About the Report
Base Year 2024Vietnam Ophthalmic Clinical Trials Market Overview
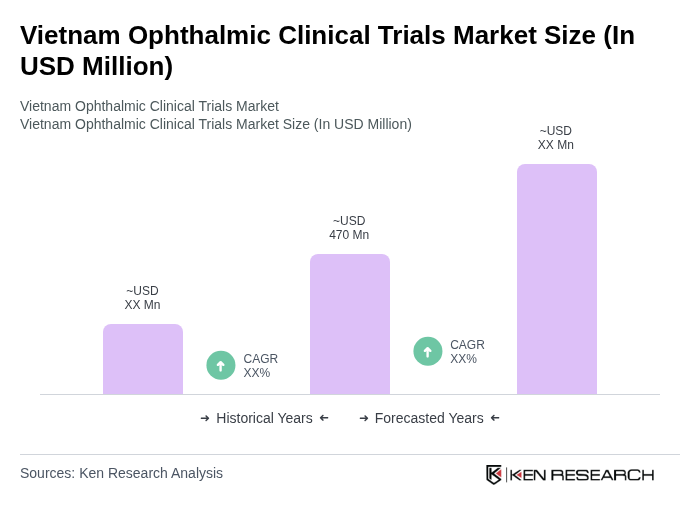
- The Vietnam Ophthalmic Clinical Trials Market is valued at USD 470 million, based on a five-year historical analysis. This growth is primarily driven by the increasing prevalence of eye diseases, rapid advancements in ophthalmic drug formulations (such as anti-VEGF and anti-glaucoma therapies), and the rising demand for innovative treatment options. The market is further supported by the expansion of clinical research organizations, the adoption of advanced surgical technologies, and the government's ongoing investment in healthcare infrastructure .
- Key cities dominating the market include Ho Chi Minh City and Hanoi, which are the primary hubs for healthcare and clinical research in Vietnam. These cities benefit from a high concentration of hospitals, research institutions, and pharmaceutical companies, facilitating collaboration and innovation in ophthalmic clinical trials. The urban population's increasing awareness of eye health, coupled with greater access to specialized care, further drives demand for clinical trials in these regions .
- In 2023, the Vietnamese government implemented Circular No. 29/2023/TT-BYT issued by the Ministry of Health, which mandates that all clinical trials must adhere to Good Clinical Practice (GCP) guidelines and requires the establishment of an independent ethics committee for trial approval. This regulation aims to ensure patient safety, enhance transparency, and improve the credibility of clinical research in the country by setting clear operational standards and compliance requirements for all stakeholders.
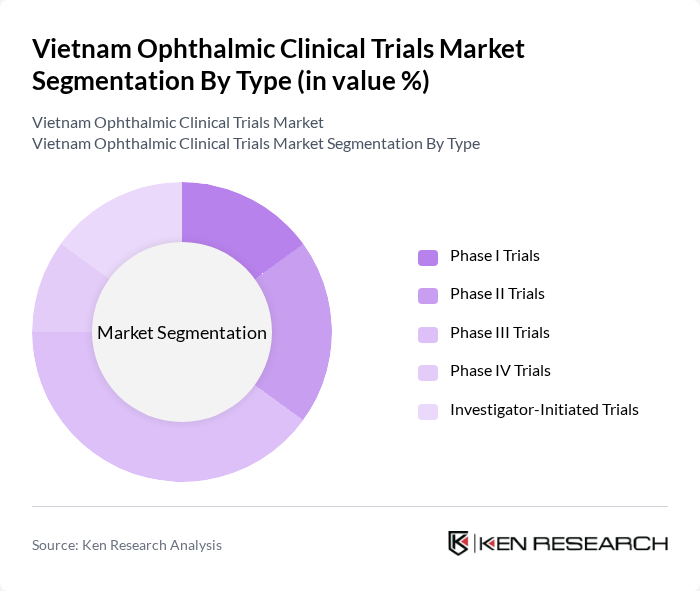
Vietnam Ophthalmic Clinical Trials Market Segmentation
By Type:The market is segmented into various types of clinical trials, including Phase I, II, III, IV, and Investigator-Initiated Trials. Among these, Phase III Trials dominate the market due to their critical role in evaluating the efficacy and safety of new treatments before regulatory approval. The increasing number of innovative therapies entering the market, particularly in retinal and glaucoma management, has led to a surge in Phase III Trials, making them a focal point for both sponsors and researchers .
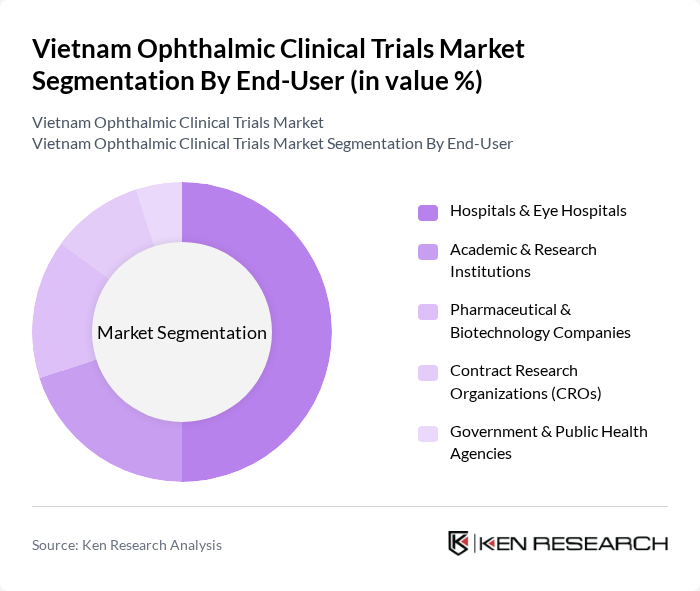
By End-User:The end-user segment includes Hospitals & Eye Hospitals, Academic & Research Institutions, Pharmaceutical & Biotechnology Companies, Contract Research Organizations (CROs), and Government & Public Health Agencies. Hospitals & Eye Hospitals are the leading end-users, as they are often the primary sites for conducting clinical trials. Their established infrastructure, access to large patient populations, and specialized ophthalmic care capabilities make them essential for the successful execution of ophthalmic clinical trials. Academic and research institutions are also increasingly involved in early-stage and investigator-initiated studies, reflecting the growing emphasis on translational research in Vietnam .
Vietnam Ophthalmic Clinical Trials Market Competitive Landscape
The Vietnam Ophthalmic Clinical Trials Market is characterized by a dynamic mix of regional and international players. Leading participants such as Novartis Vietnam, Roche Vietnam, Bayer Vietnam, Alcon Vietnam, Johnson & Johnson Vision Vietnam, Merck Sharp & Dohme (MSD) Vietnam, Santen Pharmaceutical Vietnam, Hoya Corporation Vietnam, Bausch + Lomb Vietnam, DND International Eye Hospital, Vietnam National Institute of Ophthalmology (VNIO), Eye Hospital of Ho Chi Minh City, Hanoi Eye Hospital, Allergan Vietnam, and Eyenovia Vietnam contribute to innovation, geographic expansion, and service delivery in this space.
Vietnam Ophthalmic Clinical Trials Market Industry Analysis
Growth Drivers
- Increasing Prevalence of Eye Diseases:The prevalence of eye diseases in Vietnam is rising, with an estimated3.5 million people suffering from various forms of visual impairmentas of in future. This increase is driven by factors such as an aging population, withover 10% of the population aged 60 and above, and lifestyle changes. The World Health Organization reports that by 2024, the number of individuals affected by age-related eye diseases could increase by20%, creating a pressing need for clinical trials focused on innovative treatments.
- Rising Investment in Healthcare Infrastructure:Vietnam's healthcare expenditure is projected to reach approximatelyUSD 20 billionin future, reflecting a significant increase fromUSD 15 billionin previous years. This investment is aimed at enhancing healthcare facilities and services, including ophthalmology. The government has allocatedUSD 1.5 billionspecifically for healthcare infrastructure improvements, which will facilitate the establishment of advanced clinical trial centers, thereby promoting the growth of the ophthalmic clinical trials market.
- Growing Demand for Innovative Ophthalmic Treatments:The demand for innovative treatments in ophthalmology is surging, with the market for ophthalmic drugs expected to reachUSD 1.2 billionin future. This growth is fueled by advancements in technology and an increasing focus on personalized medicine. The introduction of new therapies, such as gene therapy and biologics, is anticipated to drive clinical trials, as healthcare providers seek effective solutions for complex eye diseases, thus expanding the clinical trials landscape in Vietnam.
Market Challenges
- Limited Awareness of Clinical Trials Among Patients:A significant barrier to the growth of the ophthalmic clinical trials market in Vietnam is the limited awareness among patients regarding the benefits and availability of clinical trials. According to a survey, only30% of patients were aware of ongoing clinical trials in their area. This lack of awareness can lead to lower enrollment rates, hindering the progress of essential research and development in ophthalmology.
- Regulatory Hurdles and Lengthy Approval Processes:The regulatory landscape in Vietnam presents challenges for clinical trials, with approval processes often taking up to12 months. The Vietnam Ministry of Health has reported that the average time for trial approval can delay research initiatives significantly. These lengthy processes can deter international sponsors from conducting trials in Vietnam, limiting the market's growth potential and access to innovative treatments.
Vietnam Ophthalmic Clinical Trials Market Future Outlook
The future of the Vietnam ophthalmic clinical trials market appears promising, driven by technological advancements and increased collaboration with international research organizations. As the healthcare sector continues to evolve, the integration of digital health technologies will enhance patient engagement and streamline trial processes. Furthermore, the emphasis on real-world evidence will likely shape trial designs, ensuring that they are more patient-centric and relevant to the local population's needs, ultimately fostering innovation in ophthalmic treatments.
Market Opportunities
- Expansion of Telemedicine in Ophthalmology:The rise of telemedicine presents a significant opportunity for ophthalmic clinical trials in Vietnam. Withover 75% of the population having access to the internet, telemedicine can facilitate remote patient monitoring and consultations, increasing trial participation rates. This approach can enhance patient recruitment and retention, making clinical trials more accessible to a broader demographic.
- Collaborations with International Research Organizations:Collaborating with international research organizations can provide Vietnamese clinical trial sponsors access to advanced methodologies and funding. Such partnerships can enhance the quality of trials and attract foreign investment, which is crucial for developing innovative ophthalmic treatments. By leveraging global expertise, Vietnam can position itself as a competitive player in the ophthalmic clinical trials market.
Scope of the Report
| Segment | Sub-Segments |
|---|---|
| By Type | Phase I Trials Phase II Trials Phase III Trials Phase IV Trials Investigator-Initiated Trials |
| By End-User | Hospitals & Eye Hospitals Academic & Research Institutions Pharmaceutical & Biotechnology Companies Contract Research Organizations (CROs) Government & Public Health Agencies |
| By Therapeutic Area | Glaucoma Cataract Retinal Disorders (including Diabetic Retinopathy, AMD) Corneal Diseases Refractive Errors Uveitis & Inflammatory Diseases Others |
| By Study Design | Randomized Controlled Trials (RCTs) Observational Studies Open-Label Studies Single-Arm Trials Others |
| By Patient Demographics | Adults (18-64 years) Pediatrics (0-17 years) Geriatrics (65+ years) Others |
| By Funding Source | Government Funding Private/Industry Funding International Organizations & NGOs Academic Grants Others |
| By Geographic Focus | Urban Areas (e.g., Ho Chi Minh City, Hanoi, Da Nang) Rural Areas Others |
Key Target Audience
Investors and Venture Capitalist Firms
Government and Regulatory Bodies (e.g., Ministry of Health, Vietnam Food and Drug Administration)
Pharmaceutical Companies
Medical Device Manufacturers
Clinical Research Organizations (CROs)
Healthcare Providers and Hospitals
Industry Associations (e.g., Vietnam Ophthalmology Society)
Health Insurance Companies
Players Mentioned in the Report:
Novartis Vietnam
Roche Vietnam
Bayer Vietnam
Alcon Vietnam
Johnson & Johnson Vision Vietnam
Merck Sharp & Dohme (MSD) Vietnam
Santen Pharmaceutical Vietnam
Hoya Corporation Vietnam
Bausch + Lomb Vietnam
DND International Eye Hospital
Vietnam National Institute of Ophthalmology (VNIO)
Eye Hospital of Ho Chi Minh City
Hanoi Eye Hospital
Allergan Vietnam
Eyenovia Vietnam
Table of Contents
Market Assessment Phase
1. Executive Summary and Approach
2. Vietnam Ophthalmic Clinical Trials Market Overview
2.1 Key Insights and Strategic Recommendations
2.2 Vietnam Ophthalmic Clinical Trials Market Overview
2.3 Definition and Scope
2.4 Evolution of Market Ecosystem
2.5 Timeline of Key Regulatory Milestones
2.6 Value Chain & Stakeholder Mapping
2.7 Business Cycle Analysis
2.8 Policy & Incentive Landscape
3. Vietnam Ophthalmic Clinical Trials Market Analysis
3.1 Growth Drivers
3.1.1 Increasing prevalence of eye diseases
3.1.2 Rising investment in healthcare infrastructure
3.1.3 Growing demand for innovative ophthalmic treatments
3.1.4 Supportive government policies for clinical research
3.2 Market Challenges
3.2.1 Limited awareness of clinical trials among patients
3.2.2 Regulatory hurdles and lengthy approval processes
3.2.3 Competition from established markets
3.2.4 Shortage of skilled professionals in ophthalmology
3.3 Market Opportunities
3.3.1 Expansion of telemedicine in ophthalmology
3.3.2 Collaborations with international research organizations
3.3.3 Development of personalized medicine approaches
3.3.4 Increasing focus on pediatric ophthalmology
3.4 Market Trends
3.4.1 Adoption of digital health technologies
3.4.2 Growth of patient-centric trial designs
3.4.3 Emphasis on real-world evidence in clinical trials
3.4.4 Rising interest in gene therapy for eye diseases
3.5 Government Regulation
3.5.1 Streamlined approval processes for clinical trials
3.5.2 Enhanced ethical guidelines for patient recruitment
3.5.3 Increased funding for ophthalmic research initiatives
3.5.4 Collaboration with international regulatory bodies
4. SWOT Analysis
5. Stakeholder Analysis
6. Porter's Five Forces Analysis
7. Vietnam Ophthalmic Clinical Trials Market Market Size, 2019-2024
7.1 By Value
7.2 By Volume
7.3 By Average Selling Price
8. Vietnam Ophthalmic Clinical Trials Market Segmentation
8.1 By Type
8.1.1 Phase I Trials
8.1.2 Phase II Trials
8.1.3 Phase III Trials
8.1.4 Phase IV Trials
8.1.5 Investigator-Initiated Trials
8.2 By End-User
8.2.1 Hospitals & Eye Hospitals
8.2.2 Academic & Research Institutions
8.2.3 Pharmaceutical & Biotechnology Companies
8.2.4 Contract Research Organizations (CROs)
8.2.5 Government & Public Health Agencies
8.3 By Therapeutic Area
8.3.1 Glaucoma
8.3.2 Cataract
8.3.3 Retinal Disorders (including Diabetic Retinopathy, AMD)
8.3.4 Corneal Diseases
8.3.5 Refractive Errors
8.3.6 Uveitis & Inflammatory Diseases
8.3.7 Others
8.4 By Study Design
8.4.1 Randomized Controlled Trials (RCTs)
8.4.2 Observational Studies
8.4.3 Open-Label Studies
8.4.4 Single-Arm Trials
8.4.5 Others
8.5 By Patient Demographics
8.5.1 Adults (18-64 years)
8.5.2 Pediatrics (0-17 years)
8.5.3 Geriatrics (65+ years)
8.5.4 Others
8.6 By Funding Source
8.6.1 Government Funding
8.6.2 Private/Industry Funding
8.6.3 International Organizations & NGOs
8.6.4 Academic Grants
8.6.5 Others
8.7 By Geographic Focus
8.7.1 Urban Areas (e.g., Ho Chi Minh City, Hanoi, Da Nang)
8.7.2 Rural Areas
8.7.3 Others
9. Vietnam Ophthalmic Clinical Trials Market Competitive Analysis
9.1 Market Share of Key Players
9.2 Cross Comparison of Key Players
9.2.1 Company Name
9.2.2 Group Size (Large, Medium, or Small as per industry convention)
9.2.3 Number of Ophthalmic Clinical Trials Conducted in Vietnam
9.2.4 Clinical Trial Success Rate (Vietnam-specific)
9.2.5 Patient Recruitment Efficiency (Average Enrollment Time per Trial)
9.2.6 Average Trial Duration (Months)
9.2.7 Revenue from Ophthalmic Clinical Trials in Vietnam
9.2.8 Market Penetration Rate (Share of Total Ophthalmic Trials in Vietnam)
9.2.9 Regulatory Approval Lead Time (Average Days to Approval)
9.2.10 Adverse Event Reporting Rate
9.2.11 Investigator/Partner Network Size in Vietnam
9.2.12 Customer/Partner Satisfaction Index
9.3 SWOT Analysis of Top Players
9.4 Pricing Analysis
9.5 Detailed Profile of Major Companies
9.5.1 Novartis Vietnam
9.5.2 Roche Vietnam
9.5.3 Bayer Vietnam
9.5.4 Alcon Vietnam
9.5.5 Johnson & Johnson Vision Vietnam
9.5.6 Merck Sharp & Dohme (MSD) Vietnam
9.5.7 Santen Pharmaceutical Vietnam
9.5.8 Hoya Corporation Vietnam
9.5.9 Bausch + Lomb Vietnam
9.5.10 DND International Eye Hospital
9.5.11 Vietnam National Institute of Ophthalmology (VNIO)
9.5.12 Eye Hospital of Ho Chi Minh City
9.5.13 Hanoi Eye Hospital
9.5.14 Allergan Vietnam
9.5.15 Eyenovia Vietnam
10. Vietnam Ophthalmic Clinical Trials Market End-User Analysis
10.1 Procurement Behavior of Key Ministries
10.1.1 Ministry of Health
10.1.2 Ministry of Science and Technology
10.1.3 Ministry of Finance
10.1.4 Others
10.2 Corporate Spend on Infrastructure & Energy
10.2.1 Investment in Clinical Research Facilities
10.2.2 Funding for Technology Upgrades
10.2.3 Budget Allocation for Training Programs
10.2.4 Others
10.3 Pain Point Analysis by End-User Category
10.3.1 Hospitals
10.3.2 Research Institutions
10.3.3 Pharmaceutical Companies
10.3.4 Others
10.4 User Readiness for Adoption
10.4.1 Awareness of Clinical Trials
10.4.2 Accessibility of Information
10.4.3 Willingness to Participate
10.4.4 Others
10.5 Post-Deployment ROI and Use Case Expansion
10.5.1 Evaluation of Clinical Outcomes
10.5.2 Cost-Benefit Analysis
10.5.3 Potential for Future Trials
10.5.4 Others
11. Vietnam Ophthalmic Clinical Trials Market Future Size, 2025-2030
11.1 By Value
11.2 By Volume
11.3 By Average Selling Price
Go-To-Market Strategy Phase
1. Whitespace Analysis + Business Model Canvas
1.1 Market Gaps Identification
1.2 Business Model Development
2. Marketing and Positioning Recommendations
2.1 Branding Strategies
2.2 Product USPs
3. Distribution Plan
3.1 Urban Retail vs Rural NGO Tie-Ups
4. Channel & Pricing Gaps
4.1 Underserved Routes
4.2 Pricing Bands
5. Unmet Demand & Latent Needs
5.1 Category Gaps
5.2 Consumer Segments
6. Customer Relationship
6.1 Loyalty Programs
6.2 After-Sales Service
7. Value Proposition
7.1 Sustainability
7.2 Integrated Supply Chains
8. Key Activities
8.1 Regulatory Compliance
8.2 Branding
8.3 Distribution Setup
9. Entry Strategy Evaluation
9.1 Domestic Market Entry Strategy
9.1.1 Product Mix
9.1.2 Pricing Band
9.1.3 Packaging
9.2 Export Entry Strategy
9.2.1 Target Countries
9.2.2 Compliance Roadmap
10. Entry Mode Assessment
10.1 JV
10.2 Greenfield
10.3 M&A
10.4 Distributor Model
11. Capital and Timeline Estimation
11.1 Capital Requirements
11.2 Timelines
12. Control vs Risk Trade-Off
12.1 Ownership vs Partnerships
13. Profitability Outlook
13.1 Breakeven Analysis
13.2 Long-Term Sustainability
14. Potential Partner List
14.1 Distributors
14.2 JVs
14.3 Acquisition Targets
15. Execution Roadmap
15.1 Phased Plan for Market Entry
15.1.1 Market Setup
15.1.2 Market Entry
15.1.3 Growth Acceleration
15.1.4 Scale & Stabilize
15.2 Key Activities and Milestones
15.2.1 Milestone Planning
15.2.2 Activity Tracking
Research Methodology
Phase 1: Approach1
Desk Research
- Analysis of published reports from the Vietnam Ministry of Health regarding ophthalmic clinical trials
- Review of academic journals and articles focusing on ophthalmology and clinical research in Vietnam
- Examination of market reports from healthcare consulting firms detailing trends in ophthalmic treatments and trials
Primary Research
- Interviews with ophthalmologists and clinical trial coordinators in leading hospitals across Vietnam
- Surveys conducted with pharmaceutical companies involved in ophthalmic drug development
- Focus groups with patients participating in clinical trials to gather insights on their experiences and expectations
Validation & Triangulation
- Cross-validation of findings through multiple expert interviews and stakeholder feedback
- Triangulation of data from clinical trial registries and healthcare databases
- Sanity checks through expert panel reviews comprising industry veterans and regulatory experts
Phase 2: Market Size Estimation1
Top-down Assessment
- Estimation of the total addressable market for ophthalmic clinical trials based on national healthcare expenditure
- Segmentation of the market by therapeutic areas such as cataract, glaucoma, and retinal diseases
- Incorporation of government healthcare initiatives promoting clinical research in ophthalmology
Bottom-up Modeling
- Collection of data on the number of ongoing and completed ophthalmic clinical trials from local registries
- Estimation of trial costs based on historical data from previous studies and current market rates
- Volume x cost analysis to derive revenue potential for each segment of the ophthalmic trials market
Forecasting & Scenario Analysis
- Multi-factor regression analysis incorporating factors such as population aging and increasing prevalence of eye diseases
- Scenario modeling based on potential regulatory changes and advancements in ophthalmic technologies
- Development of baseline, optimistic, and pessimistic forecasts through 2030
Phase 3: CATI Sample Composition1
| Scope Item/Segment | Sample Size | Target Respondent Profiles |
|---|---|---|
| Cataract Clinical Trials | 100 | Ophthalmologists, Clinical Trial Managers |
| Glaucoma Treatment Studies | 60 | Research Scientists, Regulatory Affairs Specialists |
| Retinal Disease Trials | 50 | Clinical Research Coordinators, Patient Advocates |
| Innovative Ophthalmic Drug Development | 70 | Pharmaceutical Executives, Medical Affairs Managers |
| Patient Experience in Trials | 40 | Trial Participants, Healthcare Providers |
Frequently Asked Questions
What is the current value of the Vietnam Ophthalmic Clinical Trials Market?
The Vietnam Ophthalmic Clinical Trials Market is valued at approximately USD 470 million, reflecting significant growth driven by the increasing prevalence of eye diseases and advancements in ophthalmic drug formulations.