Region:Middle East
Author(s):Geetanshi
Product Code:KRAB1542
Pages:88
Published On:January 2026
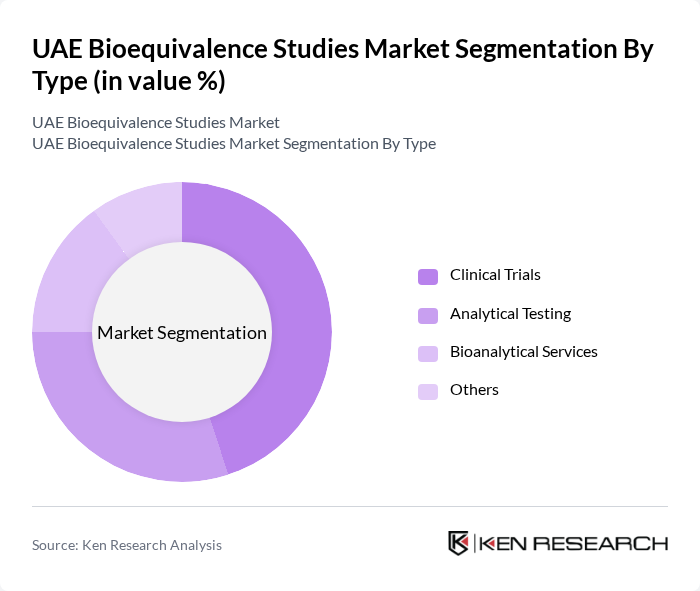
By Type:The market is segmented into Clinical Trials, Analytical Studies, Bioanalytical Services, and Others. Clinical Trials are the leading sub-segment, driven by the increasing number of new drug approvals and the growing demand for generic medications. Analytical Studies follow closely, as they are essential for ensuring the quality and efficacy of pharmaceutical products. Bioanalytical Services are also gaining traction due to the need for precise measurement of drug concentrations in biological samples.
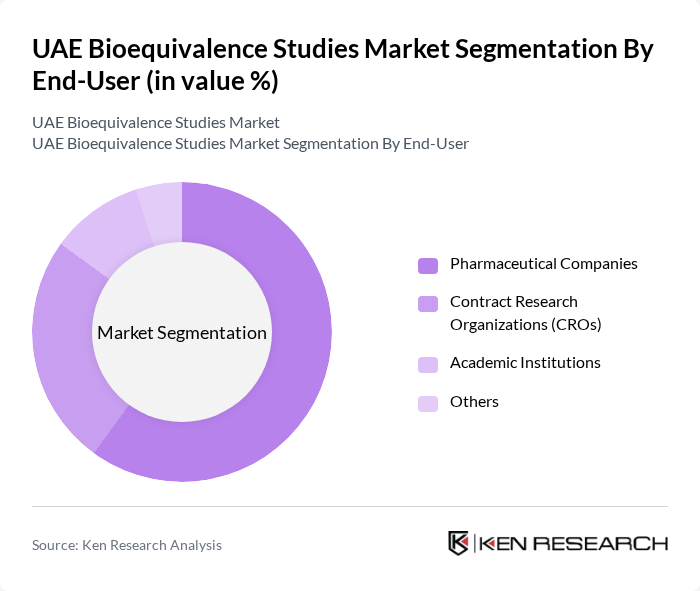
By End-User:The end-user segmentation includes Pharmaceutical Companies, Contract Research Organizations (CROs), Academic Institutions, and Others. Pharmaceutical Companies dominate this segment, as they are the primary sponsors of bioequivalence studies to support the development and approval of generic drugs. CROs are also significant players, providing specialized services to pharmaceutical companies and ensuring compliance with regulatory standards.
The UAE Bioequivalence Studies Market is characterized by a dynamic mix of regional and international players. Leading participants such as Abu Dhabi University, Gulf Pharmaceutical Industries (Julphar), Al Ain Pharmaceuticals, Dubai Science Park, Emirates Clinical Research Institute, Aster DM Healthcare, Mediclinic International, RAK Pharmaceuticals, United Arab Emirates University, Dubai Health Authority, Abu Dhabi Health Services Company (SEHA), Al Zahra Hospital, NMC Health, Burjeel Hospital, Cleveland Clinic Abu Dhabi contribute to innovation, geographic expansion, and service delivery in this space.
The UAE bioequivalence studies market is poised for significant growth, driven by increasing healthcare investments and a focus on enhancing local pharmaceutical capabilities. As the demand for generic drugs continues to rise, the government is likely to implement further supportive measures, including streamlined regulatory processes. Additionally, advancements in technology and a shift towards patient-centric approaches will shape the future landscape, fostering innovation and improving the efficiency of bioequivalence studies in the region.
| Segment | Sub-Segments |
|---|---|
| By Type | Clinical Trials Analytical Studies Bioanalytical Services Others |
| By End-User | Pharmaceutical Companies Contract Research Organizations (CROs) Academic Institutions Others |
| By Therapeutic Area | Cardiovascular Oncology Neurology Others |
| By Study Design | Single-Dose Studies Multiple-Dose Studies Others |
| By Region | Abu Dhabi Dubai Sharjah Others |
| By Service Provider Type | In-House Laboratories Outsourced Laboratories Others |
| By Funding Source | Government Funding Private Investment Grants and Subsidies Others |
| Scope Item/Segment | Sample Size | Target Respondent Profiles |
|---|---|---|
| Pharmaceutical Companies | 100 | Regulatory Affairs Managers, R&D Directors |
| Clinical Research Organizations | 50 | Project Managers, Clinical Operations Heads |
| Regulatory Bodies | 40 | Policy Makers, Compliance Officers |
| Healthcare Providers | 70 | Pharmacists, Medical Directors |
| Market Research Firms | 60 | Market Analysts, Business Development Managers |
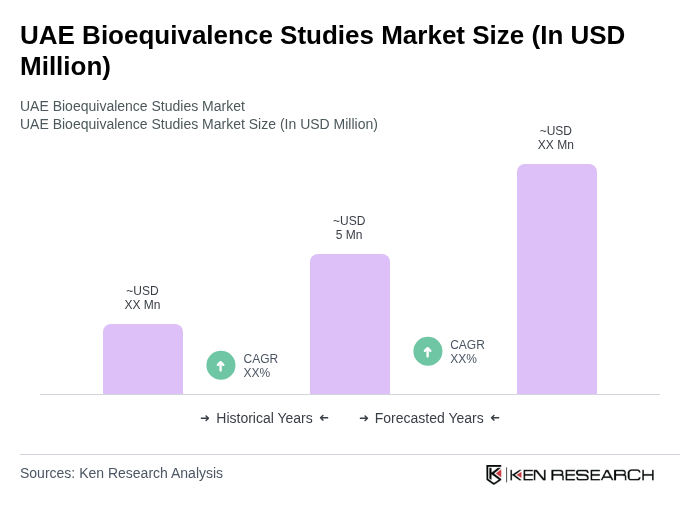
The UAE Bioequivalence Studies Market is valued at approximately USD 4 million, driven by the increasing demand for generic drugs and advancements in research methodologies and technologies that enhance the efficiency of bioequivalence studies.