About the Report
Base Year 2024UAE Ophthalmic Clinical Trials Market Overview
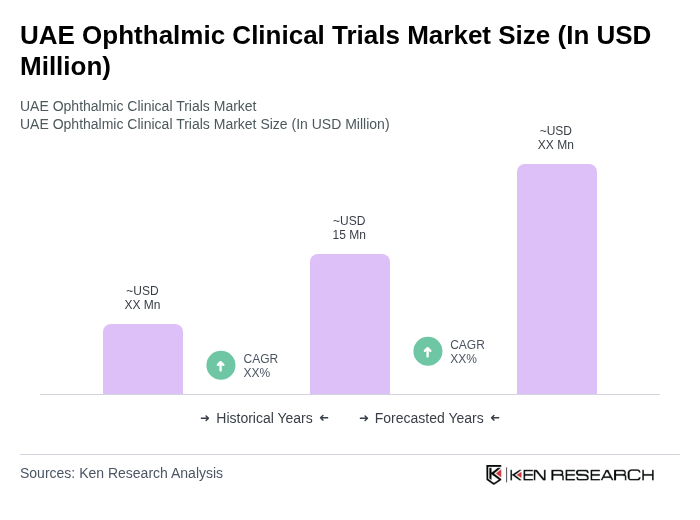
- The UAE ophthalmic clinical trials market is valued at USD 15 million, based on a five-year historical analysis. This growth is primarily driven by the increasing prevalence of chronic eye conditions such as diabetic retinopathy and cataracts, coupled with improving healthcare infrastructure and rising investments in healthcare facilities that enable the conduct of more clinical trials. These factors are bolstered by enhanced patient recruitment capabilities and public–private partnerships in ophthalmic research.
- Key cities dominating the market include Dubai and Abu Dhabi, which are recognized for their advanced healthcare systems and state-of-the-art medical facilities. The presence of leading global clinical research firms and major ophthalmic device manufacturers in these cities further strengthens their position, making them attractive locations for conducting clinical trials.
- A significant regulatory development enhancing the market is the UAE Ministry of Health’s new framework to measure health-sector R&D expenditure. This initiative promotes transparency, cooperation, and strategic investment in clinical research infrastructure, including ophthalmology, facilitating a more systematic allocation of resources and strengthening the ecosystem for conducting trials.
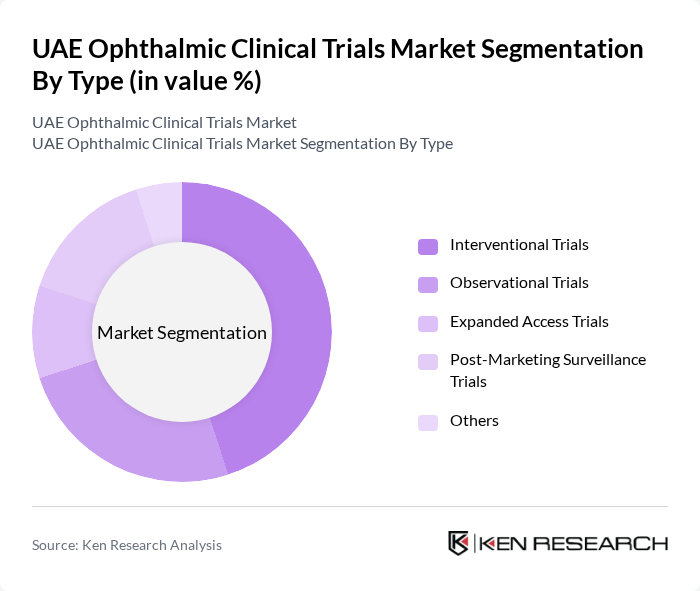
UAE Ophthalmic Clinical Trials Market Segmentation
By Type:The market is segmented into various types of clinical trials, including interventional trials, observational trials, expanded access trials, post-marketing surveillance trials, and others. Interventional trials are currently the dominant segment due to their critical role in testing new treatments and therapies, which are essential for advancing ophthalmic care. The increasing number of innovative therapies and the need for regulatory approval drive the demand for these trials.
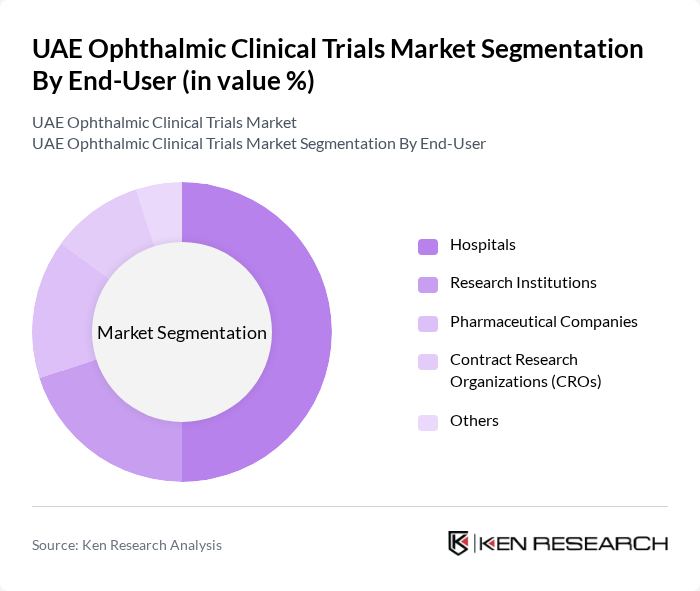
By End-User:The end-user segmentation includes hospitals, research institutions, pharmaceutical companies, contract research organizations (CROs), and others. Hospitals are the leading end-user segment, as they are often the primary sites for conducting clinical trials. The increasing collaboration between hospitals and pharmaceutical companies for research purposes is a significant factor contributing to this dominance.
UAE Ophthalmic Clinical Trials Market Competitive Landscape
The UAE Ophthalmic Clinical Trials Market is characterized by a dynamic mix of regional and international players. Leading participants such as Novartis AG, Alcon Inc., Bayer AG, Johnson & Johnson Vision, AbbVie Inc., Regeneron Pharmaceuticals, Santen Pharmaceutical Co., Ltd., Zeiss Group, Bausch + Lomb, EyePoint Pharmaceuticals, Aerie Pharmaceuticals, Ocular Therapeutix, Eyenovia, Inc., Genentech, Inc., Allergan plc contribute to innovation, geographic expansion, and service delivery in this space.
UAE Ophthalmic Clinical Trials Market Industry Analysis
Growth Drivers
- Increasing Prevalence of Eye Diseases:The UAE has witnessed a significant rise in eye diseases, with an estimated 1.5 million people affected by diabetic retinopathy alone in future. The World Health Organization reports that the prevalence of myopia is expected to reach 50% of the population by future. This growing burden of eye conditions drives the demand for clinical trials, as pharmaceutical companies seek to develop effective treatments and interventions tailored to the local population's needs.
- Advancements in Ophthalmic Research Technologies:The UAE is at the forefront of adopting cutting-edge technologies in ophthalmology, with investments exceeding AED 1 billion in research and development in future. Innovations such as gene therapy and advanced imaging techniques are enhancing the efficiency of clinical trials. These advancements not only improve patient outcomes but also attract global research organizations to conduct trials in the UAE, further stimulating market growth.
- Government Support for Clinical Research:The UAE government has allocated AED 500 million to support clinical research initiatives in future, aiming to position the country as a regional hub for medical research. This funding facilitates partnerships between public and private sectors, encouraging local institutions to engage in ophthalmic clinical trials. Additionally, streamlined regulatory processes are being implemented to expedite trial approvals, fostering a conducive environment for research and innovation.
Market Challenges
- Regulatory Hurdles and Compliance Issues:Navigating the regulatory landscape in the UAE can be complex, with stringent requirements for clinical trial approvals. In future, the average time for obtaining necessary approvals is approximately 6-12 months, which can delay research timelines. Compliance with international standards, such as Good Clinical Practice (GCP), adds another layer of complexity, potentially deterring some organizations from conducting trials in the region.
- High Costs Associated with Clinical Trials:The financial burden of conducting clinical trials in the UAE is significant, with costs averaging AED 2 million per trial in future. This includes expenses related to patient recruitment, site management, and regulatory compliance. The high cost can limit the number of trials initiated, particularly for smaller companies or startups, thereby constraining the overall growth of the ophthalmic clinical trials market in the region.
UAE Ophthalmic Clinical Trials Market Future Outlook
The future of the UAE ophthalmic clinical trials market appears promising, driven by technological advancements and increased government investment. The integration of artificial intelligence in trial designs is expected to enhance patient recruitment and data analysis, while the rise of decentralized trials will facilitate broader participation. As the healthcare infrastructure continues to improve, the UAE is likely to attract more international collaborations, positioning itself as a leader in ophthalmic research and innovation in the region.
Market Opportunities
- Expansion of Telemedicine in Ophthalmology:The growing adoption of telemedicine in the UAE presents a unique opportunity for ophthalmic clinical trials. With over 70% of the population using smartphones, remote consultations can facilitate patient recruitment and follow-up, reducing logistical challenges and costs associated with traditional trial methods.
- Collaborations with International Research Organizations:The UAE's strategic location and favorable regulatory environment make it an attractive destination for international research collaborations. By partnering with global organizations, local institutions can leverage expertise and resources, enhancing the quality and scope of ophthalmic clinical trials conducted in the region.
Scope of the Report
| Segment | Sub-Segments |
|---|---|
| By Type | Interventional Trials Observational Trials Expanded Access Trials Post-Marketing Surveillance Trials Others |
| By End-User | Hospitals Research Institutions Pharmaceutical Companies Contract Research Organizations (CROs) Others |
| By Patient Demographics | Age Groups Gender Geographic Distribution Others |
| By Therapeutic Area | Glaucoma Cataracts Retinal Disorders Corneal Diseases Others |
| By Phase of Trial | Phase I Phase II Phase III Phase IV Others |
| By Funding Source | Government Funding Private Funding Non-Profit Organizations Others |
| By Study Design | Randomized Controlled Trials Non-Randomized Trials Cross-Sectional Studies Others |
Key Target Audience
Investors and Venture Capitalist Firms
Government and Regulatory Bodies (e.g., Ministry of Health and Prevention, Emirates Health Services)
Pharmaceutical Companies
Medical Device Manufacturers
Clinical Research Organizations (CROs)
Healthcare Providers and Hospitals
Health Insurance Companies
Biotechnology Firms
Players Mentioned in the Report:
Novartis AG
Alcon Inc.
Bayer AG
Johnson & Johnson Vision
AbbVie Inc.
Regeneron Pharmaceuticals
Santen Pharmaceutical Co., Ltd.
Zeiss Group
Bausch + Lomb
EyePoint Pharmaceuticals
Aerie Pharmaceuticals
Ocular Therapeutix
Eyenovia, Inc.
Genentech, Inc.
Allergan plc
Table of Contents
Market Assessment Phase
1. Executive Summary and Approach
2. UAE Ophthalmic Clinical Trials Market Overview
2.1 Key Insights and Strategic Recommendations
2.2 UAE Ophthalmic Clinical Trials Market Overview
2.3 Definition and Scope
2.4 Evolution of Market Ecosystem
2.5 Timeline of Key Regulatory Milestones
2.6 Value Chain & Stakeholder Mapping
2.7 Business Cycle Analysis
2.8 Policy & Incentive Landscape
3. UAE Ophthalmic Clinical Trials Market Analysis
3.1 Growth Drivers
3.1.1 Increasing prevalence of eye diseases
3.1.2 Advancements in ophthalmic research technologies
3.1.3 Government support for clinical research
3.1.4 Rising demand for innovative treatment options
3.2 Market Challenges
3.2.1 Regulatory hurdles and compliance issues
3.2.2 High costs associated with clinical trials
3.2.3 Limited patient recruitment and retention
3.2.4 Competition from established markets
3.3 Market Opportunities
3.3.1 Expansion of telemedicine in ophthalmology
3.3.2 Collaborations with international research organizations
3.3.3 Growth in personalized medicine approaches
3.3.4 Increasing investment in healthcare infrastructure
3.4 Market Trends
3.4.1 Shift towards patient-centric trial designs
3.4.2 Integration of artificial intelligence in research
3.4.3 Rise of decentralized clinical trials
3.4.4 Focus on real-world evidence in ophthalmic studies
3.5 Government Regulation
3.5.1 Clinical trial registration requirements
3.5.2 Ethical guidelines for patient consent
3.5.3 Data protection regulations
3.5.4 Approval processes for new ophthalmic drugs
4. SWOT Analysis
5. Stakeholder Analysis
6. Porter's Five Forces Analysis
7. UAE Ophthalmic Clinical Trials Market Market Size, 2019-2024
7.1 By Value
7.2 By Volume
7.3 By Average Selling Price
8. UAE Ophthalmic Clinical Trials Market Segmentation
8.1 By Type
8.1.1 Interventional Trials
8.1.2 Observational Trials
8.1.3 Expanded Access Trials
8.1.4 Post-Marketing Surveillance Trials
8.1.5 Others
8.2 By End-User
8.2.1 Hospitals
8.2.2 Research Institutions
8.2.3 Pharmaceutical Companies
8.2.4 Contract Research Organizations (CROs)
8.2.5 Others
8.3 By Patient Demographics
8.3.1 Age Groups
8.3.2 Gender
8.3.3 Geographic Distribution
8.3.4 Others
8.4 By Therapeutic Area
8.4.1 Glaucoma
8.4.2 Cataracts
8.4.3 Retinal Disorders
8.4.4 Corneal Diseases
8.4.5 Others
8.5 By Phase of Trial
8.5.1 Phase I
8.5.2 Phase II
8.5.3 Phase III
8.5.4 Phase IV
8.5.5 Others
8.6 By Funding Source
8.6.1 Government Funding
8.6.2 Private Funding
8.6.3 Non-Profit Organizations
8.6.4 Others
8.7 By Study Design
8.7.1 Randomized Controlled Trials
8.7.2 Non-Randomized Trials
8.7.3 Cross-Sectional Studies
8.7.4 Others
9. UAE Ophthalmic Clinical Trials Market Competitive Analysis
9.1 Market Share of Key Players
9.2 Cross Comparison of Key Players
9.2.1 Company Name
9.2.2 Group Size (Large, Medium, or Small as per industry convention)
9.2.3 Revenue Growth Rate
9.2.4 Market Penetration Rate
9.2.5 Customer Retention Rate
9.2.6 Clinical Trial Success Rate
9.2.7 Pricing Strategy
9.2.8 Research and Development Investment
9.2.9 Regulatory Compliance Rate
9.2.10 Patient Recruitment Efficiency
9.3 SWOT Analysis of Top Players
9.4 Pricing Analysis
9.5 Detailed Profile of Major Companies
9.5.1 Novartis AG
9.5.2 Alcon Inc.
9.5.3 Bayer AG
9.5.4 Johnson & Johnson Vision
9.5.5 AbbVie Inc.
9.5.6 Regeneron Pharmaceuticals
9.5.7 Santen Pharmaceutical Co., Ltd.
9.5.8 Zeiss Group
9.5.9 Bausch + Lomb
9.5.10 EyePoint Pharmaceuticals
9.5.11 Aerie Pharmaceuticals
9.5.12 Ocular Therapeutix
9.5.13 Eyenovia, Inc.
9.5.14 Genentech, Inc.
9.5.15 Allergan plc
10. UAE Ophthalmic Clinical Trials Market End-User Analysis
10.1 Procurement Behavior of Key Ministries
10.1.1 Ministry of Health and Prevention
10.1.2 Ministry of Education
10.1.3 Ministry of Community Development
10.1.4 Others
10.2 Corporate Spend on Infrastructure & Energy
10.2.1 Investment in Clinical Research Facilities
10.2.2 Funding for Technological Upgrades
10.2.3 Budget Allocation for Training Programs
10.2.4 Others
10.3 Pain Point Analysis by End-User Category
10.3.1 Hospitals
10.3.2 Research Institutions
10.3.3 Pharmaceutical Companies
10.3.4 Others
10.4 User Readiness for Adoption
10.4.1 Awareness of Clinical Trials
10.4.2 Accessibility of Information
10.4.3 Willingness to Participate
10.4.4 Others
10.5 Post-Deployment ROI and Use Case Expansion
10.5.1 Evaluation of Clinical Outcomes
10.5.2 Cost-Benefit Analysis
10.5.3 Opportunities for Further Research
10.5.4 Others
11. UAE Ophthalmic Clinical Trials Market Future Size, 2025-2030
11.1 By Value
11.2 By Volume
11.3 By Average Selling Price
Go-To-Market Strategy Phase
1. Whitespace Analysis + Business Model Canvas
1.1 Market Gaps Identification
1.2 Value Proposition Development
1.3 Revenue Streams Analysis
1.4 Cost Structure Evaluation
1.5 Key Partnerships Exploration
1.6 Customer Segmentation
1.7 Channels of Distribution
2. Marketing and Positioning Recommendations
2.1 Branding Strategies
2.2 Product USPs
2.3 Target Audience Identification
2.4 Communication Strategy
2.5 Digital Marketing Approaches
2.6 Event Marketing Plans
2.7 Feedback Mechanisms
3. Distribution Plan
3.1 Urban Retail Strategies
3.2 Rural NGO Tie-Ups
3.3 Online Distribution Channels
3.4 Partnerships with Healthcare Providers
3.5 Logistics and Supply Chain Management
3.6 Inventory Management Solutions
3.7 Customer Service Framework
4. Channel & Pricing Gaps
4.1 Underserved Routes
4.2 Pricing Bands Analysis
4.3 Competitor Pricing Strategies
4.4 Customer Willingness to Pay
4.5 Value-Based Pricing Models
4.6 Discount and Promotion Strategies
4.7 Pricing Adjustments for Market Entry
5. Unmet Demand & Latent Needs
5.1 Category Gaps Identification
5.2 Consumer Segments Analysis
5.3 Emerging Trends in Consumer Preferences
5.4 Innovations in Product Offerings
5.5 Market Research Insights
5.6 Future Demand Projections
5.7 Strategic Recommendations
6. Customer Relationship
6.1 Loyalty Programs
6.2 After-Sales Service
6.3 Customer Feedback Mechanisms
6.4 Community Engagement Initiatives
6.5 Customer Education Programs
6.6 Relationship Management Tools
6.7 Retention Strategies
7. Value Proposition
7.1 Sustainability Initiatives
7.2 Integrated Supply Chains
7.3 Unique Selling Points
7.4 Customer-Centric Innovations
7.5 Competitive Advantages
7.6 Market Differentiation Strategies
7.7 Long-Term Value Creation
8. Key Activities
8.1 Regulatory Compliance
8.2 Branding Initiatives
8.3 Distribution Setup
8.4 Training and Development
8.5 Market Research Activities
8.6 Stakeholder Engagement
8.7 Performance Monitoring
9. Entry Strategy Evaluation
9.1 Domestic Market Entry Strategy
9.1.1 Product Mix
9.1.2 Pricing Band
9.1.3 Packaging
9.2 Export Entry Strategy
9.2.1 Target Countries
9.2.2 Compliance Roadmap
10. Entry Mode Assessment
10.1 Joint Ventures
10.2 Greenfield Investments
10.3 Mergers & Acquisitions
10.4 Distributor Model
10.5 Risk Assessment
10.6 Strategic Fit Evaluation
10.7 Long-Term Partnership Opportunities
11. Capital and Timeline Estimation
11.1 Capital Requirements
11.2 Timelines for Implementation
11.3 Financial Projections
11.4 Funding Sources
11.5 Budget Allocation
11.6 Cost Management Strategies
11.7 Risk Mitigation Plans
12. Control vs Risk Trade-Off
12.1 Ownership vs Partnerships
12.2 Risk Assessment Framework
12.3 Control Mechanisms
12.4 Partnership Evaluation Criteria
12.5 Long-Term Strategic Goals
12.6 Flexibility in Operations
12.7 Exit Strategies
13. Profitability Outlook
13.1 Breakeven Analysis
13.2 Long-Term Sustainability
13.3 Profit Margin Projections
13.4 Revenue Growth Strategies
13.5 Cost Reduction Initiatives
13.6 Financial Health Monitoring
13.7 Investment Return Analysis
14. Potential Partner List
14.1 Distributors
14.2 Joint Ventures
14.3 Acquisition Targets
14.4 Strategic Alliances
14.5 Research Collaborations
14.6 Technology Partners
14.7 Funding Partners
15. Execution Roadmap
15.1 Phased Plan for Market Entry
15.1.1 Market Setup
15.1.2 Market Entry
15.1.3 Growth Acceleration
15.1.4 Scale & Stabilize
15.2 Key Activities and Milestones
15.2.1 Timeline for Key Activities
15.2.2 Milestone Tracking
15.2.3 Performance Metrics
15.2.4 Adjustments and Revisions
Research Methodology
Phase 1: Approach1
Desk Research
- Analysis of published clinical trial registries and databases specific to ophthalmology in the UAE
- Review of market reports and white papers from healthcare organizations and regulatory bodies
- Examination of academic journals and publications focusing on ophthalmic research and clinical outcomes
Primary Research
- Interviews with ophthalmologists and clinical trial coordinators in leading UAE hospitals
- Surveys targeting pharmaceutical companies involved in ophthalmic drug development
- Focus groups with patients who have participated in ophthalmic clinical trials
Validation & Triangulation
- Cross-validation of findings through multiple expert interviews and stakeholder feedback
- Triangulation of data from clinical trial outcomes, patient demographics, and market trends
- Sanity checks through consultations with regulatory experts and industry analysts
Phase 2: Market Size Estimation1
Top-down Assessment
- Estimation of the total addressable market based on healthcare expenditure in the UAE
- Segmentation of the market by therapeutic areas, including glaucoma, cataracts, and retinal diseases
- Incorporation of government healthcare initiatives and funding for ophthalmic research
Bottom-up Modeling
- Collection of data on the number of ongoing and completed ophthalmic clinical trials in the UAE
- Estimation of average costs associated with conducting clinical trials in the ophthalmic sector
- Analysis of patient recruitment rates and retention strategies employed by clinical trial sponsors
Forecasting & Scenario Analysis
- Development of market forecasts based on historical growth rates and emerging trends in ophthalmology
- Scenario analysis considering regulatory changes, technological advancements, and demographic shifts
- Creation of baseline, optimistic, and pessimistic projections for the ophthalmic clinical trials market through 2030
Phase 3: CATI Sample Composition1
| Scope Item/Segment | Sample Size | Target Respondent Profiles |
|---|---|---|
| Ophthalmic Clinical Trial Coordinators | 100 | Clinical Research Associates, Trial Managers |
| Ophthalmologists in Private Practice | 80 | Eye Care Specialists, Medical Directors |
| Pharmaceutical Companies Conducting Trials | 70 | Clinical Development Managers, Regulatory Affairs Specialists |
| Patients Participating in Trials | 60 | Trial Participants, Patient Advocates |
| Healthcare Policy Makers | 50 | Health Economists, Policy Analysts |
Frequently Asked Questions
What is the current value of the UAE ophthalmic clinical trials market?
The UAE ophthalmic clinical trials market is valued at approximately USD 15 million, reflecting a five-year historical analysis. This growth is driven by the rising prevalence of chronic eye conditions and improvements in healthcare infrastructure.