Region:Middle East
Author(s):Dev
Product Code:KRAD3218
Pages:91
Published On:November 2025
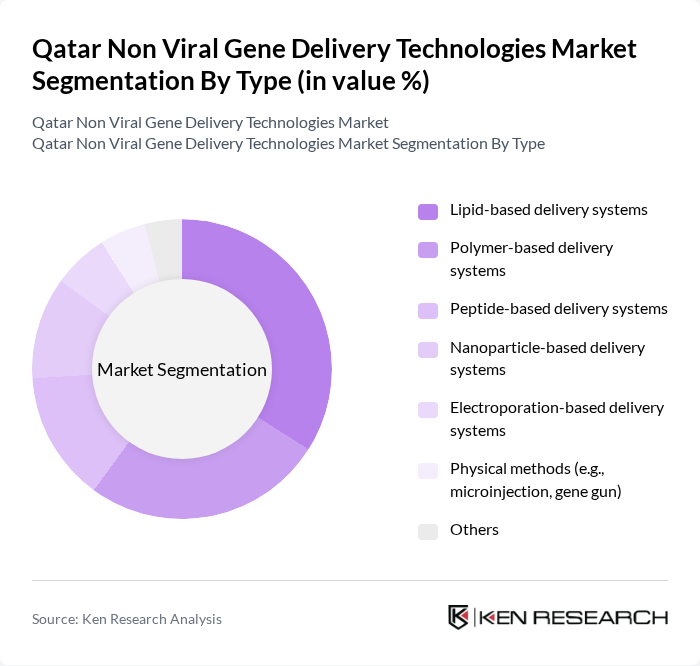
By Type:The non-viral gene delivery technologies market can be segmented into various types, including lipid-based delivery systems, polymer-based delivery systems, peptide-based delivery systems, nanoparticle-based delivery systems, electroporation-based delivery systems, physical methods (e.g., microinjection, gene gun), and others. Among these, lipid-based delivery systems are currently leading the market due to their efficiency in encapsulating genetic material and facilitating cellular uptake. The increasing focus on developing safer and more effective delivery methods has further propelled the demand for lipid-based systems .
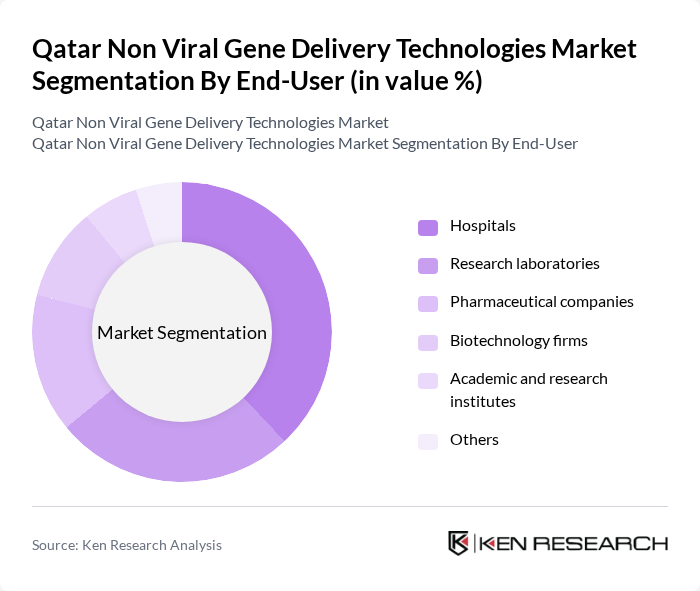
By End-User:The end-user segmentation of the non-viral gene delivery technologies market includes hospitals, research laboratories, pharmaceutical companies, biotechnology firms, academic and research institutes, and others. Hospitals are the leading end-users, driven by the increasing adoption of advanced therapeutic techniques and the need for effective treatment options for genetic disorders and cancers. The growing collaboration between hospitals and research institutions is also enhancing the utilization of these technologies in clinical settings .
The Qatar Non Viral Gene Delivery Technologies Market is characterized by a dynamic mix of regional and international players. Leading participants such as QIAGEN N.V., Thermo Fisher Scientific Inc., Lonza Group AG, Bioneer Corporation, MaxCyte, Inc., Cellectis S.A., GenScript Biotech Corporation, Merck KGaA, Bio-Rad Laboratories, Inc., Altogen Biosystems, SIRION Biotech GmbH, Innovative Cell Technologies, Inc., Sonidel Limited, Polyplus-transfection SA, Danaher Corporation contribute to innovation, geographic expansion, and service delivery in this space.
The future of the non-viral gene delivery technologies market in Qatar appears promising, driven by ongoing advancements in biotechnology and increasing collaboration between public and private sectors. As the demand for personalized medicine grows, the integration of artificial intelligence in gene delivery systems is expected to enhance treatment efficacy. Furthermore, the focus on non-invasive delivery methods will likely lead to improved patient outcomes, positioning Qatar as a regional leader in gene therapy innovations.
| Segment | Sub-Segments |
|---|---|
| By Type | Lipid-based delivery systems Polymer-based delivery systems Peptide-based delivery systems Nanoparticle-based delivery systems Electroporation-based delivery systems Physical methods (e.g., microinjection, gene gun) Others |
| By End-User | Hospitals Research laboratories Pharmaceutical companies Biotechnology firms Academic and research institutes Others |
| By Application | Cancer treatment Genetic disorder treatment Vaccine development Regenerative medicine RNA interference (RNAi) applications CRISPR/Cas9 gene editing Protein expression Others |
| By Delivery Method | Intravenous delivery Intramuscular delivery Subcutaneous delivery Oral delivery In vivo delivery Ex vivo delivery Others |
| By Target Disease | Oncological diseases Cardiovascular diseases Neurological disorders Infectious diseases Rare genetic diseases Others |
| By Region | Doha Al Rayyan Umm Salal Al Wakrah Al Khor Others |
| By Research Phase | Preclinical research Clinical trials Post-marketing surveillance Others |
| Scope Item/Segment | Sample Size | Target Respondent Profiles |
|---|---|---|
| Biotechnology Firms in Qatar | 100 | R&D Directors, Product Managers |
| Healthcare Providers and Hospitals | 90 | Clinical Researchers, Medical Directors |
| Regulatory Bodies and Health Authorities | 50 | Regulatory Affairs Specialists, Policy Makers |
| Academic Institutions and Research Centers | 70 | Professors, Research Scientists |
| Investors and Venture Capitalists in Biotech | 60 | Investment Analysts, Portfolio Managers |
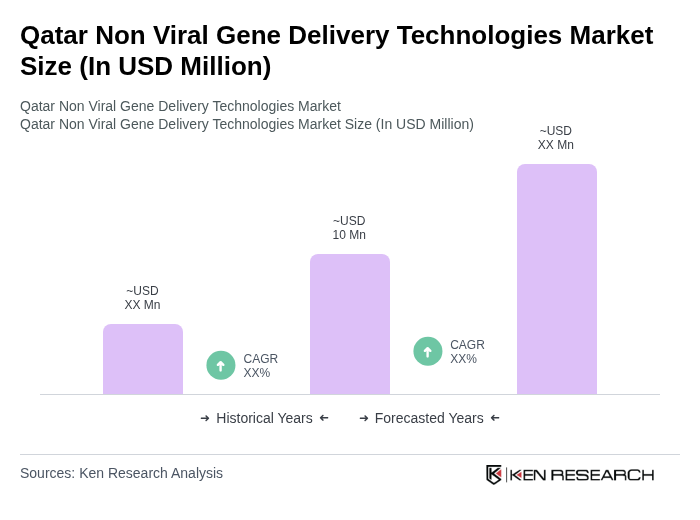
The Qatar Non Viral Gene Delivery Technologies Market is valued at approximately USD 10 million, reflecting a five-year historical analysis. This valuation is influenced by advancements in biotechnology and increasing investments in research and development.