Region:Asia
Author(s):Dev
Product Code:KRAE0098
Pages:82
Published On:December 2025
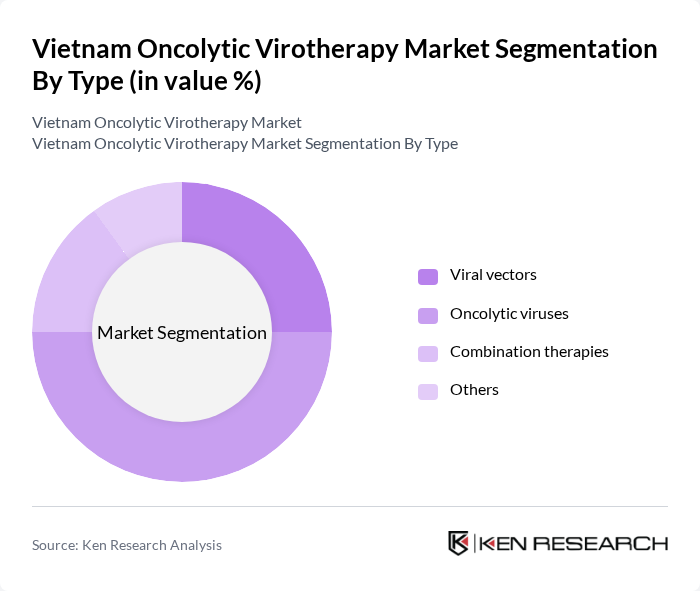
By Type:The oncolytic virotherapy market is segmented into various types, including viral vectors, oncolytic viruses, combination therapies, and others. Among these, oncolytic viruses are currently dominating the market due to their ability to selectively infect and kill cancer cells while stimulating an immune response. This effectiveness has led to increased adoption in clinical settings, making it a preferred choice for oncologists and researchers alike.
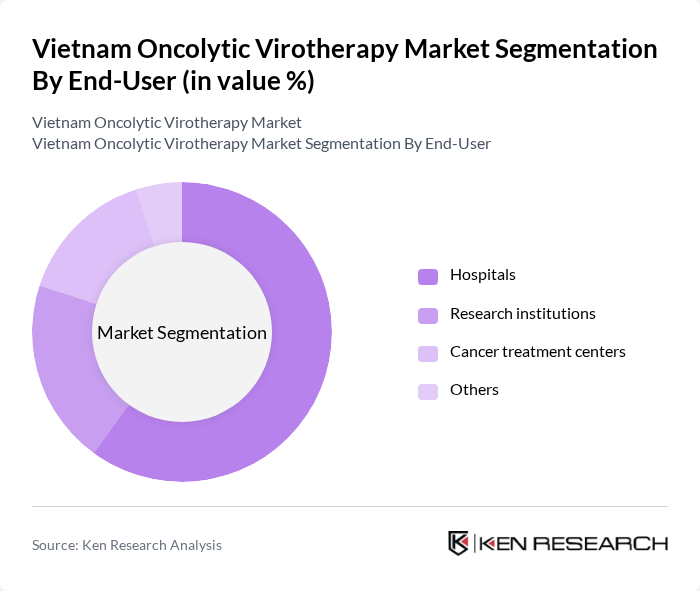
By End-User:The end-user segmentation includes hospitals, research institutions, cancer treatment centers, and others. Hospitals are the leading end-users in the oncolytic virotherapy market, primarily due to their capacity to provide comprehensive cancer care and access to advanced treatment options. The integration of virotherapy into standard treatment protocols in hospitals has significantly contributed to its market dominance.
The Vietnam Oncolytic Virotherapy Market is characterized by a dynamic mix of regional and international players. Leading participants such as Oncolytics Biotech Inc., Amgen Inc., Merck & Co., Inc., AstraZeneca, Genentech, Inc., Pfizer Inc., Novartis AG, GSK (GlaxoSmithKline), Takeda Pharmaceutical Company, Eli Lilly and Company, Regeneron Pharmaceuticals, Inc., BioNTech SE, Inovio Pharmaceuticals, Inc., Celyad Oncology SA, Sorrento Therapeutics, Inc. contribute to innovation, geographic expansion, and service delivery in this space.
The future of the oncolytic virotherapy market in Vietnam appears promising, driven by increasing cancer incidence and technological advancements. As the government enhances funding for cancer research and development, the market is likely to see a surge in innovative therapies. Additionally, the integration of telemedicine and digital health technologies will facilitate patient access to these treatments, improving overall healthcare delivery. The focus on personalized medicine will further tailor therapies to individual patient needs, enhancing treatment efficacy and patient satisfaction.
| Segment | Sub-Segments |
|---|---|
| By Type | Viral vectors Oncolytic viruses Combination therapies Others |
| By End-User | Hospitals Research institutions Cancer treatment centers Others |
| By Cancer Type | Breast cancer Lung cancer Melanoma Others |
| By Administration Route | Intravenous Intratumoral Oral Others |
| By Distribution Channel | Direct sales Online platforms Distributors Others |
| By Region | Northern Vietnam Southern Vietnam Central Vietnam Others |
| By Research Phase | Preclinical Clinical trials Post-marketing surveillance Others |
| Scope Item/Segment | Sample Size | Target Respondent Profiles |
|---|---|---|
| Oncologists in Major Hospitals | 100 | Medical Oncologists, Radiation Oncologists |
| Healthcare Administrators | 80 | Hospital Administrators, Oncology Department Heads |
| Patients Undergoing Cancer Treatment | 150 | Cancer Patients, Caregivers |
| Pharmaceutical Representatives | 70 | Sales Representatives, Product Managers |
| Regulatory Experts | 50 | Health Policy Analysts, Regulatory Affairs Specialists |
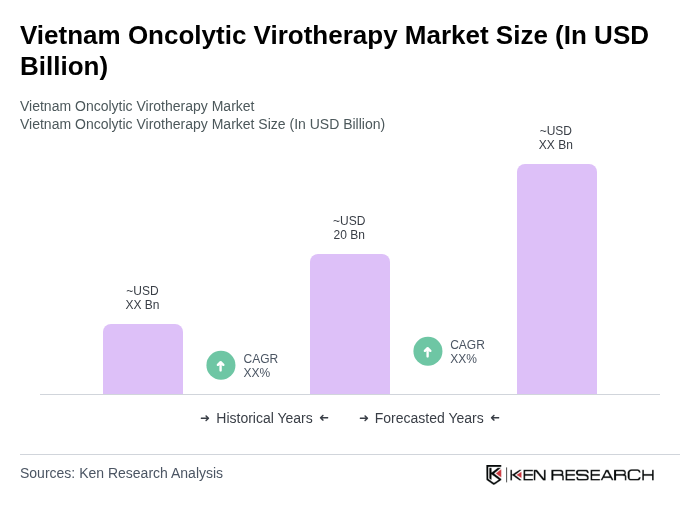
The Vietnam Oncolytic Virotherapy Market is valued at approximately USD 20 billion, reflecting significant growth driven by increasing cancer prevalence, active clinical trials, and advancements in virotherapy technologies.