Region:Middle East
Author(s):Geetanshi
Product Code:KRAA0547
Pages:92
Published On:December 2025
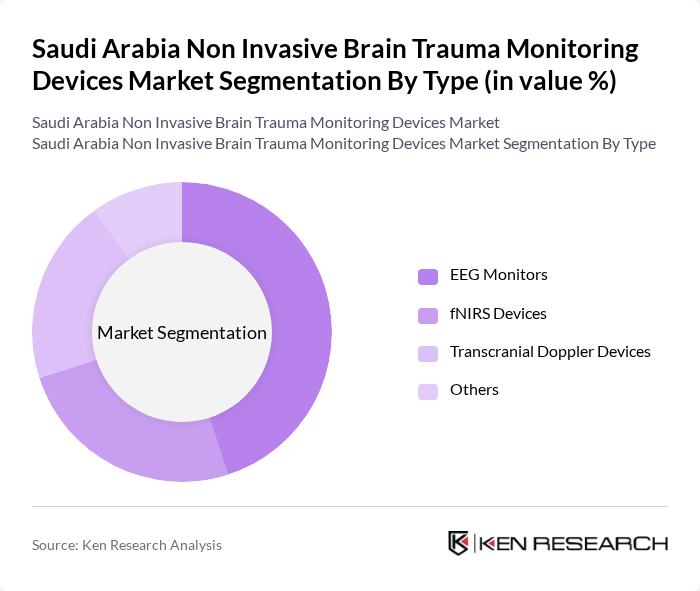
By Type:The market is segmented into various types of non-invasive brain trauma monitoring devices, including EEG monitors, fNIRS devices, transcranial Doppler devices, and others. Among these, EEG monitors are the most widely used due to their effectiveness in monitoring brain activity and diagnosing neurological conditions. The increasing prevalence of epilepsy and other neurological disorders has further propelled the demand for EEG monitors, making them a dominant force in the market.
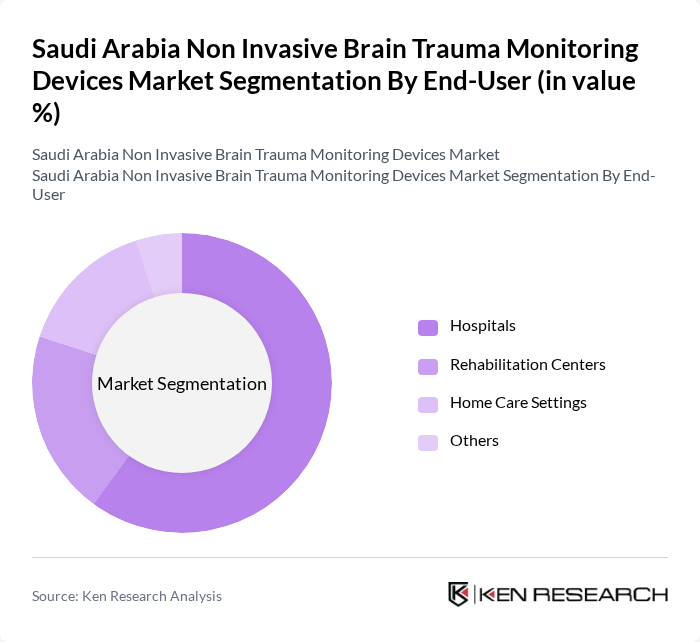
By End-User:The end-user segmentation includes hospitals, rehabilitation centers, home care settings, and others. Hospitals are the primary end-users of non-invasive brain trauma monitoring devices, driven by the need for advanced diagnostic tools in emergency and critical care settings. The increasing number of hospitals equipped with modern healthcare technologies has significantly contributed to the growth of this segment.
The Saudi Arabia Non Invasive Brain Trauma Monitoring Devices Market is characterized by a dynamic mix of regional and international players. Leading participants such as Medtronic, Philips Healthcare, Siemens Healthineers, GE Healthcare, Nihon Kohden Corporation, BrainScope Company, Inc., Natus Medical Incorporated, Ceribell, Inc., NeuroWave Systems, Inc., Compumedics Limited, Advanced Brain Monitoring, Inc., Cadwell Industries, Inc., EEG Info, Zeto, Inc., Cognionics, Inc. contribute to innovation, geographic expansion, and service delivery in this space.
The future of the non-invasive brain trauma monitoring devices market in Saudi Arabia appears promising, driven by ongoing digital healthcare transformation and substantial investments in data infrastructure. The government's commitment to integrating digital solutions into healthcare, exemplified by initiatives like the digital diabetes command center, indicates a readiness to adopt advanced monitoring technologies. Additionally, the supportive regulatory environment and emerging technology readiness in public sectors will likely foster innovation and collaboration, enhancing market growth opportunities in the coming years.
| Segment | Sub-Segments |
|---|---|
| By Type | EEG Monitors fNIRS Devices Transcranial Doppler Devices Others |
| By End-User | Hospitals Rehabilitation Centers Home Care Settings Others |
| By Application | Emergency Care Post-Surgery Monitoring Research and Development Others |
| By Distribution Channel | Direct Sales Online Sales Distributors Others |
| By Region | Central Region Eastern Region Western Region Southern Region |
| By Technology | Non-invasive Imaging Techniques Monitoring Software Data Analytics Tools Others |
| By Policy Support | Government Grants Tax Incentives Research Funding Others |
| Scope Item/Segment | Sample Size | Target Respondent Profiles |
|---|---|---|
| Neurosurgery Departments | 100 | Neurosurgeons, Clinical Directors |
| Emergency Care Units | 80 | Emergency Medicine Physicians, Trauma Coordinators |
| Medical Device Manufacturers | 60 | Product Managers, R&D Engineers |
| Healthcare Policy Makers | 50 | Health Administrators, Policy Analysts |
| Patient Advocacy Groups | 40 | Advocacy Leaders, Patient Representatives |
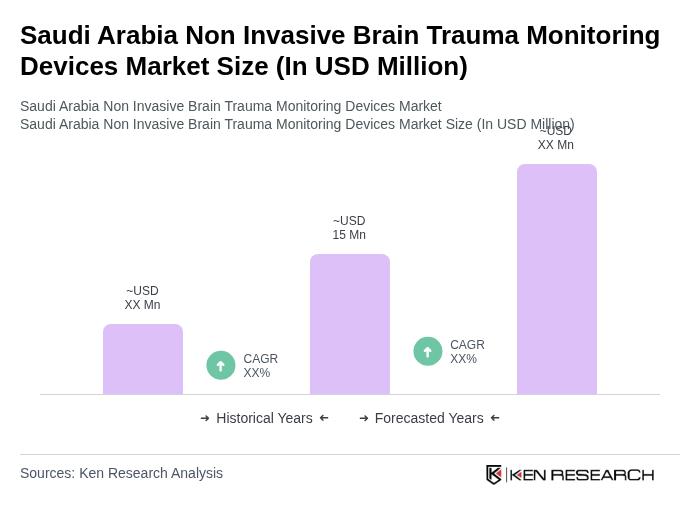
The Saudi Arabia Non Invasive Brain Trauma Monitoring Devices Market is valued at approximately USD 15 million, reflecting a growing demand driven by the increasing incidence of neurological disorders and advancements in healthcare infrastructure.