and Milestones
15.2.1 Milestone Planning
15.2.2 Activity Tracking
Research Methodology
Phase 1: Approach1
Desk Research
- Analysis of existing literature on virtual clinical trials in the Middle East
- Review of market reports from healthcare and technology research firms
- Examination of regulatory frameworks and guidelines from regional health authorities
Primary Research
- Interviews with clinical trial coordinators and project managers in healthcare institutions
- Surveys targeting pharmaceutical companies and biotech firms involved in virtual trials
- Focus groups with healthcare professionals and patients participating in virtual trials
Validation & Triangulation
- Cross-validation of findings through multiple data sources, including academic journals and industry publications
- Triangulation of insights from primary interviews with secondary data trends
- Sanity checks conducted through expert panels comprising industry veterans and regulatory experts
Phase 2: Market Size Estimation1
Top-down Assessment
- Estimation of market size based on healthcare expenditure and digital health adoption rates
- Segmentation of the market by therapeutic areas and types of virtual trials
- Incorporation of government initiatives promoting digital health solutions in the region
Bottom-up Modeling
- Collection of data on the number of virtual trials conducted by leading pharmaceutical companies
- Estimation of average costs associated with virtual trial implementation and management
- Volume x cost analysis based on trial phases and participant recruitment strategies
Forecasting & Scenario Analysis
- Multi-factor regression analysis considering technological advancements and patient engagement trends
- Scenario modeling based on varying levels of regulatory support and market adoption rates
- Development of baseline, optimistic, and pessimistic forecasts through 2030
Phase 3: CATI Sample Composition1
| Scope Item/Segment | Sample Size | Target Respondent Profiles |
|---|---|---|
| Pharmaceutical Companies | 100 | Clinical Research Managers, Regulatory Affairs Specialists |
| Healthcare Institutions | 80 | Trial Coordinators, Medical Directors |
| Biotech Firms | 70 | Product Development Managers, Clinical Operations Leads |
| Patient Advocacy Groups | 50 | Patient Representatives, Community Outreach Coordinators |
| Regulatory Bodies | 30 | Policy Makers, Compliance Officers |
Frequently Asked Questions
What is the current value of the Middle East Virtual Clinical Trials Market?
The Middle East Virtual Clinical Trials Market is valued at approximately USD 10 billion, driven by advancements in digital health infrastructure and the increasing prevalence of chronic diseases, which support the adoption of decentralized trial models.