Region:Middle East
Author(s):Geetanshi
Product Code:KRAE1118
Pages:100
Published On:February 2026
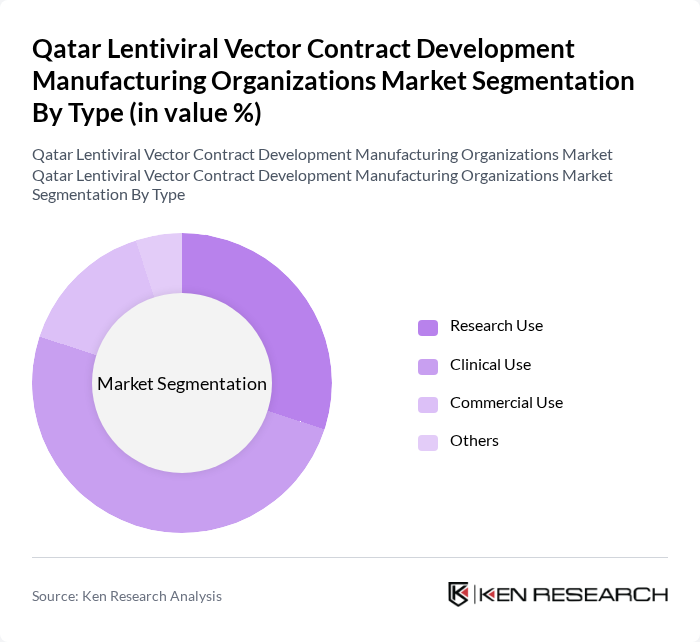
By Type:The market is segmented into various types, including Research Use, Clinical Use, Commercial Use, and Others. Among these, Clinical Use is currently the leading sub-segment, driven by the increasing number of clinical trials and the growing demand for personalized medicine. Research Use also holds a significant share as academic institutions and research organizations continue to explore the potential of lentiviral vectors in gene therapy applications.
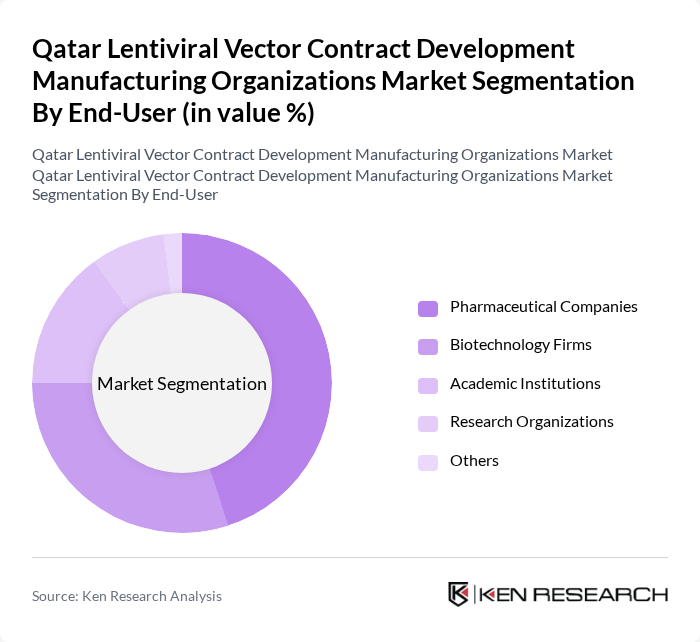
By End-User:The end-user segmentation includes Pharmaceutical Companies, Biotechnology Firms, Academic Institutions, Research Organizations, and Others. Pharmaceutical Companies are the dominant end-user, as they invest heavily in the development of gene therapies and require reliable manufacturing partners for lentiviral vectors. Biotechnology Firms also play a crucial role, focusing on innovative therapies and often collaborating with academic institutions for research purposes.
The Qatar Lentiviral Vector Contract Development Manufacturing Organizations Market is characterized by a dynamic mix of regional and international players. Leading participants such as Novartis AG, Gilead Sciences, Inc., Merck KGaA, Pfizer Inc., Bristol-Myers Squibb Company, Roche Holding AG, Sanofi S.A., Amgen Inc., Takeda Pharmaceutical Company Limited, Bluebird Bio, Inc., Cellectis S.A., Kite Pharma, Inc., Voyager Therapeutics, Inc., Sangamo Therapeutics, Inc., Oxford Biomedica PLC contribute to innovation, geographic expansion, and service delivery in this space.
The future of the lentiviral vector contract development manufacturing organizations market in Qatar appears promising, driven by increasing investments in biotechnology and a growing focus on personalized medicine. As the demand for gene therapies continues to rise, organizations are likely to enhance their manufacturing capabilities. Additionally, collaborations with academic institutions will foster innovation, leading to the development of novel therapeutic applications that address unmet medical needs, thereby expanding the market landscape significantly in future.
| Segment | Sub-Segments |
|---|---|
| By Type | Research Use Clinical Use Commercial Use Others |
| By End-User | Pharmaceutical Companies Biotechnology Firms Academic Institutions Research Organizations Others |
| By Application | Gene Therapy Vaccine Development Cell Therapy Others |
| By Delivery Method | In Vivo Ex Vivo Others |
| By Production Scale | Small Scale Medium Scale Large Scale Others |
| By Region | Doha Al Rayyan Umm Salal Others |
| By Regulatory Compliance | FDA Compliance EMA Compliance Others |
| Scope Item/Segment | Sample Size | Target Respondent Profiles |
|---|---|---|
| Gene Therapy Development | 100 | R&D Directors, Gene Therapy Scientists |
| Regulatory Compliance in Vector Production | 80 | Regulatory Affairs Managers, Quality Assurance Officers |
| Market Trends in Biomanufacturing | 90 | Business Development Managers, Market Analysts |
| Investment in Biotechnology Startups | 70 | Venture Capitalists, Biotechnology Investors |
| Healthcare Policy Impact on CDMOs | 60 | Healthcare Policy Analysts, Industry Consultants |
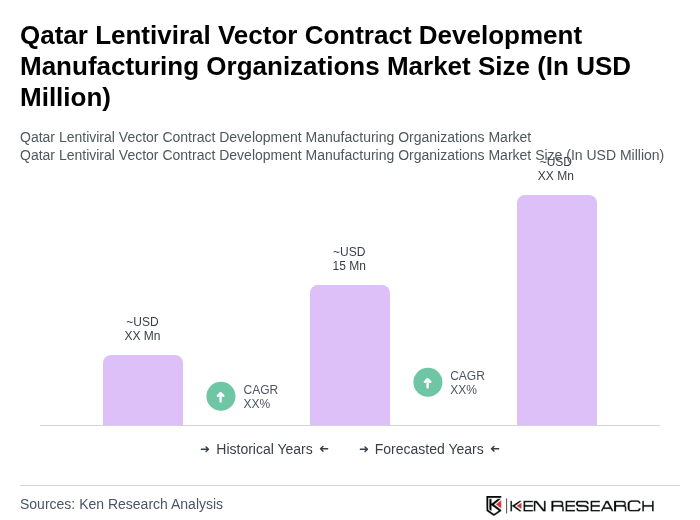
The Qatar Lentiviral Vector Contract Development Manufacturing Organizations Market is valued at approximately USD 15 million, reflecting a five-year historical analysis driven by the increasing demand for gene therapies and rising investments in biotechnology.