Region:Asia
Author(s):Geetanshi
Product Code:KRAE0720
Pages:93
Published On:December 2025
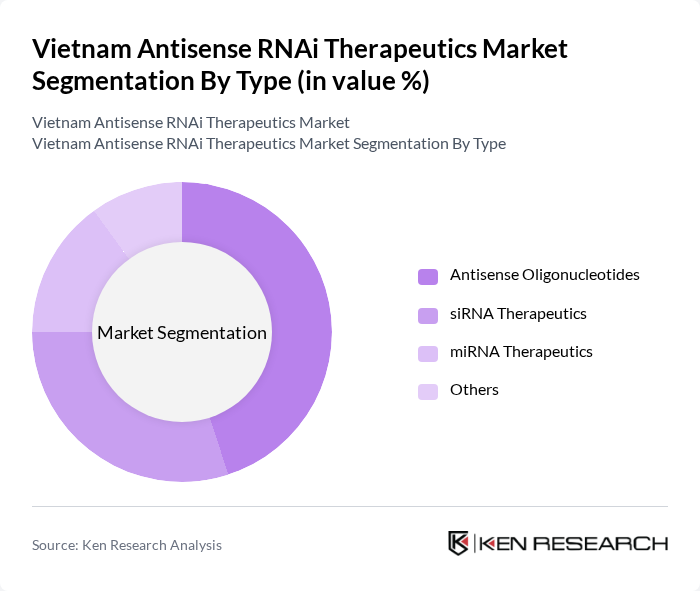
By Type:The market is segmented into various types of therapeutics, including Antisense Oligonucleotides, siRNA Therapeutics, miRNA Therapeutics, and Others. Among these, Antisense Oligonucleotides are currently leading the market due to their established efficacy in treating genetic disorders and their growing application in oncology. The increasing focus on personalized medicine and targeted therapies is further propelling the demand for this segment.
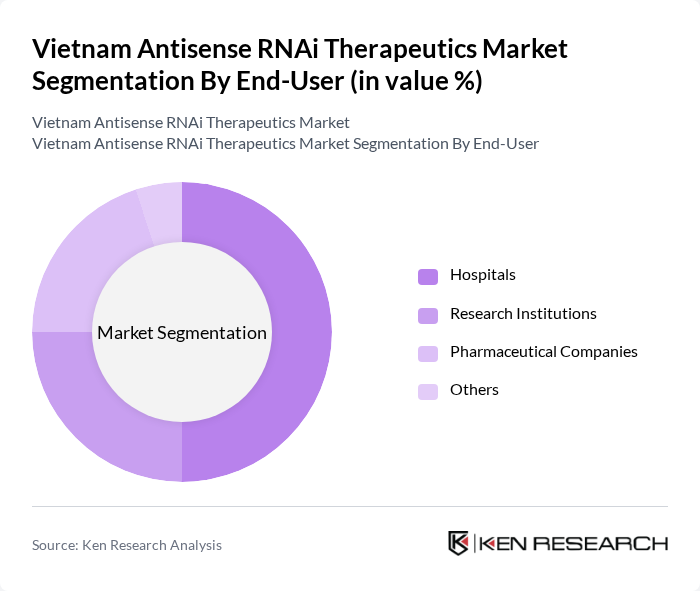
By End-User:The end-user segmentation includes Hospitals, Research Institutions, Pharmaceutical Companies, and Others. Hospitals are the dominant end-user in the market, driven by the increasing number of patients requiring advanced therapeutic options for chronic diseases. The growing adoption of innovative treatment protocols in hospitals is significantly contributing to the expansion of this segment.
The Vietnam Antisense RNAi Therapeutics Market is characterized by a dynamic mix of regional and international players. Leading participants such as Roche, Alnylam Pharmaceuticals, Ionis Pharmaceuticals, Moderna, Inc., Silence Therapeutics, Arrowhead Pharmaceuticals, Dicerna Pharmaceuticals, Regulus Therapeutics, Wave Life Sciences, Codiak BioSciences, Avidity Biosciences, Antisense Therapeutics, Genevant Sciences, N-Lorem Foundation, Eton Pharmaceuticals contribute to innovation, geographic expansion, and service delivery in this space.
The future of the antisense RNAi therapeutics market in Vietnam appears promising, driven by ongoing advancements in precision medicine and increasing collaboration between biotech firms and academic institutions. As the healthcare landscape evolves, the demand for targeted therapies is expected to rise, leading to more innovative solutions. Additionally, the integration of artificial intelligence in drug discovery processes is likely to enhance the efficiency of developing RNAi therapeutics, paving the way for breakthroughs in treatment options for genetic disorders and cancers.
| Segment | Sub-Segments |
|---|---|
| By Type | Antisense Oligonucleotides siRNA Therapeutics miRNA Therapeutics Others |
| By End-User | Hospitals Research Institutions Pharmaceutical Companies Others |
| By Application | Cancer Treatment Cardiovascular Diseases Infectious Diseases Others |
| By Delivery Method | Lipid Nanoparticles Viral Vectors Electroporation Others |
| By Distribution Channel | Direct Sales Online Sales Distributors Others |
| By Region | Northern Vietnam Southern Vietnam Central Vietnam Others |
| By Research Phase | Preclinical Clinical Trials Marketed Products Others |
| Scope Item/Segment | Sample Size | Target Respondent Profiles |
|---|---|---|
| Pharmaceutical Companies in RNAi | 100 | R&D Directors, Product Managers |
| Healthcare Professionals | 80 | Oncologists, Geneticists |
| Regulatory Bodies | 50 | Regulatory Affairs Specialists, Policy Makers |
| Academic Researchers | 70 | University Professors, Research Scientists |
| Biotechnology Investors | 60 | Venture Capitalists, Investment Analysts |
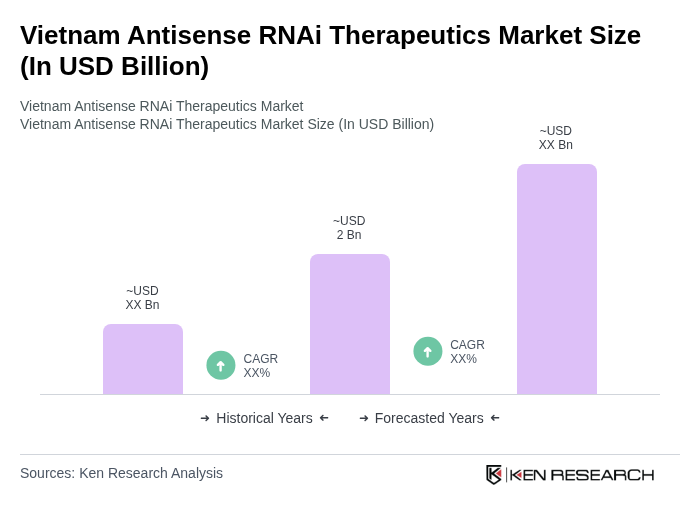
The Vietnam Antisense RNAi Therapeutics Market is valued at approximately USD 2 billion, reflecting significant growth driven by the rising demand for biologics and advanced therapies, particularly in response to the increasing prevalence of chronic diseases.