About the Report
Base Year 2024Listen to the audio summary
Vietnam Opioid Market Overview
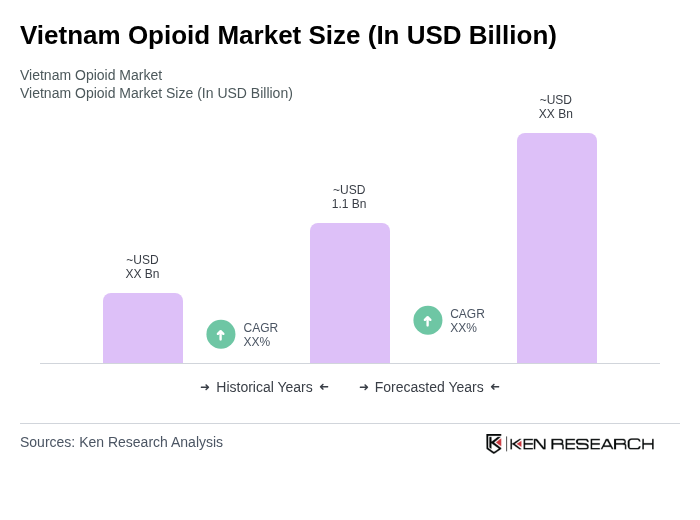
- The Vietnam Opioid Market is valued at USD 1.1 billion, based on a five-year historical analysis of prescription drugs and controlled medicines within the broader Vietnam pharmaceuticals market, which itself has been valued in the mid?single?digit billion range in recent years. This growth is primarily driven by the increasing prevalence of chronic pain conditions and cancer, alongside rising demand for effective pain management solutions and controlled analgesics. The market is also supported by expanding healthcare infrastructure, greater access to hospital?based care, and growing use of prescription medicines in clinical settings, particularly in urban and tertiary hospitals.
- Key cities dominating the market include Ho Chi Minh City and Hanoi, which are the largest urban centers in Vietnam and act as primary hubs for the country’s pharmaceutical and hospital markets. These cities have a higher concentration of central and provincial hospitals, oncology centers, and private clinics, creating stronger demand for pain management therapies, including opioids, and ensuring higher consumption of prescription medicines. Additionally, the presence of leading domestic manufacturers, importers, and distributors in southern Vietnam and Hanoi supports robust supply chains and enhances market growth for controlled drugs.
- Opioid prescription, distribution, and use in Vietnam are governed by a strict controlled?substances framework, particularly Decree No. 105/2021/ND-CP on management of narcotic substances and precursors issued by the Government of Vietnam, which details licensing, storage, record?keeping, and reporting obligations for medical facilities handling narcotic medicines including opioids. The regulation requires prescribing physicians and dispensing facilities to comply with standardized prescription forms, quantity limits, and documentation procedures and mandates monitoring of narcotic drug imports, distribution, and use through centralized reporting managed by competent health and drug?control authorities. In parallel, Ministry of Health circulars on narcotic and psychotropic medicines provide detailed professional and training requirements for prescribers and pharmacists involved in opioid?based pain management and substitution therapy.
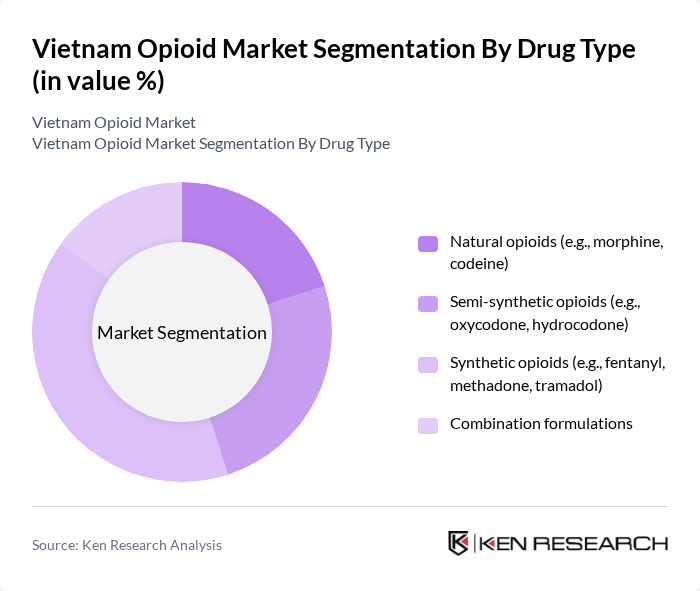
Vietnam Opioid Market Segmentation
By Drug Type:The opioid market can be segmented into four main drug types: natural opioids, semi-synthetic opioids, synthetic opioids, and combination formulations. Natural opioids, such as morphine and codeine, are derived from the opium poppy and are widely used for moderate to severe pain relief, especially in hospital and oncology settings. Semi-synthetic opioids, including oxycodone and hydrocodone, are modified versions of natural opioids and are commonly used internationally for moderate to severe pain, though in Vietnam their use is more restricted and concentrated in hospital settings where stronger analgesia is needed. Synthetic opioids, like fentanyl, methadone, and tramadol, are entirely man-made and are known for their potency and flexibility in dosage forms; tramadol and fentanyl are important components of pain management, while methadone is central to opioid substitution therapy programs in Vietnam. Combination formulations typically include a mix of weak opioids such as codeine or tramadol with non-opioid analgesics (for example, paracetamol) to enhance pain relief for acute and moderate pain while aiming to limit opioid dose. Within this structure, synthetic opioids and tramadol?based products have seen particularly strong uptake due to their clinical effectiveness, availability in multiple dosage strengths, and their use across both hospital and outpatient settings.
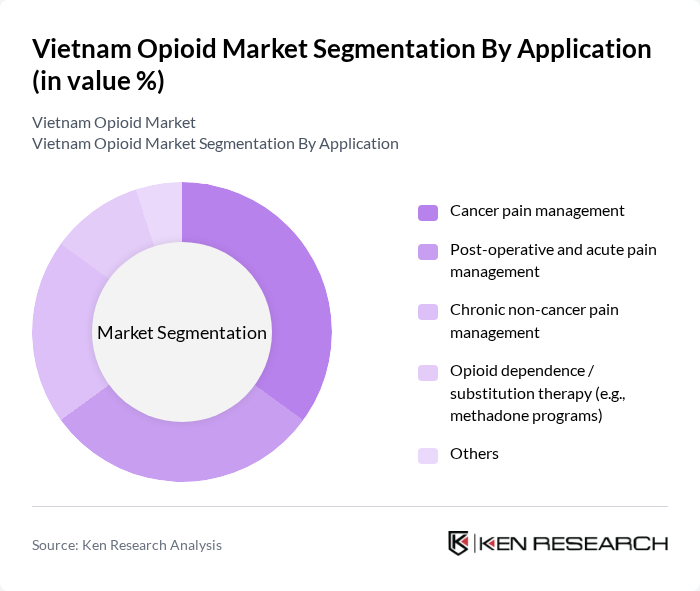
By Application:The opioid market is segmented based on applications, including cancer pain management, post-operative and acute pain management, chronic non-cancer pain management, opioid dependence/substitution therapy, and others. Cancer pain management is a significant segment due to the increasing incidence and prevalence of cancer in Vietnam and the emphasis on integrating morphine and other strong opioids into palliative and end?of?life care. Post-operative and acute pain management is also crucial, as surgical volumes are rising with the expansion of public and private hospital capacity and wider access to elective procedures, necessitating short?term opioid use in inpatient settings. Chronic non-cancer pain management is gaining traction as awareness of musculoskeletal disorders, neuropathic pain, and other long-term conditions increases, although guidelines promote careful benefit–risk assessment and use of multimodal pain strategies. Opioid dependence/substitution therapy, particularly through methadone maintenance treatment programs supported by the Ministry of Health and international partners, plays a central role in addressing heroin and other opioid use disorders and has been scaled up across many provinces. Currently, cancer pain management and hospital-based acute pain remain core application areas in terms of opioid volume, supported by the growing cancer patient population and continued strengthening of tertiary care services.
Vietnam Opioid Market Competitive Landscape
The Vietnam Opioid Market is characterized by a dynamic mix of regional and international players. Leading participants such as Vietnam Pharmaceutical Corporation (VINAPHARM), DHG Pharmaceutical JSC (Hau Giang Pharmaceutical), Domesco Medical Import-Export JSC, Traphaco JSC, Bidiphar JSC (Binh Dinh Pharmaceutical), Central Pharmaceutical Company No. 1 (CPC1), Vimedimex Medi-Pharma JSC, S.P.M JSC (SPM Pharmaceutical), Imexpharm Corporation, OPC Pharmaceutical JSC, Mekophar Chemical Pharmaceutical JSC, Pymepharco JSC, Savipharm JSC, Boston Pharma JSC, Bepharco (Ben Tre Pharmaceutical JSC) contribute to innovation, geographic expansion, and service delivery in this space.
Vietnam Opioid Market Industry Analysis
Growth Drivers
- Increasing Prevalence of Chronic Pain:The Vietnam healthcare system reports that approximately 30% of the population suffers from chronic pain conditions, translating to around 26 million individuals. This rising prevalence is driving the demand for effective pain management solutions, including opioids. The World Health Organization estimates that chronic pain affects about 20% of adults globally, indicating a significant need for opioid therapies in Vietnam, particularly as the population ages and lifestyle diseases become more common.
- Rising Demand for Palliative Care:Vietnam's aging population, projected to reach 20% in the coming years, is increasing the demand for palliative care services. In future, the Ministry of Health aims to expand palliative care programs, which currently serve only 5% of patients in need. This expansion is crucial as it aligns with the global trend of integrating opioids into palliative care, ensuring that patients receive adequate pain relief during end-of-life care, thus driving opioid market growth.
- Expanding Healthcare Infrastructure:Vietnam's healthcare expenditure is expected to reach $22 billion in future, reflecting a 10% increase from previous years. This investment is aimed at enhancing healthcare facilities and services, particularly in urban areas. Improved access to healthcare services facilitates better pain management practices, including opioid prescriptions. The government’s commitment to healthcare infrastructure development is essential for meeting the growing demand for opioid therapies in the country.
Market Challenges
- Regulatory Restrictions on Opioid Prescriptions:Vietnam has stringent regulations governing opioid prescriptions, with only 0.5% of patients receiving opioids for chronic pain management. These regulations are primarily due to concerns over misuse and addiction. The Ministry of Health is working to balance patient needs with regulatory frameworks, but the current restrictions hinder access to necessary pain relief for many patients, limiting market growth potential.
- Stigma Associated with Opioid Use:There is a significant stigma surrounding opioid use in Vietnam, with many healthcare providers and patients fearing addiction. This stigma results in under-prescription of opioids, with only 12% of healthcare professionals feeling comfortable prescribing them for chronic pain. Educational initiatives are needed to address misconceptions and promote responsible opioid use, as overcoming this stigma is crucial for expanding the market and improving patient outcomes.
Vietnam Opioid Market Future Outlook
The Vietnam opioid market is poised for transformation as healthcare policies evolve to address pain management more effectively. With a focus on integrating technology and personalized medicine, the market is likely to see innovative opioid formulations and telemedicine solutions emerge in future. Additionally, increased collaboration between healthcare providers and educational initiatives will enhance awareness of pain management options, ultimately improving patient access to necessary treatments and fostering a more supportive environment for opioid use.
Market Opportunities
- Development of New Opioid Formulations:There is a significant opportunity for pharmaceutical companies to develop new opioid formulations that minimize addiction risks while providing effective pain relief. Innovations such as abuse-deterrent formulations can cater to the growing demand for safer pain management options, potentially increasing market share and improving patient outcomes.
- Expansion of Telemedicine for Pain Management:The rise of telemedicine presents a unique opportunity to enhance access to pain management services, particularly in rural areas. By leveraging technology, healthcare providers can offer consultations and prescriptions remotely, addressing the barriers posed by limited access to healthcare facilities and improving patient adherence to pain management regimens.
Scope of the Report
| Segment | Sub-Segments |
|---|---|
| By Drug Type | Natural opioids (e.g., morphine, codeine) Semi-synthetic opioids (e.g., oxycodone, hydrocodone) Synthetic opioids (e.g., fentanyl, methadone, tramadol) Combination formulations |
| By Application | Cancer pain management Post-operative and acute pain management Chronic non-cancer pain management Opioid dependence / substitution therapy (e.g., methadone programs) Others |
| By Route of Administration | Oral Injectable Transdermal Others |
| By End-User | Public hospitals Private hospitals Specialized pain and oncology clinics Substance use treatment centers / methadone clinics Others |
| By Distribution Channel | Hospital pharmacies Retail pharmacies / drug stores Online / e-pharmacies Government and NGO procurement programs Others |
| By Patient Demographics | Adults Geriatric Pediatric Others |
Key Target Audience
Investors and Venture Capitalist Firms
Government and Regulatory Bodies (e.g., Ministry of Health, Ministry of Public Security)
Manufacturers and Producers
Distributors and Retailers
Pharmaceutical Wholesalers
Healthcare Providers and Institutions
Non-Governmental Organizations (NGOs) focused on drug policy
Insurance Companies and Health Plans
Players Mentioned in the Report:
Vietnam Pharmaceutical Corporation (VINAPHARM)
DHG Pharmaceutical JSC (Hau Giang Pharmaceutical)
Domesco Medical Import-Export JSC
Traphaco JSC
Bidiphar JSC (Binh Dinh Pharmaceutical)
Central Pharmaceutical Company No. 1 (CPC1)
Vimedimex Medi-Pharma JSC
S.P.M JSC (SPM Pharmaceutical)
Imexpharm Corporation
OPC Pharmaceutical JSC
Mekophar Chemical Pharmaceutical JSC
Pymepharco JSC
Savipharm JSC
Boston Pharma JSC
Bepharco (Ben Tre Pharmaceutical JSC)
Table of Contents
Market Assessment Phase
1. Executive Summary and Approach
2. Vietnam Opioid Market Overview
2.1 Key Insights and Strategic Recommendations
2.2 Vietnam Opioid Market Overview
2.3 Definition and Scope
2.4 Evolution of Market Ecosystem
2.5 Timeline of Key Regulatory Milestones
2.6 Value Chain & Stakeholder Mapping
2.7 Business Cycle Analysis
2.8 Policy & Incentive Landscape
3. Vietnam Opioid Market Analysis
3.1 Growth Drivers
3.1.1 Increasing prevalence of chronic pain
3.1.2 Rising demand for palliative care
3.1.3 Expanding healthcare infrastructure
3.1.4 Government initiatives for pain management
3.2 Market Challenges
3.2.1 Regulatory restrictions on opioid prescriptions
3.2.2 Stigma associated with opioid use
3.2.3 Risk of addiction and misuse
3.2.4 Limited access in rural areas
3.3 Market Opportunities
3.3.1 Development of new opioid formulations
3.3.2 Expansion of telemedicine for pain management
3.3.3 Partnerships with healthcare providers
3.3.4 Increasing awareness and education on pain management
3.4 Market Trends
3.4.1 Shift towards personalized medicine
3.4.2 Growth of non-opioid alternatives
3.4.3 Integration of technology in pain management
3.4.4 Focus on patient-centered care
3.5 Government Regulation
3.5.1 Prescription monitoring programs
3.5.2 Guidelines for opioid prescribing
3.5.3 Restrictions on opioid manufacturing
3.5.4 Education and training requirements for prescribers
4. SWOT Analysis
5. Stakeholder Analysis
6. Porter's Five Forces Analysis
7. Vietnam Opioid Market Market Size, 2019-2024
7.1 By Value
7.2 By Volume
7.3 By Average Selling Price
8. Vietnam Opioid Market Segmentation
8.1 By Drug Type
8.1.1 Natural opioids (e.g., morphine, codeine)
8.1.2 Semi-synthetic opioids (e.g., oxycodone, hydrocodone)
8.1.3 Synthetic opioids (e.g., fentanyl, methadone, tramadol)
8.1.4 Combination formulations
8.2 By Application
8.2.1 Cancer pain management
8.2.2 Post-operative and acute pain management
8.2.3 Chronic non-cancer pain management
8.2.4 Opioid dependence / substitution therapy (e.g., methadone programs)
8.2.5 Others
8.3 By Route of Administration
8.3.1 Oral
8.3.2 Injectable
8.3.3 Transdermal
8.3.4 Others
8.4 By End-User
8.4.1 Public hospitals
8.4.2 Private hospitals
8.4.3 Specialized pain and oncology clinics
8.4.4 Substance use treatment centers / methadone clinics
8.4.5 Others
8.5 By Distribution Channel
8.5.1 Hospital pharmacies
8.5.2 Retail pharmacies / drug stores
8.5.3 Online / e-pharmacies
8.5.4 Government and NGO procurement programs
8.5.5 Others
8.6 By Patient Demographics
8.6.1 Adults
8.6.2 Geriatric
8.6.3 Pediatric
8.6.4 Others
9. Vietnam Opioid Market Competitive Analysis
9.1 Market Share of Key Players
9.2 Cross Comparison of Key Players
9.2.1 Company name
9.2.2 Ownership type (state-owned, private, multinational subsidiary)
9.2.3 Vietnam opioid portfolio breadth (number of opioid SKUs / molecules)
9.2.4 Opioid segment revenue (USD/VND) and 3–5 year CAGR
9.2.5 Share of opioids in total pharmaceutical revenue (%)
9.2.6 Hospital formulary coverage (% of Tier-1/central hospitals served)
9.2.7 Distribution reach (number of provinces / pharmacies covered)
9.2.8 Average tender win rate in public procurement (%)
9.2.9 Gross margin on key opioid products (%)
9.2.10 R&D and registration pipeline (number of new opioid / pain products)
9.2.11 Regulatory compliance indicators (GMP/GDP certifications, inspections passed)
9.2.12 Pharmacovigilance and safety performance (ADR incidence, recalls)
9.3 SWOT Analysis of Top Players
9.4 Pricing Analysis
9.5 Detailed Profile of Major Companies
9.5.1 Vietnam Pharmaceutical Corporation (VINAPHARM)
9.5.2 DHG Pharmaceutical JSC (Hau Giang Pharmaceutical)
9.5.3 Domesco Medical Import-Export JSC
9.5.4 Traphaco JSC
9.5.5 Bidiphar JSC (Binh Dinh Pharmaceutical)
9.5.6 Central Pharmaceutical Company No. 1 (CPC1)
9.5.7 Vimedimex Medi-Pharma JSC
9.5.8 S.P.M JSC (SPM Pharmaceutical)
9.5.9 Imexpharm Corporation
9.5.10 OPC Pharmaceutical JSC
9.5.11 Mekophar Chemical Pharmaceutical JSC
9.5.12 Pymepharco JSC
9.5.13 Savipharm JSC
9.5.14 Boston Pharma JSC
9.5.15 Bepharco (Ben Tre Pharmaceutical JSC)
10. Vietnam Opioid Market End-User Analysis
10.1 Procurement Behavior of Key Ministries
10.1.1 Ministry of Health
10.1.2 Ministry of Finance
10.1.3 Ministry of Labor, Invalids and Social Affairs
10.1.4 Others
10.2 Corporate Spend on Infrastructure & Energy
10.2.1 Healthcare infrastructure investments
10.2.2 Research and development funding
10.2.3 Training and education programs
10.2.4 Others
10.3 Pain Point Analysis by End-User Category
10.3.1 Hospitals
10.3.2 Clinics
10.3.3 Home healthcare providers
10.3.4 Others
10.4 User Readiness for Adoption
10.4.1 Awareness of opioid therapies
10.4.2 Training for healthcare professionals
10.4.3 Patient education initiatives
10.4.4 Others
10.5 Post-Deployment ROI and Use Case Expansion
10.5.1 Cost savings from effective pain management
10.5.2 Improved patient outcomes
10.5.3 Expansion into new therapeutic areas
10.5.4 Others
11. Vietnam Opioid Market Future Size, 2025-2030
11.1 By Value
11.2 By Volume
11.3 By Average Selling Price
Go-To-Market Strategy Phase
1. Whitespace Analysis + Business Model Canvas
1.1 Market Gaps Identification
1.2 Business Model Development
2. Marketing and Positioning Recommendations
2.1 Branding Strategies
2.2 Product USPs
3. Distribution Plan
3.1 Urban Retail vs Rural NGO Tie-ups
4. Channel & Pricing Gaps
4.1 Underserved Routes
4.2 Pricing Bands
5. Unmet Demand & Latent Needs
5.1 Category Gaps
5.2 Consumer Segments
6. Customer Relationship
6.1 Loyalty Programs
6.2 After-sales Service
7. Value Proposition
7.1 Sustainability
7.2 Integrated Supply Chains
8. Key Activities
8.1 Regulatory Compliance
8.2 Branding
8.3 Distribution Setup
9. Entry Strategy Evaluation
9.1 Domestic Market Entry Strategy
9.1.1 Product Mix
9.1.2 Pricing Band
9.1.3 Packaging
9.2 Export Entry Strategy
9.2.1 Target Countries
9.2.2 Compliance Roadmap
10. Entry Mode Assessment
10.1 JV
10.2 Greenfield
10.3 M&A
10.4 Distributor Model
11. Capital and Timeline Estimation
11.1 Capital Requirements
11.2 Timelines
12. Control vs Risk Trade-Off
12.1 Ownership vs Partnerships
13. Profitability Outlook
13.1 Breakeven Analysis
13.2 Long-term Sustainability
14. Potential Partner List
14.1 Distributors
14.2 JVs
14.3 Acquisition Targets
15. Execution Roadmap
15.1 Phased Plan for Market Entry
15.1.1 Market Setup
15.1.2 Market Entry
15.1.3 Growth Acceleration
15.1.4 Scale & Stabilize
15.2 Key Activities and Milestones
15.2.1 Milestone Planning
15.2.2 Activity Tracking
Research Methodology
Phase 1: Approach1
Desk Research
- Analysis of government reports on opioid usage and regulations in Vietnam
- Review of academic publications and white papers on opioid market trends
- Examination of industry reports from health organizations and NGOs focused on drug abuse
Primary Research
- Interviews with healthcare professionals, including pharmacists and doctors
- Surveys conducted with patients and caregivers regarding opioid usage and perceptions
- Focus groups with addiction specialists to understand treatment trends and challenges
Validation & Triangulation
- Cross-validation of findings with data from international health organizations
- Triangulation of qualitative insights from interviews with quantitative data from surveys
- Sanity checks through expert panel reviews comprising healthcare policymakers and economists
Phase 2: Market Size Estimation1
Top-down Assessment
- Estimation of total opioid consumption based on national health statistics
- Segmentation of market size by therapeutic categories and demographics
- Incorporation of government initiatives aimed at opioid regulation and control
Bottom-up Modeling
- Volume estimates derived from prescription data across major hospitals and clinics
- Cost analysis based on pricing structures from pharmaceutical distributors
- Calculation of market size using volume x price methodology for various opioid products
Forecasting & Scenario Analysis
- Multi-factor regression analysis incorporating economic indicators and healthcare access
- Scenario modeling based on potential changes in regulatory frameworks and public health campaigns
- Development of baseline, optimistic, and pessimistic forecasts through 2030
Phase 3: CATI Sample Composition1
| Scope Item/Segment | Sample Size | Target Respondent Profiles |
|---|---|---|
| Healthcare Providers | 120 | Doctors, Pharmacists, Nurses |
| Patients Using Opioids | 100 | Chronic Pain Patients, Post-Surgery Patients |
| Addiction Treatment Centers | 80 | Therapists, Counselors, Program Directors |
| Regulatory Bodies | 50 | Policy Makers, Health Officials |
| Pharmaceutical Distributors | 70 | Sales Managers, Supply Chain Coordinators |
Frequently Asked Questions
What is the current value of the Vietnam Opioid Market?
The Vietnam Opioid Market is valued at approximately USD 1.1 billion, driven by the increasing prevalence of chronic pain conditions and cancer, along with a rising demand for effective pain management solutions and controlled analgesics.