Region:Asia
Author(s):Rebecca
Product Code:KRAB2056
Pages:80
Published On:January 2026
By Molecule / Product Type:The market is segmented into various product types, including small-molecule injectables, biosimilar injectables, highly potent/cytotoxic injectables, long-acting depot injectables, complex formulations (liposomal, emulsions, etc.), and others. This structure aligns with the global specialty generics landscape, where injectables represent the largest revenue share within specialty generics due to rapid onset of action and suitability for complex, hospital-based therapies. Among these, small-molecule injectables dominate the market due to their widespread use in treating infections, peri?operative conditions, and chronic diseases, and because small-molecule generics account for the largest share and fastest growth within Vietnam’s generic drugs market overall. The increasing demand for affordable treatment options, expansion of hospital pharmacies, and wider physician and patient acceptance of parenteral generics contribute to the strong performance of this subsegment.
By Therapeutic Area:The therapeutic areas covered in the market include oncology, autoimmune & inflammatory diseases, infectious diseases, cardiometabolic disorders, central nervous system disorders, and others. Oncology is the leading therapeutic area, consistent with global specialty generics trends where oncology represents the largest share of specialty generic usage due to high prevalence, long treatment durations, and extensive reliance on injectable cytotoxic and targeted therapies. In Vietnam, cancer incidence has been rising alongside other chronic conditions, driving demand for cost-effective injectable chemotherapy and supportive?care generics in hospital settings. The growing focus on personalized medicine, wider adoption of biosimilar monoclonal antibodies, and expansion of oncology centers further enhances the demand for specialty injectables in this segment.
The Vietnam Specialty Injectable Generics Market is characterized by a dynamic mix of regional and international players. Leading participants such as DHG Pharmaceutical JSC (DHG Pharma), Traphaco JSC, Domesco Medical Import-Export JSC (Domesco), Hau Giang Pharmaceutical – Branches & subsidiaries, S.P.M JSC (SPM Pharmaceutical), Vimedimex Medi-Pharma JSC, Bidiphar JSC (Binh Dinh Pharmaceutical), Imexpharm Corporation, OPC Pharmaceutical JSC, Pymepharco JSC, Mekophar Chemical Pharmaceutical JSC, Sanofi Vietnam Co., Ltd., Pfizer (Vietnam) Co., Ltd., Novartis Vietnam Co., Ltd., GSK Vietnam Co., Ltd. contribute to innovation, geographic expansion, and service delivery in this space.
The future of the specialty injectable generics market in Vietnam appears promising, driven by increasing healthcare investments and a growing focus on chronic disease management. As the government continues to promote generics through policy initiatives, the market is likely to witness enhanced competition and innovation. Additionally, the integration of advanced manufacturing technologies will streamline production processes, further supporting the availability of affordable injectables. Overall, the market is poised for significant growth as healthcare access improves and patient awareness increases.
| Segment | Sub-Segments |
|---|---|
| By Molecule / Product Type | Small-molecule injectables Biosimilar injectables Highly potent / cytotoxic injectables Long-acting depot injectables Complex formulations (liposomal, emulsions, etc.) Others |
| By Therapeutic Area | Oncology Autoimmune & inflammatory diseases Infectious diseases Cardiometabolic disorders Central nervous system disorders Others |
| By Route of Administration | Intravenous (IV) Intramuscular (IM) Subcutaneous (SC) Intrathecal / intra-articular / others |
| By End-User | Public hospitals Private hospitals Specialty clinics & cancer centers Home- and community-based care Others |
| By Distribution Channel | Hospital tender / ETC channel Retail pharmacy / OTC channel Specialty pharmacies Direct sales to institutions Others |
| By Packaging Type | Vials Ampoules Pre-filled syringes & pens Cartridges Others |
| By Region | Northern Vietnam Central Vietnam Southern Vietnam Others |
| Scope Item/Segment | Sample Size | Target Respondent Profiles |
|---|---|---|
| Hospital Pharmacists | 100 | Pharmacy Directors, Clinical Pharmacists |
| Healthcare Providers | 80 | General Practitioners, Specialists |
| Generic Manufacturers | 70 | Product Managers, Business Development Executives |
| Regulatory Bodies | 50 | Regulatory Affairs Managers, Compliance Officers |
| Market Analysts | 40 | Market Research Analysts, Industry Consultants |
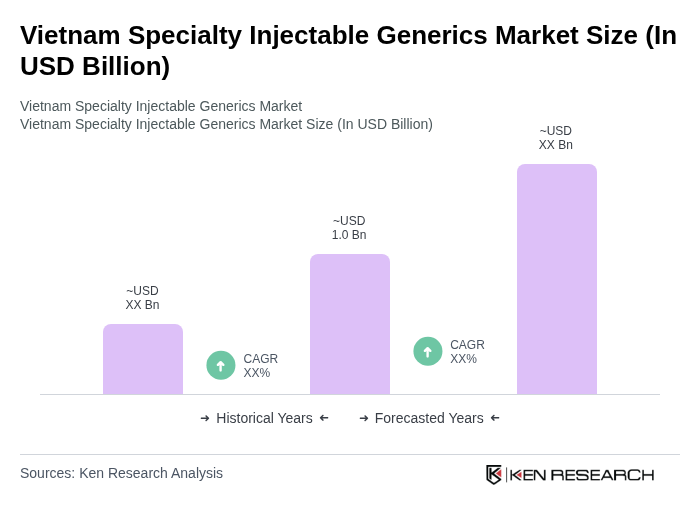
The Vietnam Specialty Injectable Generics Market is valued at approximately USD 1.0 billion, reflecting a significant growth driven by the increasing prevalence of chronic diseases and the demand for cost-effective treatment options.