Region:Asia
Author(s):Dev
Product Code:KRAC1326
Pages:99
Published On:December 2025
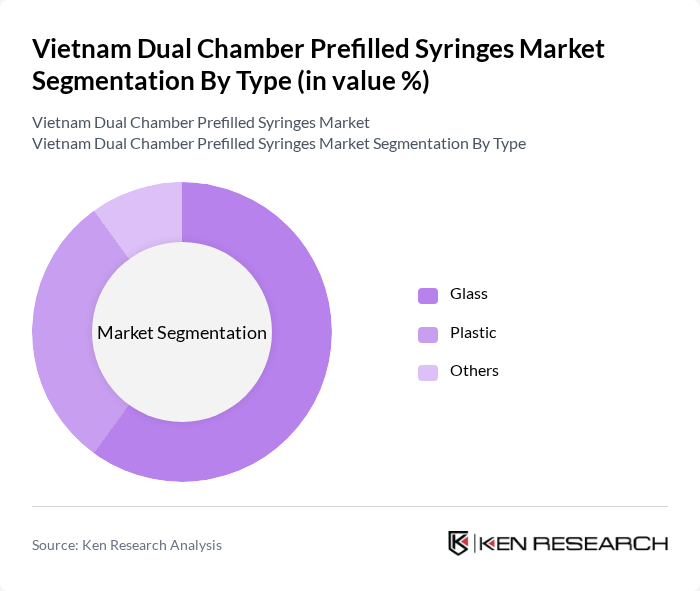
By Type:The dual chamber prefilled syringes market is segmented into glass, plastic, and others. Glass syringes dominate the market due to their inert nature, which ensures the stability of sensitive biologics. The preference for glass is driven by its ability to maintain the integrity of the drug, especially in the case of biologics that require precise dosing and stability. Plastic syringes are gaining traction due to their lightweight and shatterproof characteristics, making them suitable for various applications.
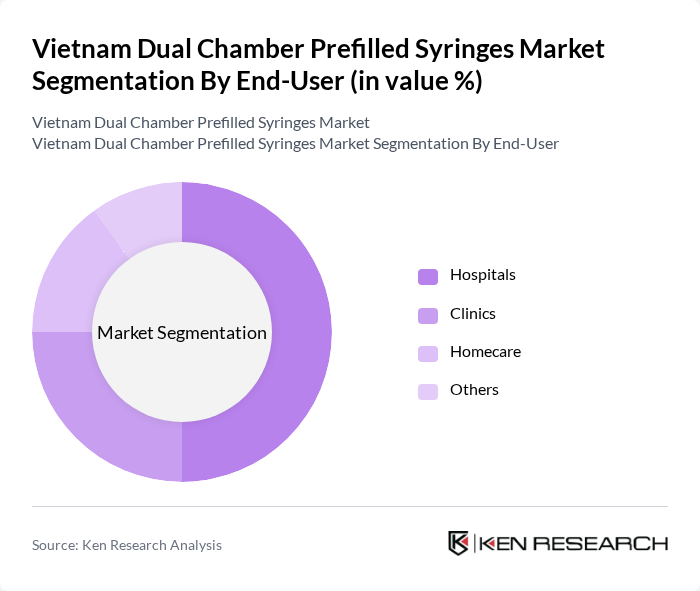
By End-User:The end-user segmentation includes hospitals, clinics, homecare, and others. Hospitals are the leading end-users of dual chamber prefilled syringes, primarily due to their high patient turnover and the need for efficient drug delivery systems. The increasing number of outpatient procedures and the growing trend of home healthcare are also contributing to the rise in demand from clinics and homecare settings, as patients prefer convenient and safe administration methods.
The Vietnam Dual Chamber Prefilled Syringes Market is characterized by a dynamic mix of regional and international players. Leading participants such as Becton Dickinson, Gerresheimer AG, Schott AG, West Pharmaceutical Services, and Terumo contribute to innovation, geographic expansion, and service delivery in this space.
The future of the Vietnam dual chamber prefilled syringes market appears promising, driven by increasing healthcare demands stemming from an aging population and rising chronic disease prevalence. The adoption of digital regulatory frameworks, such as the Common Submission Dossier Template (CSDT) in future, will streamline market entry processes, enhancing the speed of product launches. Additionally, the focus on sustainability and innovation in medical devices will likely shape the market landscape, encouraging the development of eco-friendly solutions tailored to local needs.
| Segment | Sub-Segments |
|---|---|
| By Type | Glass Plastic Others |
| By End-User | Hospitals Clinics Homecare Others |
| By Therapeutic Area | Oncology Autoimmune Diseases Infectious Diseases Others |
| By Distribution Channel | Direct Sales Distributors Online Sales Others |
| By Region | Northern Vietnam Southern Vietnam Central Vietnam Others |
| By Packaging Type | Single-use Multi-use Others |
| By Product Configuration | Fixed-dose Variable-dose Others |
| Scope Item/Segment | Sample Size | Target Respondent Profiles |
|---|---|---|
| Hospital Administration | 100 | Hospital Managers, Procurement Officers |
| Pharmacy Sector | 80 | Pharmacists, Pharmacy Managers |
| Healthcare Professionals | 120 | Doctors, Nurses, Medical Technicians |
| Manufacturers of Prefilled Syringes | 60 | Product Managers, R&D Heads |
| Regulatory Bodies | 50 | Regulatory Affairs Specialists, Compliance Officers |
The Vietnam Dual Chamber Prefilled Syringes Market is valued at approximately USD 40 million, reflecting a significant growth trend driven by increased healthcare awareness and the rising prevalence of chronic diseases.