About the Report
Base Year 2024Saudi Arabia Alpha 1 Antitrypsin Deficiency Disease Market Overview
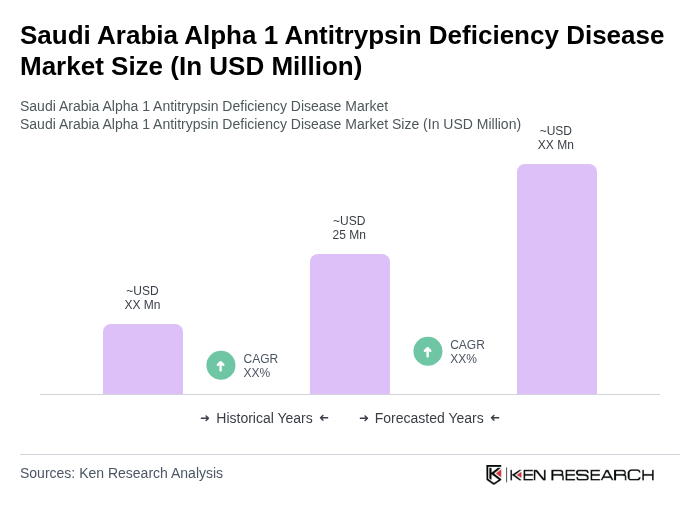
- The Saudi Arabia Alpha 1 Antitrypsin Deficiency Disease market is valued at USD 25 million, based on a five-year historical analysis. This growth is primarily driven by increasing awareness of genetic disorders, advancements in diagnostic technologies, rising prevalence of AATD-related respiratory and liver diseases, and the growing adoption of augmentation therapies. The healthcare sector's focus on personalized medicine and targeted therapies has further propelled the market's expansion.
- Key cities such as Riyadh, Jeddah, and Dammam dominate the market due to their advanced healthcare infrastructure and concentration of specialized medical facilities. These urban centers are equipped with state-of-the-art diagnostic and treatment options, attracting patients from across the region. Additionally, the presence of leading pharmaceutical companies and research institutions in these cities fosters innovation and enhances treatment accessibility.
- The Health Sector Transformation Program, launched by the Ministry of Health in 2021, addresses genetic disorders through enhanced screening, genetic testing standards requiring SFDA approval for diagnostics, and compliance mandates for healthcare providers to integrate rare disease protocols, including Alpha 1 Antitrypsin Deficiency management in specialized centers. This initiative includes funding for research, public awareness campaigns, and the establishment of specialized clinics, which are expected to enhance patient outcomes and increase the overall market size.
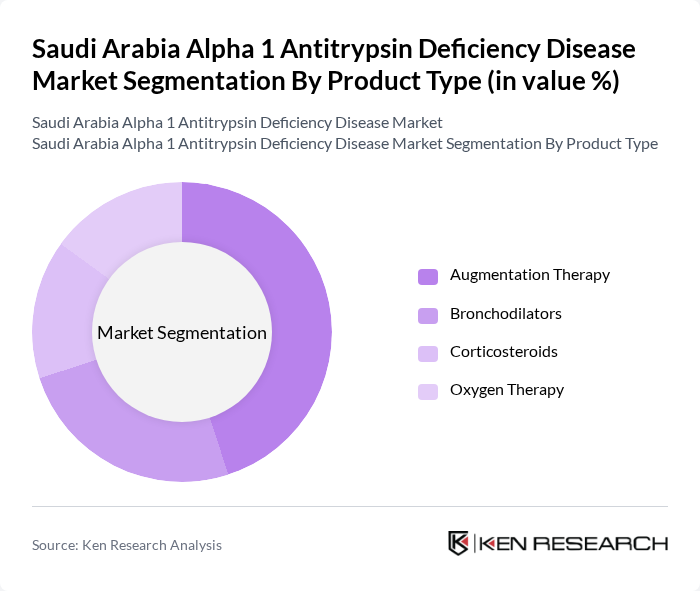
Saudi Arabia Alpha 1 Antitrypsin Deficiency Disease Market Segmentation
By Product Type:The product type segmentation includes Augmentation Therapy, Bronchodilators, Corticosteroids, and Oxygen Therapy. Among these, Augmentation Therapy is the leading subsegment due to its critical role in managing the deficiency by replacing the missing protein through intravenous infusion of purified alpha-1 antitrypsin to restore protective levels in the lungs. The increasing adoption of this therapy, driven by its effectiveness in slowing emphysema progression, improving lung function and quality of life, has solidified its dominance in the market.
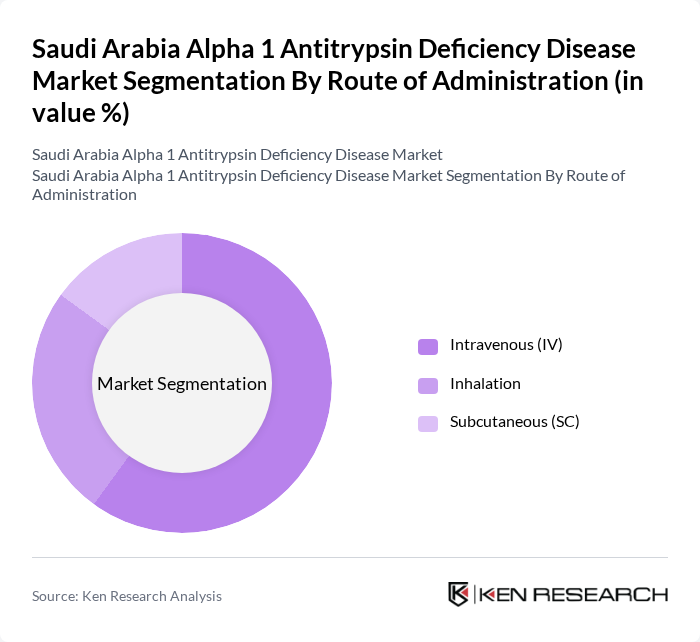
By Route of Administration:This segmentation includes Intravenous (IV), Inhalation, and Subcutaneous (SC) routes. The Intravenous (IV) route is the most prevalent due to its rapid absorption and effectiveness in delivering therapies directly into the bloodstream. This method is particularly favored in clinical settings for patients requiring immediate treatment, thus leading to its significant market share.
Saudi Arabia Alpha 1 Antitrypsin Deficiency Disease Market Competitive Landscape
The Saudi Arabia Alpha 1 Antitrypsin Deficiency Disease Market is characterized by a dynamic mix of regional and international players. Leading participants such as Grifols, CSL Behring, Takeda Pharmaceutical Company, AstraZeneca, Vertex Pharmaceuticals, Alnylam Pharmaceuticals, Shire (now part of Takeda), Biogen, AbbVie, Sanofi, Regeneron Pharmaceuticals, Amgen, Novartis, Roche, Merck & Co. contribute to innovation, geographic expansion, and service delivery in this space.
Saudi Arabia Alpha 1 Antitrypsin Deficiency Disease Market Industry Analysis
Growth Drivers
- Increasing Prevalence of Alpha 1 Antitrypsin Deficiency:The prevalence of Alpha 1 Antitrypsin Deficiency (AATD) in Saudi Arabia is estimated to be around 1 in 2,500 individuals, translating to approximately 13,000 affected people. This rising incidence is driven by genetic factors and lifestyle changes, necessitating increased healthcare resources and attention. The World Health Organization (WHO) has noted a growing need for specialized care, which is expected to boost the demand for diagnostic and therapeutic services in the region.
- Rising Awareness and Diagnosis Rates:Awareness campaigns and educational initiatives have led to a significant increase in diagnosis rates of AATD in Saudi Arabia. In future, it is projected that over 60% of healthcare providers will be familiar with AATD, up from 40% previously. This increase is supported by government health programs that aim to enhance early detection, thereby improving patient outcomes and driving demand for treatment options, as highlighted by the Saudi Ministry of Health.
- Advancements in Treatment Options:The introduction of new therapies, including enzyme replacement therapies and gene therapy, has transformed the treatment landscape for AATD. In future, the market is expected to see a 30% increase in the availability of innovative treatments, driven by collaborations between local pharmaceutical companies and international biotech firms. This advancement not only enhances patient care but also stimulates market growth by attracting investment in research and development.
Market Challenges
- Limited Access to Specialized Healthcare:Access to specialized healthcare services for AATD remains a significant challenge in Saudi Arabia. Approximately 70% of patients live in rural areas where specialized facilities are scarce. This geographical disparity limits timely diagnosis and treatment, leading to poorer health outcomes. The Saudi government is working to address this issue, but the current infrastructure still lags behind urban centers, impacting overall patient care.
- High Cost of Treatment:The financial burden of AATD treatment is substantial, with annual costs exceeding SAR 200,000 (approximately USD 53,000) per patient for advanced therapies. This high cost poses a barrier to access for many patients, particularly those without comprehensive health insurance. The Saudi healthcare system is under pressure to find sustainable solutions to subsidize these costs, which could hinder treatment uptake and overall market growth.
Saudi Arabia Alpha 1 Antitrypsin Deficiency Disease Market Future Outlook
The future of the Alpha 1 Antitrypsin Deficiency market in Saudi Arabia appears promising, driven by increasing healthcare investments and a focus on innovative treatment solutions. The government is expected to enhance healthcare infrastructure, facilitating better access to specialized services. Additionally, the rise of telemedicine is likely to improve patient engagement and follow-up care. As awareness continues to grow, the market is poised for significant advancements in both treatment options and patient support systems, fostering a more robust healthcare environment.
Market Opportunities
- Development of New Therapies:There is a significant opportunity for the development of new therapies targeting AATD, particularly gene therapies that could offer long-term solutions. With an estimated investment of SAR 500 million (approximately USD 133 million) in R&D, local biotech firms are well-positioned to innovate and meet the growing demand for effective treatments, potentially transforming patient care.
- Expansion of Healthcare Infrastructure:The Saudi government is investing heavily in healthcare infrastructure, with plans to allocate SAR 100 billion (approximately USD 26.6 billion) in future. This investment aims to enhance access to specialized care for AATD patients, particularly in underserved regions. Improved facilities and services will likely lead to better diagnosis and treatment rates, creating a favorable environment for market growth.
Scope of the Report
| Segment | Sub-Segments |
|---|---|
| By Product Type | Augmentation Therapy Bronchodilators Corticosteroids Oxygen Therapy |
| By Route of Administration | Intravenous (IV) Inhalation Subcutaneous (SC) |
| By Patient Age Group | Pediatric Adult |
| By Application/End-User | Hospitals Specialty Clinics Pharmacies |
| By Distribution Channel | Hospital Pharmacies Retail Pharmacies Online Pharmacies |
| By Disease Manifestation | Pulmonary Liver Others |
Key Target Audience
Investors and Venture Capitalist Firms
Government and Regulatory Bodies (e.g., Saudi Food and Drug Authority, Ministry of Health)
Pharmaceutical Manufacturers
Biotechnology Companies
Healthcare Providers and Hospitals
Patient Advocacy Groups
Health Insurance Companies
Clinical Research Organizations
Players Mentioned in the Report:
Grifols
CSL Behring
Takeda Pharmaceutical Company
AstraZeneca
Vertex Pharmaceuticals
Alnylam Pharmaceuticals
Shire (now part of Takeda)
Biogen
AbbVie
Sanofi
Regeneron Pharmaceuticals
Amgen
Novartis
Roche
Merck & Co.
Table of Contents
Market Assessment Phase
1. Executive Summary and Approach
2. Saudi Arabia Alpha 1 Antitrypsin Deficiency Disease Market Overview
2.1 Key Insights and Strategic Recommendations
2.2 Saudi Arabia Alpha 1 Antitrypsin Deficiency Disease Market Overview
2.3 Definition and Scope
2.4 Evolution of Market Ecosystem
2.5 Timeline of Key Regulatory Milestones
2.6 Value Chain & Stakeholder Mapping
2.7 Business Cycle Analysis
2.8 Policy & Incentive Landscape
3. Saudi Arabia Alpha 1 Antitrypsin Deficiency Disease Market Analysis
3.1 Growth Drivers
3.1.1 Increasing prevalence of Alpha 1 Antitrypsin Deficiency
3.1.2 Rising awareness and diagnosis rates
3.1.3 Advancements in treatment options
3.1.4 Supportive government policies and initiatives
3.2 Market Challenges
3.2.1 Limited access to specialized healthcare
3.2.2 High cost of treatment
3.2.3 Lack of awareness among healthcare providers
3.2.4 Regulatory hurdles in drug approval
3.3 Market Opportunities
3.3.1 Development of new therapies
3.3.2 Expansion of healthcare infrastructure
3.3.3 Collaborations with international organizations
3.3.4 Increasing investment in research and development
3.4 Market Trends
3.4.1 Growing focus on personalized medicine
3.4.2 Rise of telemedicine and digital health solutions
3.4.3 Increased patient advocacy and support groups
3.4.4 Shift towards preventive healthcare
3.5 Government Regulation
3.5.1 Regulatory frameworks for drug approval
3.5.2 Policies promoting research and innovation
3.5.3 Guidelines for patient access to treatments
3.5.4 Incentives for local manufacturing of therapies
4. SWOT Analysis
5. Stakeholder Analysis
6. Porter's Five Forces Analysis
7. Saudi Arabia Alpha 1 Antitrypsin Deficiency Disease Market Market Size, 2019-2024
7.1 By Value
7.2 By Volume
7.3 By Average Selling Price
8. Saudi Arabia Alpha 1 Antitrypsin Deficiency Disease Market Segmentation
8.1 By Product Type
8.1.1 Augmentation Therapy
8.1.2 Bronchodilators
8.1.3 Corticosteroids
8.1.4 Oxygen Therapy
8.2 By Route of Administration
8.2.1 Intravenous (IV)
8.2.2 Inhalation
8.2.3 Subcutaneous (SC)
8.3 By Patient Age Group
8.3.1 Pediatric
8.3.2 Adult
8.4 By Application/End-User
8.4.1 Hospitals
8.4.2 Specialty Clinics
8.4.3 Pharmacies
8.5 By Distribution Channel
8.5.1 Hospital Pharmacies
8.5.2 Retail Pharmacies
8.5.3 Online Pharmacies
8.6 By Disease Manifestation
8.6.1 Pulmonary
8.6.2 Liver
8.6.3 Others
9. Saudi Arabia Alpha 1 Antitrypsin Deficiency Disease Market Competitive Analysis
9.1 Market Share of Key Players
9.2 Cross Comparison of Key Players
9.2.1 Company Name
9.2.2 Market Revenue (USD Million)
9.2.3 Revenue Growth Rate (CAGR %)
9.2.4 Market Share (%)
9.2.5 Product Portfolio Breadth
9.2.6 Geographic Presence
9.2.7 Regulatory Approvals
9.2.8 R&D Investment Level
9.2.9 Distribution Network Strength
9.2.10 Strategic Partnerships
9.3 SWOT Analysis of Top Players
9.4 Pricing Analysis
9.5 Detailed Profile of Major Companies
9.5.1 Grifols
9.5.2 CSL Behring
9.5.3 Takeda Pharmaceutical Company
9.5.4 AstraZeneca
9.5.5 Vertex Pharmaceuticals
9.5.6 Alnylam Pharmaceuticals
9.5.7 Shire (now part of Takeda)
9.5.8 Biogen
9.5.9 AbbVie
9.5.10 Sanofi
9.5.11 Regeneron Pharmaceuticals
9.5.12 Amgen
9.5.13 Novartis
9.5.14 Roche
9.5.15 Merck & Co.
10. Saudi Arabia Alpha 1 Antitrypsin Deficiency Disease Market End-User Analysis
10.1 Procurement Behavior of Key Ministries
10.1.1 Budget Allocation for Rare Diseases
10.1.2 Procurement Processes
10.1.3 Collaboration with Healthcare Providers
10.1.4 Others
10.2 Corporate Spend on Infrastructure & Energy
10.2.1 Investment in Healthcare Facilities
10.2.2 Funding for Research Initiatives
10.2.3 Partnerships with Pharmaceutical Companies
10.2.4 Others
10.3 Pain Point Analysis by End-User Category
10.3.1 Access to Treatment
10.3.2 Affordability of Medications
10.3.3 Availability of Specialized Care
10.3.4 Others
10.4 User Readiness for Adoption
10.4.1 Awareness Levels
10.4.2 Training Needs for Healthcare Providers
10.4.3 Patient Education Programs
10.4.4 Others
10.5 Post-Deployment ROI and Use Case Expansion
10.5.1 Measurement of Treatment Outcomes
10.5.2 Long-term Cost Savings
10.5.3 Expansion of Treatment Protocols
10.5.4 Others
11. Saudi Arabia Alpha 1 Antitrypsin Deficiency Disease Market Future Size, 2025-2030
11.1 By Value
11.2 By Volume
11.3 By Average Selling Price
Go-To-Market Strategy Phase
1. Whitespace Analysis + Business Model Canvas
1.1 Identification of Market Gaps
1.2 Business Model Development
2. Marketing and Positioning Recommendations
2.1 Branding Strategies
2.2 Product USPs
3. Distribution Plan
3.1 Urban Retail vs Rural NGO Tie-ups
4. Channel & Pricing Gaps
4.1 Underserved Routes
4.2 Pricing Bands
5. Unmet Demand & Latent Needs
5.1 Category Gaps
5.2 Consumer Segments
6. Customer Relationship
6.1 Loyalty Programs
6.2 After-sales Service
7. Value Proposition
7.1 Sustainability
7.2 Integrated Supply Chains
8. Key Activities
8.1 Regulatory Compliance
8.2 Branding
8.3 Distribution Setup
9. Entry Strategy Evaluation
9.1 Domestic Market Entry Strategy
9.1.1 Product Mix
9.1.2 Pricing Band
9.1.3 Packaging
9.2 Export Entry Strategy
9.2.1 Target Countries
9.2.2 Compliance Roadmap
10. Entry Mode Assessment
10.1 JV
10.2 Greenfield
10.3 M&A
10.4 Distributor Model
11. Capital and Timeline Estimation
11.1 Capital Requirements
11.2 Timelines
12. Control vs Risk Trade-Off
12.1 Ownership vs Partnerships
13. Profitability Outlook
13.1 Breakeven Analysis
13.2 Long-term Sustainability
14. Potential Partner List
14.1 Distributors
14.2 JVs
14.3 Acquisition Targets
15. Execution Roadmap
15.1 Phased Plan for Market Entry
15.1.1 Market Setup
15.1.2 Market Entry
15.1.3 Growth Acceleration
15.1.4 Scale & Stabilize
15.2 Key Activities and Milestones
15.2.1 Milestone Planning
15.2.2 Activity Tracking
Research Methodology
Phase 1: Approach1
Desk Research
- Analysis of published epidemiological studies on Alpha 1 Antitrypsin Deficiency in Saudi Arabia
- Review of healthcare expenditure reports and market access documents from the Ministry of Health
- Examination of pharmaceutical market reports and health technology assessments relevant to AAT deficiency treatments
Primary Research
- Interviews with pulmonologists and hepatologists specializing in Alpha 1 Antitrypsin Deficiency
- Surveys with healthcare providers and pharmacists regarding treatment availability and patient demographics
- Focus groups with patient advocacy groups to understand patient experiences and treatment challenges
Validation & Triangulation
- Cross-validation of findings with international studies on AAT deficiency prevalence and treatment outcomes
- Triangulation of data from healthcare providers, patient interviews, and market reports
- Sanity checks through expert panel discussions with key opinion leaders in the field
Phase 2: Market Size Estimation1
Top-down Assessment
- Estimation of the total population affected by Alpha 1 Antitrypsin Deficiency based on national health statistics
- Analysis of treatment costs and healthcare resource allocation for managing AAT deficiency
- Incorporation of government health initiatives and funding for rare diseases in Saudi Arabia
Bottom-up Modeling
- Collection of data on the number of diagnosed patients and treatment regimens from healthcare facilities
- Estimation of market size based on the average cost of therapies and patient adherence rates
- Volume x cost analysis for different treatment modalities, including enzyme replacement therapy and supportive care
Forecasting & Scenario Analysis
- Multi-factor regression analysis incorporating demographic trends, healthcare access, and treatment advancements
- Scenario modeling based on potential changes in healthcare policy and insurance coverage for AAT deficiency treatments
- Baseline, optimistic, and pessimistic projections for market growth through 2030
Phase 3: CATI Sample Composition1
| Scope Item/Segment | Sample Size | Target Respondent Profiles |
|---|---|---|
| Healthcare Providers | 120 | Pulmonologists, Hepatologists, General Practitioners |
| Pharmaceutical Distributors | 80 | Sales Managers, Product Managers |
| Patient Advocacy Groups | 50 | Patient Representatives, Support Group Leaders |
| Health Insurance Companies | 60 | Policy Analysts, Claims Managers |
| Research Institutions | 40 | Clinical Researchers, Epidemiologists |
Frequently Asked Questions
What is the current market value of Alpha 1 Antitrypsin Deficiency Disease in Saudi Arabia?
The Saudi Arabia Alpha 1 Antitrypsin Deficiency Disease market is valued at approximately USD 25 million, reflecting a five-year historical analysis that highlights growth driven by increased awareness, advancements in diagnostics, and rising prevalence of related diseases.