Region:Middle East
Author(s):Rebecca
Product Code:KRAD7372
Pages:87
Published On:December 2025
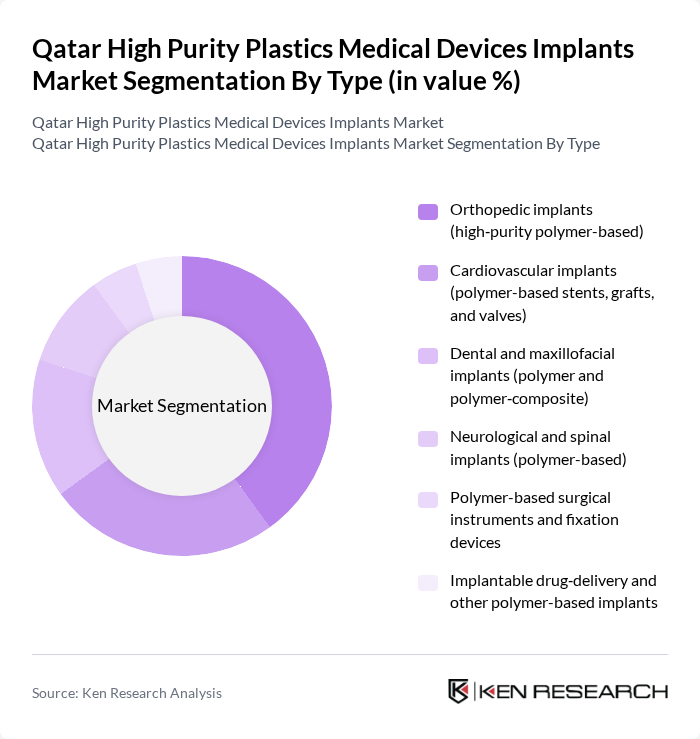
By Type:The market is segmented into various types of implants, each catering to specific medical needs. The subsegments include orthopedic implants, cardiovascular implants, dental and maxillofacial implants, neurological and spinal implants, polymer-based surgical instruments, and implantable drug-delivery systems. This structure is aligned with the global high-purity plastics for medical devices and implants market, where implantable devices, orthopedic and cardiovascular applications are the major consumers of high-purity polymers. Among these, orthopedic implants are the most dominant due to the rising incidence of musculoskeletal disorders, osteoarthritis, and trauma cases, as well as the increasing number of joint replacement and spine surgeries performed annually in Qatar’s public and private hospitals. The demand for high-purity polymer-based orthopedic implants is driven by their superior biocompatibility, wear resistance, radiolucency, and mechanical properties, which support their use in joint components, spinal cages, and fixation devices and make them a preferred choice for surgeons seeking lighter, corrosion?free alternatives to metals.
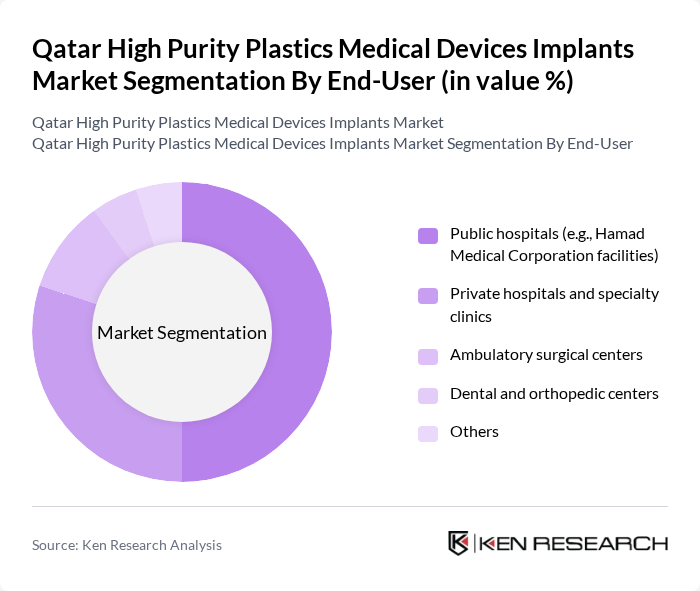
By End-User:The market is segmented based on end-users, including public hospitals, private hospitals, ambulatory surgical centers, dental and orthopedic centers, and others. Public hospitals, particularly those under the Hamad Medical Corporation, dominate the market due to their extensive patient base, advanced trauma, orthopedic, cardiovascular and oncology centers, and their central role in complex surgeries and implant procedures in Qatar. The increasing number of surgeries performed in these institutions, alongside national programs to expand elective surgery capacity and specialized centers, coupled with government support for healthcare infrastructure and universal health coverage initiatives, drives the demand for high-purity plastic implants in public hospitals. Private hospitals, day-surgery centers, and specialized dental and orthopedic clinics are also steadily increasing their use of polymer-based implants and devices as they expand minimally invasive and outpatient surgical offerings.
The Qatar High Purity Plastics Medical Devices Implants Market is characterized by a dynamic mix of regional and international players. Leading participants such as Medtronic plc, Johnson & Johnson (DePuy Synthes, Ethicon), Stryker Corporation, Boston Scientific Corporation, Zimmer Biomet Holdings, Inc., Abbott Laboratories, B. Braun Melsungen AG, Smith & Nephew plc, NuVasive, Inc., CONMED Corporation, Aesculap AG (a B. Braun company), Orthofix Medical Inc., Integra LifeSciences Holdings Corporation, Halyard Health, Inc. (now Avanos Medical, Inc.), Medline Industries, LP contribute to innovation, geographic expansion, and service delivery in this space, reflecting their strong global positions in high-purity polymer-based implants, surgical instruments, and interventional devices.
The future of the Qatar high purity plastics medical devices implants market appears promising, driven by ongoing advancements in technology and increasing healthcare investments. As the healthcare infrastructure expands, the integration of smart technologies and personalized medicine will likely enhance patient outcomes. Additionally, the growing emphasis on sustainability may lead to a shift towards biodegradable materials, aligning with global trends. These factors will create a dynamic environment for innovation and growth in the sector.
| Segment | Sub-Segments |
|---|---|
| By Type | Orthopedic implants (high?purity polymer-based) Cardiovascular implants (polymer-based stents, grafts, and valves) Dental and maxillofacial implants (polymer and polymer?composite) Neurological and spinal implants (polymer-based) Polymer-based surgical instruments and fixation devices Implantable drug?delivery and other polymer-based implants |
| By End-User | Public hospitals (e.g., Hamad Medical Corporation facilities) Private hospitals and specialty clinics Ambulatory surgical centers Dental and orthopedic centers Others |
| By Material Type | Ultra-high-molecular-weight polyethylene (UHMWPE) Polyether ether ketone (PEEK) Polytetrafluoroethylene (PTFE) and expanded PTFE (ePTFE) Bioabsorbable polymers (PLA, PGA, PLGA, PDO) Silicone and other high-purity medical-grade elastomers Others |
| By Application | Joint reconstruction and orthopedic surgery Cardiovascular and endovascular interventions Dental restoration and oral surgery Spinal and neurosurgery Implantable drug-delivery and pain?management systems Others |
| By Distribution Channel | Direct sales to hospitals and health systems Local distributors and agents Group purchasing and tender-based procurement Online and e?procurement platforms Others |
| By Region | Doha Al Rayyan Umm Salal Al Wakrah Others |
| By Regulatory Compliance | CE Marking (EU?MDR compliant devices) FDA Approval (PMA, 510(k)) ISO 10993 and related biocompatibility standards ISO 13485 and Qatari MoPH/SCH approvals Others |
| Scope Item/Segment | Sample Size | Target Respondent Profiles |
|---|---|---|
| Orthopedic Implants Market | 100 | Surgeons, Hospital Administrators |
| Cardiovascular Devices Sector | 80 | Cardiologists, Procurement Managers |
| Dental Implant Market | 70 | Dentists, Dental Clinic Owners |
| Regulatory Compliance Insights | 60 | Quality Assurance Managers, Regulatory Affairs Specialists |
| Research & Development Trends | 90 | R&D Directors, Material Scientists |
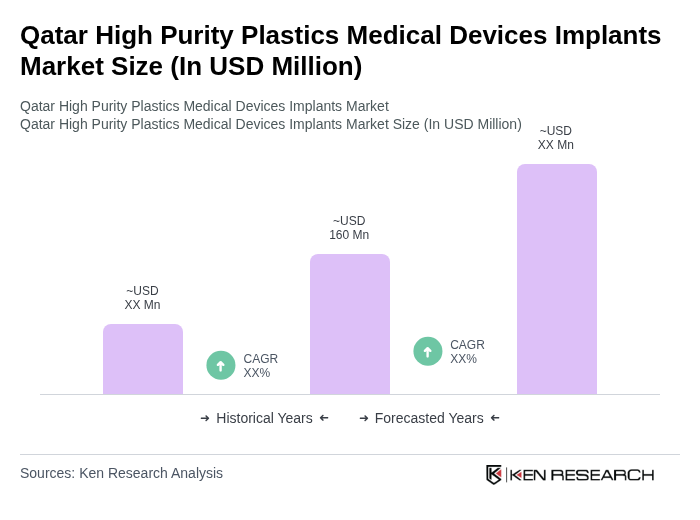
The Qatar High Purity Plastics Medical Devices Implants Market is valued at approximately USD 160 million, reflecting growth driven by advanced medical technologies, increased healthcare expenditure, and a rising aging population requiring surgical interventions.